| |
Virologic response and resistance to adefovir in patients with chronic hepatitis B, & Entecavir Salvage Therapy
|
| |
| |
Journal of Hepatology
Volume 44, Issue 2, Pages 283-290 (February 2006)
Scott K. Fung, Hee Bok Chae, Robert J. Fontana, Hari Conjeevaram, Jorge Marrero, Kelly Oberhelman, Munira Hussain, Anna S.F. Lok
Division of Gastroenterology, University of Michigan, Ann Arbor, MI, USA
".....a pooled study of 467 patients with lamivudine-resistant HBV demonstrated that resistance to adefovir was seen only in those who stopped lamivudine. Similarly, in our study, none of 16 patients who continued to receive lamivudine plus adefovir had adefovir resistance compared to 5 of 18 (28%) who were switched to adefovir monotherapy...... the currently approved dose of adefovir is suboptimal is further supported by recent studies showing its inferiority compared to tenofovir 300mg.... An unexpected finding in our study was the association between HBV genotype D and adefovir resistance in 4 of 6 patients....."
ABSTRACT
Background: The incidence and risk factors for adefovir-resistant HBV have not been clearly defined.
Aims: To characterize the virologic response to adefovir, to determine the rate of adefovir resistance and to explore factors associated with initial virologic response (IVR) and adefovir resistance.
Methods: All hepatitis B patients who received adefovir for ≥6 months at our center were prospectively monitored for virologic response and adefovir resistance.
Results: Forty three patients were included; mean treatment duration was 18 months (range 6-45). Thirty four (79%) patients had prior lamivudine. IVR was observed in 44% patients and associated with higher pretreatment ALT (P=0.05) and the absence of HBeAg (P=0.02). Six (14%) patients were found to have adefovir-resistant mutations. The cumulative probability of genotypic resistance to adefovir at month 24 was 22%. Patients with adefovir resistance were more likely to have been switched from lamivudine to adefovir monotherapy (P=0.01), to be older (P=0.04), and to be infected with HBV genotype D (P=0.02).
3.4. Cumulative probability and factors associated with adefovir resistance
During a mean follow-up of 18±9 months, 6 (14%) patients were confirmed to have adefovir-resistant mutations including four who failed to achieve IVR and two who had IVR with subsequent virologic breakthrough in one. The cumulative probability of genotypic resistance to adefovir at 12, 18, and 24 months was estimated to be 0, 16 and 22%, respectively (Fig. 3). The patients who developed adefovir resistance were older (55 vs. 44 years, P=0.04) and the duration of lamivudine overlap was significantly shorter than those who did not have adefovir resistance (0.2 vs. 3.6 months, P<0.001). Among the 34 patients who had previous lamivudine treatment, all five patients with adefovir resistance received adefovir alone in contrast to 9 of 29 patients who did not develop adefovir resistance (100 vs. 31%, P=0.01). Despite a low overall prevalence, HBV genotype D was significantly more common among patients with adefovir resistance (67 vs. 11%, P=0.02). No significant difference in gender, race, HBeAg status, HBV DNA or ALT levels was detected between patients with and without adefovir resistance (Table 3). Of the 20 patients who failed to achieve IVR and were not detected to have adefovir-resistant mutations at rt181 or rt236, only one was found to have a mutation (rtN238H, asparagine to histidine mutation) that had been reported to be associated with decreased response to adefovir [11].
Conclusions: Roughly 50% of patients failed to achieve IVR on adefovir. The cumulative probability of adefovir resistance at 2 years was 22%. Our data suggest that combination of lamivudine and adefovir may prevent emergence of adefovir resistance in patients with lamivudine-resistant HBV.
3.5. Patients with adefovir resistance
All six patients with adefovir resistance were men. Five patients had lamivudine breakthrough and were switched to adefovir monotherapy, while one (no. 6) received adefovir as de novo therapy (Table 4). Two patients (nos. 4, 5) with adefovir-resistant mutation achieved IVR. One patient (no. 5) had low baseline HBV DNA and viral reduction of only 2.2 log10copies/ml with subsequent virologic breakthrough, while the other (no. 4) patient had viral reduction of 4.3 log10copies/ml at month 6 and stable HBV DNA level after the detection of adefovir-resistant mutation.
Adefovir-resistant mutation was detected at a mean of 18±3 months (range, 14-24) after treatment was initiated. At the first detection of genotypic resistance, four patients had HBV DNA >6 log10copies/ml and five had ALT >2 X upper limit of normal (ULN). Specific adefovir-resistant mutations included rtA181V (three patients) and rtN236T (three patients). Two patients (nos. 5, 6) had additional mutations. One patient (no. 2) had an episode of variceal bleeding after adefovir resistance was detected, but no patient died (Table 4).
After the detection of resistance, adefovir was discontinued in four patients, three of these patients were switched to entecavir (nos. 1, 2, 5) (Fig. 4) and 1 to tenofovir (no. 3). The fifth patient (no. 6) continued adefovir and lamivudine was added. For these five patients, mean reduction in HBV DNA was 3.3±2.3 log10copies/ml, three patients had undetectable HBV DNA and two had normal ALT at the last follow-up. The sixth patient (no. 4) did not receive salvage therapy since HBV DNA levels stabilized at approximately 4 log10copies/ml and ALT levels remained normal on continued adefovir therapy.
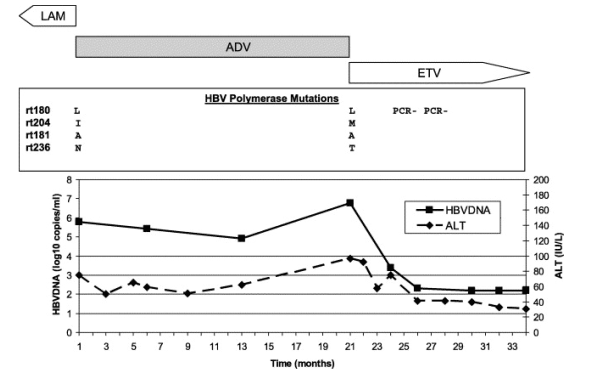
Fig. 4. Clinical course of a patient with adefovir resistance. The clinical course and response to salvage therapy for a representative patient with adefovir resistance is depicted. Antiviral treatment is indicated above the graph: lamivudine was given for >1 year previously (data not shown), adefovir was given for 21 months and entecavir therapy is ongoing. HBV polymerase gene mutations are represented in the boxed panel. The solid line represents HBV DNA levels and the dashed line ALT levels. This patient was described in a previous case series, with follow-up on entecavir of only 8 months [14]. LAM, lamivudine; ADV, adefovir; ETV, entecavir; rt, reverse transcriptase; PCR, polymerase chain reaction.
1. Introduction
Adefovir dipivoxil (adefovir) is a nucleotide analog with activity against wild type and lamivudine-resistant hepatitis B virus (HBV) [1-5]. A prospective trial that randomized 59 HBeAg-positive patients with lamivudine-resistant HBV to ongoing lamivudine, adefovir plus lamivudine, or adefovir alone found that mean reduction in HBV DNA (4 log10 vs. 3.6 log10copies/ml) at 48 weeks was similar in the adefovir monotherapy and combination therapy groups, suggesting that there was no benefit to continuing lamivudine in patients with lamivudine-resistant HBV who are switched to adefovir [2].
Resistance to adefovir is less common and occurs later in the course of hepatitis B treatment compared to lamivudine [6,7]. Data from a study of 998 patients undergoing long-term lamivudine indicate that resistance increased from 23% in year 1 to 71% in year 4 [8]. On the other hand, rates of resistance to adefovir based on a clinical trial of 124 HBeAg-negative nucleoside-naive patients treated with adefovir were estimated to be 0% in year 1, 3% in year 2, 11% in year 3 and 18% in year 4 [6]. Mutations that have been confirmed to confer resistance to adefovir include rtN236T (substitution of asparagine by threonine) and rtA181V/T (substitution of alanine by valine or threonine) [9,10]. Other mutations in the HBV polymerase gene have been reported to be associated with reduced susceptibility to adefovir but the significance of these mutations is unclear [11]. In vitro studies indicate that rtN236T and rtA181V lead to only minor reductions in sensitivity to adefovir (approximately, 3-13-fold) [12,13]. Despite this, significant viral rebound and hepatic decompensation were reported underscoring the importance of close monitoring for drug resistance [14].
The primary aim of this study was to characterize the virologic response to adefovir in a group of patients treated at a tertiary care center. A secondary aim was to determine the probability of adefovir resistance, and to explore clinical and virological factors associated with initial virologic response (IVR) and adefovir resistance.
4. Discussion
In this study, we found a higher probability of resistance to adefovir -22% after 2 years-than has been previously reported in clinical trials of nucleoside-naive hepatitis B patients [6,16]. Possible explanations for this difference may be related to a high proportion of patients with prior lamivudine resistance that was switched to adefovir monotherapy and referral bias. Because of our ability to test for antiviral-resistant mutations, patients who have adefovir treatment initiated by outside gastroenterologists and a suboptimal response or breakthrough are more likely to be referred to our center than those who have a satisfactory response. However, when we restrict our analysis to patients who had adefovir treatment initiated in our clinic, the cumulative probability of adefovir resistance remained high, 20% at 2 years.
Risk factors associated with lamivudine resistance include high baseline HBV DNA, elevated ALT, high body mass index, prior treatment with famciclovir and suboptimal initial virologic response [17]. In a study of 159 patients, lamivudine resistance was more common among those with HBV DNA >3 log10copies/ml after 6 months of treatment compared to those with HBV DNA ≦3 log10copies/ml (63 vs.13%) (OR 11.5, 95% CI 3-40) [18]. Similarly, in a clinical trial of telbivudine, all 26 patients with HBV DNA >4 log10copies/ml compared to 0/30 patients with undetectable HBV DNA at week 24 developed viral breakthrough at week 52 [19]. Limited data suggest that a suboptimal response to adefovir may be associated with resistance. In a recent study of 185 HBeAg-negative patients, a reduced response to adefovir (<2.5 log10copies/ml reduction in HBV DNA compared to baseline) at 48 weeks was seen in 4/6 (67%) patients who developed resistance to adefovir during long term follow-up [16]. In an analysis of data pooled from five clinical studies, 67% of adefovir-treated patients who had HBV DNA >6 log10copies/ml at week 48 were found to have adefovir resistance [6,20]. Similarly, in the present study, 4 of 6 (67%) patients who developed adefovir resistance failed to achieve an initial virologic response.
In the present study, less than half of the patients achieved IVR: only 44% patients had HBV DNA <4 log10copies/ml at month 6, and only 47% patients had >3 log10copies/ml reduction in HBV DNA after 6 months of treatment. Our data are in agreement with a study of HBeAg-positive patients treated with adefovir for 48 weeks, in which 25% patients had <2.2 log10 reduction in HBV DNA and another 25% had 2.2-3.5 log10 decrease in HBV DNA [21]. Several factors may account for this suboptimal response to adefovir including non-compliance, low antiviral activity due to inadequate dose, or inefficient conversion from the prodrug (adefovir dipivoxil) to the active compound (adefovir diphosphate) due to interpatient variability in hepatic deaminase activity. In addition, viral factors such as pre-existing resistant mutations and patient factors may influence the risk of drug resistance. Similar to a previous report [21], most (20/24) of our patients without an IVR demonstrated no evidence of adefovir-resistant mutations during a mean follow-up of 18 months. It is possible that adefovir-resistant mutations were present, but constituted <5% of the viral population. However, 14 of these 20 patients were serially tested with no evidence of adefovir-resistant mutations. We believe that the low rate of IVR is likely a reflection of inadequate dosing of adefovir (10mg) since this dose was chosen because of safety concerns with higher doses (30mg), which are more potent. That the currently approved dose of adefovir is suboptimal is further supported by recent studies showing its inferiority compared to tenofovir 300mg [22,23]. Nonetheless, it is possible that host factors that affect bioavailability of the active compound may play a role in virologic response to adefovir. In our study, most patients who failed to achieve IVR did not have further viral reduction during continued treatment, indicating that these patients were non-responders and not slow responders (Fig. 2).
The role of continuing lamivudine treatment in patients with lamivudine-resistant HBV who are receiving adefovir is controversial. Continued exposure to lamivudine may select for compensatory mutations that serve to restore replication fitness of lamivudine-resistant HBV [24]. Several studies found no added benefit of continuing lamivudine in combination with adefovir compared to adefovir alone in terms of viral suppression, HBeAg seroconversion or frequency of ALT flares over 1 year [2,25]. However, a pooled study of 467 patients with lamivudine-resistant HBV demonstrated that resistance to adefovir was seen only in those who stopped lamivudine [26]. Similarly, in our study, none of 16 patients who continued to receive lamivudine plus adefovir had adefovir resistance compared to 5 of 18 (28%) who were switched to adefovir monotherapy.
An unexpected finding in our study was the association between HBV genotype D and adefovir resistance in 4 of 6 patients. In a study of 694 patients treated with adefovir, HBV DNA suppression was comparable for HBV genotypes A-D [27]. Studies on lamivudine treatment have not found an association between HBV genotype and rate of lamivudine resistance but genotype appeared to be correlated with the specific mutation selected [28]. Further studies involving a large number of patients with various genotypes are required to explore the potential association between HBV genotype and antiviral-resistant mutation.
The long-term outcome of patients who develop adefovir-resistant HBV is not well-described. Despite the small change in antiviral susceptibility of adefovir-resistant mutation, we previously reported that viral rebound and hepatic decompensation may occur in some patients with advanced disease [14]. In the current study, viral rebound to >6 log10copies/ml and ALT flares >2 X ULN occurred in 4 and 5 patients, respectively, and one patient had a variceal bleed. In vitro studies indicate that adefovir-resistant HBV strains are susceptible to lamivudine, clevudine, emtricitabine, À-l nucleosides, entecavir and tenofovir [29]. Lamivudine has been used successfully in clinical studies, but the durability of the response is unknown in patients who have a history of lamivudine resistance. In the present study, three patients who were switched to entecavir had a rapid virologic response, which was maintained for up to 12 months but the optimal antiviral salvage strategy for patients with adefovir-resistant HBV remains unclear.
Limitations of our study include the small number of patients as well as potential referral bias. Half of our patients had bridging fibrosis or cirrhosis, 80% had previously been treated with lamivudine and most had breakthrough infection. Therefore, the results of our study may not be generalized to patients treated in the community.
In conclusion, our data demonstrate that the initial virologic response to adefovir is variable. Only half of our patients achieved IVR at 6 months and most of these patients failed to achieve further viral suppression during continued treatment. We also found a higher probability of resistance to adefovir (22% after 2 years) compared to previous studies. Our study showed that HBV genotype D, older age, and use of adefovir monotherapy in patients previously treated with lamivudine were associated with an increased risk of adefovir resistance. If confirmed in larger studies, treatment guidelines should recommend the addition of adefovir in hepatitis B patients with lamivudine resistance rather than substitution. Further studies are needed to determine if HBV genotype is related to the risk of antiviral resistance.
3. Results
3.1. Baseline characteristics of patients prior to adefovir
A total of 43 patients were included in this analysis. Adefovir was started by physicians at the University of Michigan Liver Clinic in 34 (79%) patients and by outside physicians in 9 (21%) patients. Thirty-eight (88%) patients were men and the mean age was 46±12 years. Sixteen (37%) patients were Asian, 25 (58%) were Caucasian, and 2 (4%) of other races. Twenty-nine (67%) patients were HBeAg-positive, mean ALT was 201 IU/L (range, 23-743 IU/L) and mean HBV DNA was 7.4±1.6 log10copies/ml. The most common HBV genotypes were A (46%), B (8%), C (21%) and D (25%). Nine patients (21%) received adefovir monotherapy as de novo treatment, while 34 patients had prior lamivudine-treatment (mean 26±13 months). Twenty-nine (85%) of these previously treated patients had documented breakthrough of whom 11 had confirmed genotypic resistance to lamivudine while five patients received adefovir due to concerns about future lamivudine resistance. Eighteen of 34 (53%) patients who had prior lamivudine treatment were switched to adefovir monotherapy, while 16 (47%) received adefovir plus lamivudine combination therapy. Lamivudine breakthrough was documented in 15 of 18 patients switched to adefovir monotherapy and in 14 of 16 patients switched to combination of adefovir and lamivudine. No significant differences in demographic or baseline laboratory features were found among patients with and without previous lamivudine therapy (Table 1).
3.2. Virologic response to adefovir
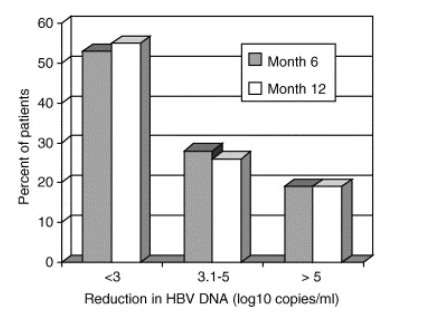
At the start of adefovir, all patients had HBV DNA >4 log10copies/ml and 35 (81%) had HBV DNA >6 log10copies/ml. At month 6, the mean reduction in HBV DNA was 2.8 log10copies/ml (range <1-5.9) and the proportion of patients with ≦3 log10, 3.1-5 log10 and >5 log10copies/ml reduction was 53, 28 and 19%, respectively (Fig. 1). Similarly at month 12, the mean reduction in HBV DNA was 2.9 log10copies/ml (range, <1-6.7) and the proportion with ≦3 log10, 3.1-5 log10 and >5 log10copies/ml reduction was 55, 26 and 19%, respectively. Similar results were observed when the nine patients who had adefovir initiated by outside physicians were excluded. Among the 34 patients with prior lamivudine treatment, HBV DNA reduction at month 6 was less in patients switched to adefovir monotherapy compared to those who received lamivudine plus adefovir but the difference was not significant (2.5 vs. 3.4 log10copies/ml, P=0.10).
Fig. 1. Virologic response to adefovir. Percent of patients with reduction in HBV DNA by <3 log10, 3.1-5 log10 and >5 log10copies/ml at month 6 (shaded bars) and at month 12 (open bars) is illustrated.
3.3. Initial virologic response
Initial virologic response (IVR) defined as HBV DNA <4 log10copies/ml at month 6 was achieved in 19 (44%) patients. There was no difference in age, gender, HBV DNA levels, proportion of patients with prior lamivudine therapy or duration of previous lamivudine treatment between the patients with and without IVR (Table 2). However, patients who achieved IVR were less likely to be HBeAg-positive (47 vs. 83%, P=0.02) and had higher baseline ALT levels (260 vs. 149 IU/L, P=0.049) compared to those who did not have IVR.
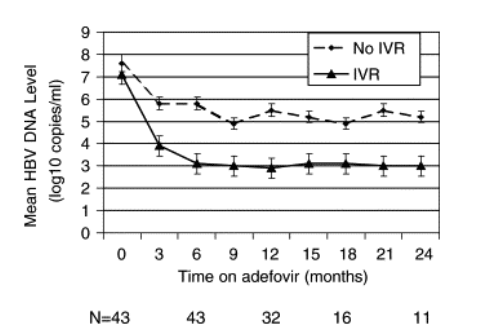
As expected, the 19 patients who achieved IVR had a more marked drop in HBV DNA levels during the first 6 months, mean reduction of 4 vs. 1.8 log10copies/ml in the 24 patients with no IVR (P<0.001). Among the 17 patients with IVR who had completed 12 months treatment, 8 (47%) and 10 (59%) patients had undetectable HBV DNA at months 6 and 12, respectively. Of the 24 patients who did not have IVR, 17 had completed 12 months of treatment, 4 (24%) had >1 log10copies/ml reduction in HBV DNA between month 6 and 12, and 2 (12%) had undetectable HBV DNA at month 12 (Fig. 2). These data indicate that very few patients who failed to achieve IVR had further viral suppression beyond 6 months.
Fig. 2. Virologic response according to initial virologic response. Patients who achieved an initial virologic response are represented by the solid line; patients who did not have an initial virologic response are represented by the dashed line.
3.6. Nephrotoxicity
A reproducible increase in serum creatinine of >0.5mg/dl above baseline was observed in only 1 (2%) patient 18 months after starting adefovir. This patient required a dose reduction (10mg every other day) with improvement in creatinine level. All others patients received 10mg daily dose throughout the study period.
2. Patients and methods
2.1. Patients
All adult HBV patients with compensated chronic hepatitis B referred to the University of Michigan Liver Clinic from June 2001 to January 2005 who had received adefovir for at least 6 months were included in this analysis. This study included patients who received adefovir for lamivudine-resistant HBV as well as patients who received adefovir as de novo treatment. Written informed consent was obtained from all patients and approval for this study was obtained from the Institutional Review Board at the University of Michigan.
Liver panel, creatinine and quantitative HBV DNA were tested every 3 months and patients were seen every 6 months. Serial serum samples were collected on a regular basis during adefovir treatment and after the detection of adefovir resistance. All samples were stored at -80C. Testing for adefovir-resistant HBV mutation was performed in all patients who had received adefovir for ≥6 months and did not achieve an initial virologic response (HBV DNA <4 log10copies/ml after 6 months of therapy) or had virologic breakthrough (≥1 log10copies/ml increase in HBV DNA on at least two occasions after an initial virologic response). Clinical and laboratory data of each patient were reviewed.
2.2. HBV DNA quantification
HBV DNA was measured using a PCR assay, COBAS Amplicor HB Monitor Assay (Roche, Branchburg, NJ), with a lower limit of detection of 200copies/ml. Serial dilutions were performed for samples exceeding 5.3 log10copies/ml.
2.3. Testing for antiviral resistance
HBV DNA was extracted using the QIAamp DNA Blood Mini Kit (Valencia, CA) according to the manufacturer's instructions. The presence of HBV polymerase gene mutations was determined using the InnoLiPA DR2 line probe assay (InnoGenetics, Ghent, Belgium). This assay is based on the DR1 assay described previously [15], but has been updated to include probes that detect lamivudine-resistant mutations at rt80 and rt173 and adefovir-resistant mutations at rt181 and rt236. Line probe results were confirmed with bidirectional automated sequencing at the DNA sequencing core facility (University of Michigan Medical Center, Ann Arbor, MI) using the standard protocol for the Applied Biosystems DNA Sequencer 377 (Perkin Elmer Corp., Foster City, CA) [15]. This amplicon covers domains A, B, C, D, and E of the reverse transcriptase region of the HBV polymerase.
2.4. Definitions
Initial virologic response was defined as HBV DNA <4 log10copies/ml after 6 months on treatment. Virologic breakthrough was defined as ≥1 log10 increase in HBV DNA from nadir on two consecutive occasions after an initial virologic response or an initial decline in HBV DNA by >2 log10copies/ml.
2.5. Statistical analyses
Statistical testing was performed using SPSS version 13.0 (SPSS Inc., Chicago, IL). Results are reported as mean ±SD. HBV DNA levels were logarithmically transformed for analysis. Continuous variables were compared using the 2-tailed student's t-test. Categorical data were compared using the two-tailed X2 test or Fisher's exact test. Factors associated with an initial virologic response and adefovir resistance were analyzed by univariate analysis. The cumulative probability of adefovir resistance was estimated by Kaplan-Meier analysis. P-values ≦0.05 were considered statistically significant
|
|
| |
| |
|
|
|