 |
 |
 |
| |
Response to TMC114 is Based on Genotypic/Phenotypic Resistance: POWER 1/2/3 pooled analysis
|
| |
| |
Phenotypic and genotypic determinants of TMC114 (darunavir) resistance: POWER 1, 2 and 3 pooled analysis
Reported by Jules Levin
8th International Congress on Drug Therapy in HIV Infection, Glasgow, UK, 12-16 November 2006.
De Meyer S,1 Vangeneugden T,1 Lefebvre E,2 Van Marck H,1 Azijn H,1 De Baere I,1 Van Baelen B,1 de Béthune M-P1
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, PA, USA
The objective of this combined analysis was to investigate the influence of baseline geno- and phenotype on virological outcome to treatment with DRV/r
(600/100mg bid dose) in the POWER 1 and 2 trials and POWER 3 analysis.
AUTHOR CONCLUSIONS
Baseline DRV FC (fold-change) was a strong predictor of virological response to treatment with DRV/r
- by analysing the relationship between virological response and DRV FC,
FC CCOs of 10 and 40 were generated.
The virological response rate to DRV/r was reduced when there were >/=10 IAS-USA12 PI resistance-associated mutations.
A set of specific baseline mutations were identified that were associated with a diminished virological response to DRV/r at Week 24 in the POWER studies
- these DRV resistance-associated mutations were V11I, V32I, L33F, I47V, I50V, I54L/M, G73S, L76V, I84V and L89V
- importantly, they occurred in a background of multiple mutations (median of at least 10 PI resistance-associated mutations).14
In the POWER studies, the DRV resistance-associated mutation count provides a useful tool to predict virological response to DRV/r and is a better predictor of virological outcome than the number of IAS-USA12 PI resistance-associated mutations.
A decreased virological response to DRV/r was seen in patients with >/=3 DRV resistance-associated mutations at baseline in a background of other IAS-USA PI resistance-associated mutations; however, regardless of the type of baseline DRV resistance-associated mutation, the virological response to DRV/r remained higher than that observed with CPI(s).
The predictive value of the identified set of DRV resistance-associated mutations was confirmed by analysis of a panel of 9,668 HIV-1 clinical isolates where isolates with
These tools apply to the patient population studied in the POWER trials, and will be subject to change pending additional data in different patient populations (naïve [TMC114-C211 study] and early treatment-experienced [TMC114-C214 study]).
INTRODUCTION
The new HIV-1 protease inhibitor (PI), darunavir (DRV; TMC114), has significant
in-vitro antiretroviral (ARV) activity against both wild-type virus and multi-drug-resistant HIV-1 strains.1
DRV is co-administered with low-dose ritonavir (DRV/r) and DRV/r 600/100mg
bid is indicated for the treatment of HIV-1 infection in ARV treatmentexperienced
adult patients, such as those with HIV-1 strains resistant to more
than one PI.2
Patient data were combined in a pooled analysis of the randomised, controlled
POWER 1 and 2 (TMC114-C213 and C202) trials3-6 where treatment experienced, HIV-1-infected patients received DRV/r plus an optimized background regimen (OBR). At 48 weeks, 45% of patients receiving DRV/r 600/100mg bid achieved viral load (plasma HIV-1 RNA; VL) <50 copies/mL,
compared with 10% of patients receiving control PIs (CPI[s]) plus an OBR.7 DRV/r was generally well tolerated, with no dose-response relationship for
safety.7,8
Efficacy data from an additional 246 treatment-experienced patients initiating treatment at the DRV/r 600/100mg bid dose in two non-randomised, open-label
trials (POWER 3 [TMC114-C215 and C208]) showed that 40% of patients had
VL <50 copies/mL at Week 24.9 DRV/r was also generally well tolerated in this
analysis.
Methods
Design of POWER 1, 2 and 3
All the trials had a similar design in terms of inclusion/exclusion criteria and
patients' baseline characteristics were similar in all the trials.
Inclusion criteria for the POWER 1, 2 and 3 were:
- failing to respond to a PI-containing regimen at screening
- prior treatment with >/=2 NRTIs for at least 3 months, with >/=1 NNRTI in a failing regimen, and with 31 PI for at least 3 months
- plasma VL >1,000 copies/mL
- >/=1 primary PI mutation based on the IAS-USA guidelines from 2003 (POWER 1 and 2)10 or 2004 (POWER 3).11
In addition to DRV/r 600/100mg bid, patients received an OBR, which consisted of two or more NRTIs with or without enfuvirtide, and excluded NNRTIs. Selection of the OBR by the investigators was based on genotypic resistance testing (Virtual PhenotypeTM from Virco) and prior ARV treatment history.
The study protocols were reviewed and approved by the appropriate institutional ethics committee(s) and health authorities, and were conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients.
Analysis of POWER 1, 2 and 3
Analyses were performed on all patients who were always treated with DRV/r 600/100mg bid plus an OBR and reached Week 24 or discontinued earlier (n=377; cut-off date of 24 September 2005; ITT [intent-to-treat]) in POWER 1, 2 and 3.
Pheno- and genotyping of plasma viruses were determined by the Antivirogram® and virco®TYPE HIV-1 assays, respectively.
Linear modelling (ANCOVA [analysis of covariance]) was performed to assess the relationship between baseline DRV fold change (FC) in EC50 (50% effective concentration) and the Week 24 virological response (change in log10 VL, noncompleter= failure [NC=F]). Baseline DRV FC, use of enfuvirtide and baseline VL were covariates. Clinical cut-offs (CCOs) were defined using this model, specifically for the subgroup of patients not using enfuvirtide: inverse prediction was applied to estimate which FC (and its 95% CI [confidence interval]) corroborates with a decrease of 1 log10 VL versus baseline.
Week 24 analyses of the virological endpoints of change in VL, >/=1 log10 reduction in VL and VL <50 copies/mL, were performed on the ITT population
(NC=F and time to loss of virological response [TLOVR], respectively).
A panel of 9,668 HIV-1 clinical isolates that were submitted for routine resistance testing at Virco was used to check the predictive value of the DRV resistance-associated mutations identified in this study. These isolates included
screening samples from the POWER trials.
Results
Effect of baseline DRV FC on response to DRV/r and determination of
DRV CCO values
Baseline DRV FC was a significant predictor of virological response to DRV/r treatment (p<0.0001). Linear modelling of the relationship between DRV FC and response allowed two CCOs to be determined; these values were DRV FC of 10 and 40.
At baseline, 70% of viral isolates from patients had DRV FC 40. At Week 24, response to treatment varied according to DRV FC at baseline (Figure 1).
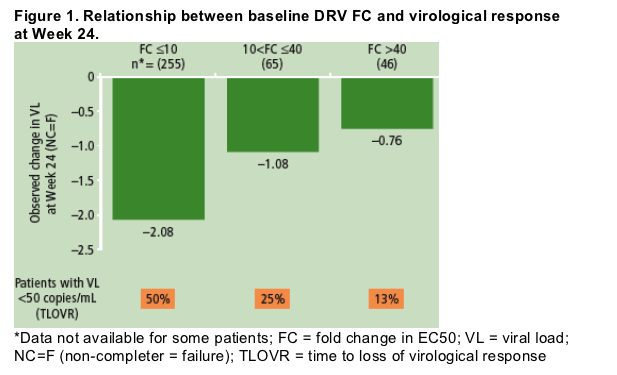
Effect of baseline mutations on response to DRV/r
Reduction of the virological response rate to DRV/r (i.e., /=10 PI
resistance-associated mutations (as defined by IAS-USA)12 present at baseline
(Figure 2).
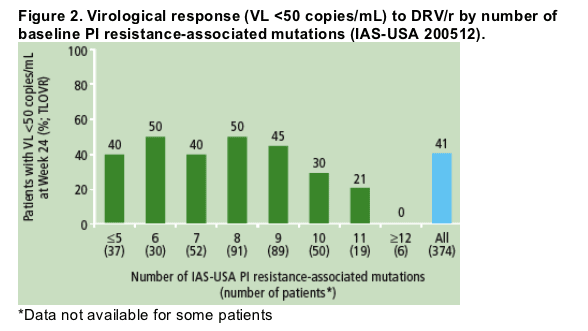
Although reduced response was associated with the presence of a greater number of PI resistance-associated mutations, these PI resistance-associated mutations did not provide a sufficiently precise prediction of response (Figure 2).
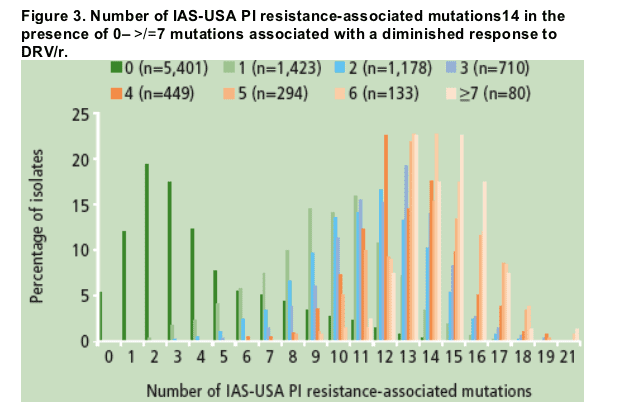
Instead, a specific set of mutations was identified that, when present at baseline, was associated with a diminished response to DRV/r at Week 24; these mutations were V11I, V32I, L33F, I47V, I50V, I54L/M, G73S, L76V, I84V and L89V13
- they were observed in association with a median of at least 10 PI resistance-associated mutations (Figure 3).14
Regardless of the type of baseline DRV resistance-associated mutation, the virological response to DRV/r remained higher than that observed with CPI(s).
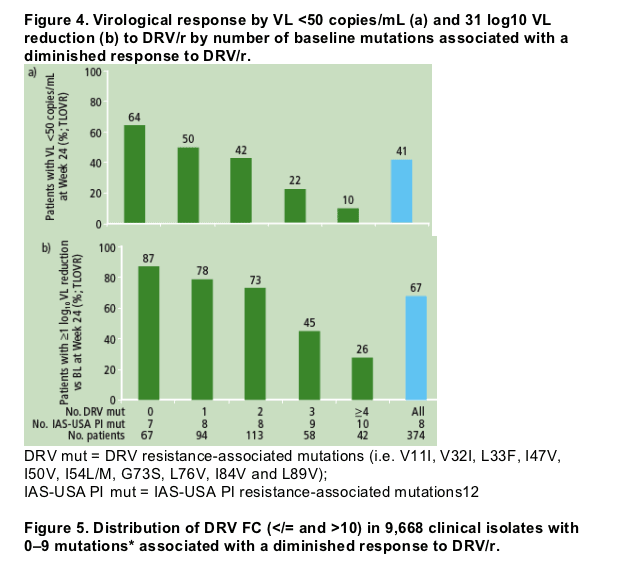
Response was assessed by the number of baseline mutations present that were associated with a diminished response to DRV/r (Figures 4a and 4b). The mean change in log10 VL was -2.1, -1.12 and -0.46 log10 copies/mL for 0-2, 3 and >/=4 DRV resistance-associated baseline mutations, respectively
- a decreased virological response to DRV/r was seen in patients who had >/=3 DRV resistance-associated mutations present at baseline in a background of other IAS-USA PI resistance-associated mutations.12
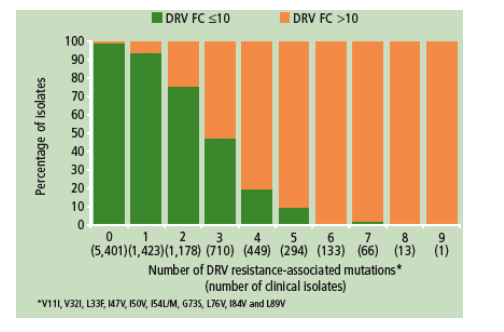
These analyses were confirmed using a panel of 9,668 HIV-1 clinical isolates;
isolates with < /=2 mutations in a background of multiple mutations had a median
FC <10 demonstrating the predictive value of the set of DRV resistance associated mutations (Figure 5).
References
1. De Meyer S, et al. Antimicrob Agents Chemother 2005;49:2314-21.
2. Tibotec Inc. PREZISTATM (darunavir) Prescribing Information. June 2006 [cited 3 October 2006]. Available from: http://www.tibotectherapeutics.com/pi.jsp.
3. Katlama C, et al. 3rd IAS Conference on HIV Pathogenesis and Treatment, Rio de Janeiro, Brazil, 24-27 July 2005. Abstract WeOaLB0.102.
4. Grinsztejn B, et al. 3rd IAS Conference on HIV Pathogenesis and Treatment, Rio de Janeiro, Brazil, 24-27 July 2005. Abstract WePeLB6.201.
5. Wilkin T, et al. 45th Interscience Conference on Antimicrobial Agents and Chemotherapy,Washington DC, USA, 16-19 December 2005. Abstract 2860.
6. Berger D, et al. 45th Interscience Conference on Antimicrobial Agents and Chemotherapy,Washington DC, USA, 16-19 December 2005. Abstract H-1094.
7. Lazzarin A, et al. 16th International AIDS Conference, Toronto, Canada, 13-18 August 2006. Abstract TUAB0104.
8. Sekar V, et al. 13th Conference on Retroviruses and Opportunistic Infections, Denver, CO, USA, 5-9 February 2006. Abstract J-121.
9. Molina JM, et al. HIV Med 2006;7(Suppl. 1):12. Abstract P4.
10. D'Aquila RT, et al. Top HIV Med 2003;11:92-6.
11. Johnson VA, et al. Top HIV Med 2004;12:119-24.
12. Johnson VA, et al. Top HIV Med 2005;13:51-7.
13. De Meyer S, et al. 13th Conference on Retroviruses and Opportunistic Infections, Denver, USA, 5-9 February 2006. Abstract 157.
14. Johnson VA, et al. Top HIV Med 2005;13:125-31.
|
| |
|
 |
 |
|
|