| |
Confronting chronic hepatitis B virus infection in HIV: new diagnostic tools and more weapons
|
| |
| |
AIDS: Volume 20(3) 14 February 2006 p 451-453
Soriano, Vincent; Sheldon, Julie; Ramos, Belen; Nunez, Marina
From the Department of Infectious Diseases, Hospital Carlos III, Madrid, Spain.
Hepatitis B virus (HBV) is a small enveloped DNA virus that replicates by reverse transcription of the pregenomic RNA using a virus-encoded polymerase. This enzyme lacks proofreading activity, which results in wide virus sequence heterogeneity. Phylogenetic analyses have led to the recognition of eight HBV genotypes (A to H) on the basis of > 8% divergence in the complete genome. As shown in Table 1, evidence for differences in geographic distribution, viral markers, liver pathogenicity and treatment susceptibility for HBV genotypes have emerged in recent years [1-3].
Multiple factors influence liver disease progression in patients with chronic HBV infection, including age at infection, serum HBV DNA titres, alcohol consumption and concomitant infection with hepatitis C or delta viruses. Concurrent HIV infection is associated with accelerated liver fibrosis progression [4]. Given the growing evidence for a distinct behaviour of HBV genotypes, studies on the molecular epidemiology of HBV in HIV-positive individuals are very welcomed [5,6]. In this issue of AIDS, Lacombe et al. [7] report the results of a cross-sectional study conducted in 2002 and 2003 in seven French hospitals, in which 104 HBV/HIV-coinfected patients were identified and underwent a liver biopsy. HBV genotype A was the most prevalent (72%), followed by genotype G (12%), D (10%) and E (6%). Interestingly, advanced liver fibrosis (METAVIR scores F2-F4) was recognized in 69% of patients and the main predictor of advanced liver fibrosis was infection by HBV genotype G.
Recent guidelines [8,9] have recommended that serum HBV soluble antigen (HBsAg) should be screened in all HIV-infected individuals. In those who are positive, full serological assessment of markers of HBV infection should be performed, including testing of HBV e antigen (HBeAg) and anti-HBeAg antibody, and plasma HBV DNA measurement. HBV treatment must be considered for patients with active disease and viral replication [10,11]. In HBV-monoinfected subjects, the indication for therapy until recent years often relied on histological findings. With the advent of new oral drugs and the chance to measure serum HBV DNA levels, the need for a liver biopsy has diminished [10,11]. Moreover, new non-invasive tools for assessing liver fibrosis, such as elastography (FibroScan) [12] and/or serum fibrosis markers [13], have allowed liver biopsies to be avoided in many circumstances, further providing an opportunity for periodic longitudinal assessment of these patients.
While there is an association between serum HBV DNA titres and liver damage in patients with chronic HBV infection, the minimal threshold of plasma HBV DNA associated with progressive liver disease is unknown. Recent guidelines in HIV-negative persons with chronic HBV hepatitis have recommended that treatment should be considered for HBeAg-positive patients with serum HBV DNA > 105 copies/ml (100,000 c/ml) [10,11]. A lower threshold seems to be appropriated for HBeAg-negative patients and those with decompensated cirrhosis (thresholds of 104 (10,000 c/ml) and 103 copies/ml (1,000 c/ml), respectively). At this time, the recommendations for treating HBV in the setting of HIV coinfection are that an individual decision should be made based additionally on considerations related to HIV status and, particularly, the need for antiretroviral therapy [8,9]. Clearly, coexistence of HIV infection with chronic HBV infection makes necessary an integrated approach for the appropriate management of both infections.
For patients with detectable HBV viraemia and who have already received lamivudine, HBV resistance should be suspected. Lacombe and colleagues [7] found lamivudine-resistant HBV-associated mutations in more than two-thirds of their HBV/HIV-coinfected population. Emtricitabine is not useful in this setting against HBV, and switching to or adding tenofovir may be advisable. If HIV replication is already suppressed, adding adefovir or entecavir are also reasonable options in this subset of patients.
The introduction of tenofovir into the HBV armamentarium for HIV-coinfected patients (Table 2) has provided new hopes [14-20]. The high genetic barrier for resistance to nucleotide reverse transcriptase inhibitors (adefovir and tenofovir) represents an advantage with respect to the L-nucleosides lamivudine and emtricitabine. Furthermore, these two families of drugs do not show cross-resistance, which makes possible salvage interventions and more attractive combination therapy [21]. A new nucleoside analogue, entecavir, which shows high potency and robust resistance barrier, has recently been approved for the treatment of HBV. However, no doubt resistance will emerge as result of prolonged therapy with any of these compounds, as has recently been pointed out with tenofovir [22].
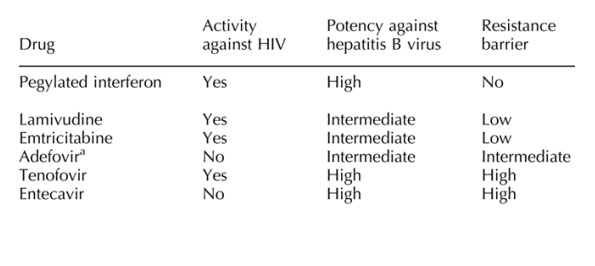
Comments by authors. Lamivudine: >30% resistance at 12 months, excellent tolerance. FTC: equivalent to lamivudine (3TC), coformulated with tenofovir, called Truvada. Adefovir: useful in ESLD & 3TC failures, 15% resistance at 4 yrs. Tenofovir: good tolerance, useful in 3TC & adefovir failures. Entecavir: useful but less potent in 3TC failures (than tenofovir), good tolerance.
In summary, the availability of new HBV diagnostic tools, including genotyping, viral load measurement and resistance testing has come to maturity. Their use will allow more adequate approaches to chronic HBV hepatitis. These tests may permit estimation of the risk of progression of HBV-related liver disease and the tailoring of HBV therapy and monitoring on an individual basis. Consequently, efforts should be made to introduce them into the clinic as soon as possible. Clearly, a new era has begun and this is time to change our approach to HBV in the HIV setting.
|
|
| |
| |
|
|
|