| |
Major role of hepatitis B genotypes in liver fibrosis during coinfection with HIV: HBV genotype G found to be associated with liver fibrosis
|
| |
| |
AIDS: Volume 20(3) 14 February 2006 p 419-427
Lacombe, Karinea,b,c; Massari, Veroniquea,b; Girard, Pierre-Mariec; Serfaty, Lawrenced; Gozlan, Joele; Pialoux, Gillesg; Mialhes, Patrickk; Molina, Jean-Michelh; Lascoux-Combe, Carolinei; Wendum, Dominiquef; Carrat, Fabricea,b,c; Zoulim, Fabienj,k
From the aInserm U707
bUniversity Pierre et Marie Curie
cInfectious and Tropical Diseases Service
dHepatogastroenterology Service
eBacteriology and Virology Service
fAnatomy-Pathology Service, Hospital Saint-Antoine, Paris
gInfectious and Tropical Diseases Service, Hospital Tenon
hThe Infectious and Tropical Diseases Service
iInternal Medicine Service, Hospital Saint-Louis
jInserm U271
kHepatology Service, Hospital Hotel-Dieu, Lyon, France.
"....this study...found that genotype G was the strongest independent factor associated with liver fibrosis in HBV/HIV coinfected patients.... In conclusion, these results highlight the possible role of HBV genotype in the prognosis of HIV/HBV coinfection. Concomitant to early treatment of chronic HBV infection, determination of HBV genotype should be considered as part of the management of patients with multiple risk factors for accelerated liver fibrogenesis such as dual infection with HIV and so treated with potentially hepatotoxic drugs...."
Heptatitis B virus genomic factors associated with liver fibrosis
All the liver biopsies were performed after the year 2000 [mean interval between liver biopsy and inclusion, 154 days (SD, 341)]. The mean length of the liver biopsy specimens was 18 mm (SD, 8.2). The histological findings as a fibrosis score were: F0, 15; F1, 21; F2, 43; F3, 15; and F4, 12. Activity score was A0, 15; A1, 52; A2, 24; and A3, 13 (Table 4). The reference group used for multiple regression analysis comprised 70 patients with a METAVIR score > F1.
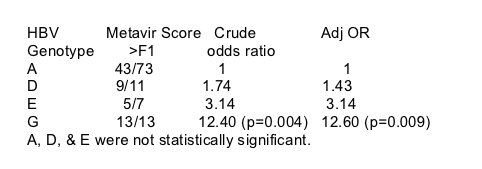
In univariate analysis, HBV genotype G and pre-core mutations were associated with a METAVIR score > F1. Age per year (OR, 1.09; 95% CI, 1.02-1.16), the duration of HIV infection (per year OR, 1.11; 95% CI, 1.02-1.21), previous AIDS-defining events and exposure to efavirenz (per month of treatment OR, 1.05; 95% CI, 1.01-1.09) abacavir, zidovudine and protease inhibitors were also significantly associated with more advanced liver fibrosis. No association was found between use and duration of use of anti-HBV drugs and the level of fibrosis. Lamivudine, tenofovir and interferon were prescribed in 92, 16 and 32 patients, respectively, with a respective mean duration of 52.1 months (SD, 23.9), 5.6 months (SD, 4.2) and 15.0 months (SD, 23.2). The rate of fibrosis > F1 was 64/92 in the lamivudine group, 13/16 in the tenofovir group and 26/32 in the interferon group, not statistically different from the rate of fibrosis in patients not treated (P = 0.2 for lamivudine; P = 0.25 for tenofovir; P = 0.07 for interferon).
The duration of HIV infection was kept in the multivariate model because of its clinical relevance (age and the duration of HIV infection are strongly colinear). After adjustment for the duration of HIV infection, prior anti-HIV and anti-HBV treatment, pre-core mutations, current alcohol intake, sex, body mass index and concomitant chronic HCV or HDV coinfection, three factors remained independently associated with liver fibrosis. HBV genotype G was the strongest determinant of more advanced liver fibrosis (OR, 12.60; 95% CI, 1.72-infinity). Efavirenz exposure and the duration of HIV infection were also independently associated with advanced fibrosis. The generalized estimating equation analysis, used to test for a possible clustering effect owing to variations among the four histologists, showed that the results were not altered by adding a random effect to the parameter estimation. Therefore, reader heterogeneity did not influence the association between liver fibrosis and the HBV genotype, efavirenz treatment or the duration of HIV infection.
Discussion
This study has identified three independent factors associated with advanced liver fibrosis in HIV/HBV-coinfected patients, and obtained new information on the independent role of the HBV genotype in liver fibrogenesis.
HBV genotype G was recently discovered in a population of French and American patients infected with HBV [15], and its prevalence in HIV/HBV-coinfected patients has not previously been assessed. Genotype G is thought to occur mainly in Western countries and to be transmitted by men having sex with men [16]. The relatively high frequency of this genotype in our study population may, therefore, reflect the predominance of male homosexuals and supports previous data.
The phylogenetic characteristics of HBV genotype G have been studied [17] but their clinical implications are unknown. The role of the viral genotype in HBV-associated liver fibrogenesis has rarely been investigated and the data are controversial. Indeed, some studies of Asian populations have shown that HBV genotype C is associated with more severe liver disease and more rapid onset of hepatocellular carcinoma, while other studies involving different clades of the same viral genotypes or longer follow-up periods showed no significant differences between HBV genotypes B and C [17-21]. European studies did not identify the HBV genotype as an independent predictive factor of severe liver disease, although they predated the discovery of HBV genotype G [7,22]. None of these studies referred to HIV/HBV-coinfected patients, in whom HAART-induced liver fibrosis must be considered [23]. We adjusted our analysis of HBV genomic characteristics for variables possibly associated with liver fibrosis (HAART, alcohol intake, sex, body mass index) and found that genotype G was the strongest independent factor associated with liver fibrosis. HBV genomic characteristics were evenly distributed in the study population and did not influence the likelihood of liver biopsy, thus limiting this possible selection bias. Pre-core stop codon mutations were also associated with more severe fibrosis in univariate analysis, but not after adjustment for the HBV genotype. This might be explained by the fact that HBV genotype G strains have two stop codons in the pre-core region, at positions 2 and 28, preventing translation of the HBeAg precursor and leading to a predominance of pre-core mutants in HBV genotype G-infected patients [15,17]. This was confirmed in our study, as HBV genotype G was the genotype most often associated with pre-core mutations. This led to a biased association between pre-core mutations and liver fibrosis in univariate analysis, which disappeared after adjustment for the HBV genotype. This finding is potentially important, as HBeAg-negative chronic HBV infection is classically associated with more advanced fibrosis and with cirrhosis [24]. One possibility is that pre-core mutant strains are associated with a longer duration of infection; this, in turn, may explain the increased frequency of cirrhosis. Alternatively, the fluctuating nature of HBV disease (notably with exacerbations of raised alanine aminotransferase) in patients with HBV pre-core mutations may also accelerate the progression of liver fibrosis. The impact of the HBV genotype and pre-core mutations is controversial in patients infected by HBV alone [7,8,10,25]. In contrast, in HIV/HBV-coinfected patients, liver fibrogenesis might be related more to the HBV genotype (genotype G) than to pre-core mutants.
HBV genotype A predominated in these HIV/HBV-coinfected patients living in Europe, in keeping with the results of two small recent studies in which 57% and 92% of patients were infected by HBV genotype A [16,26]. The relatively high prevalence of genotype E in our population may be explained by the large participation of patients originating from West Africa, where this genotype is the most prevalent in the HBV-monoinfected population [27].
The observed association between efavirenz exposure and the degree of liver fibrosis may not be a causal one. Indeed, the temporal relationship between efavirenz exposure and the onset of liver fibrosis could not be firmly established because of the cross-sectional design of the study. However, it is noteworthy that the duration of efavirenz exposure showed a linear correlation with the risk of fibrosis. Other NNRTI, such as nevirapine, have previously been linked to a higher risk of hepatoxicity in HIV-infected patients with HCV or HBV coinfection [28]. A recently published retrospective study of nevirapine in HIV/HCV-coinfected patients concluded that nevirapine accelerated liver fibrogenesis [29], but prospective studies are needed to draw firm conclusions on the impact of nevirapine and efavirenz on liver fibrosis. All other associations between HAART and liver fibrosis in our study disappeared after adjustment for the duration of HIV infection.
The last factor associated with fibrosis was the duration of HIV infection. Given the colinearity of age and the duration of HIV infection, and as the duration of HIV infection was more strongly associated than age with liver fibrosis in univariate analysis, we chose to keep this parameter in the multivariate model. Many studies have shown HIV coinfection is a factor in HBV disease progression and that the risk of liver fibrosis increases with age and the duration of hepatotrophic virus infection [30]. Unfortunately, in our study, we were not able to determine the direct association between the duration of HBV infection and liver fibrosis as the diagnosis of chronic HBsAg carriage was often incidental, and few patients recalled having symptoms of acute infection. The influence of the duration of HBV infection in the context of HIV still needs to be clarified through longitudinal studies.
In conclusion, these results highlight the possible role of HBV genotype in the prognosis of HIV/HBV coinfection. Concomitant to early treatment of chronic HBV infection, determination of HBV genotype should be considered as part of the management of patients with multiple risk factors for accelerated liver fibrogenesis such as dual infection with HIV and so treated with potentially hepatotoxic drugs.
Abstract
Background: Little is know about the determinants of liver fibrosis progression and genomic variability in hepatitis B virus (HBV) in HIV/HBV-coinfected patients.
Methods: A cross-sectional analysis examined common characteristics of HBV infection in an ongoing cohort study of 308 patients with both HIV-1-positive Western blot and plasma HBV surface antigen (HBsAg) seropositivity. Risk factors for liver fibrosis were studied in a subset of 104 patients for whom liver biopsy and complete HBV genomic analysis were available. Analysis was performed by exact multiple regression analysis.
Results: Mean age of the study population was 40.3 years, with a ratio male to female of 5.3 and a mean duration of HIV infection of 9.3 years. In the subset of 104 patients, plasma HBV e antigen (HBeAg) in HBV-replicative patients could not be detected in 28.4% and lamivudine-resistant mutants were detected in 67.8%. HBV genotype A was the most frequent genotype (73/104) and 25 patients were infected by the usually rare genotype G. METAVIR fibrosis score was rated F2-F4 in 70 patients. After adjustment for the most common known determinants of liver fibrosis, HBV genotype G [odds ratio (OR), 12.60; 95% confidence interval (CI), 1.72-infinite; P < 0.009], efavirenz exposure (OR, 3.55; 95% CI, 1.14-12.14; P < 0.03), and the duration of HIV infection (3.86; 95% CI, 1.27-12.64; P < 0.01) were strongly associated with the risk of grade F2-F4 fibrosis......".... The observed association between efavirenz exposure and the degree of liver fibrosis may not be a causal one...."
Conclusion: HBV genotype G is a determinant of liver fibrosis in HIV/HBV-coinfected patients and HBV genotyping should be considered as part of the management of patients with multiple risk factors for rapid progression of liver fibrosis.
Introduction
The advent of HAART has transformed the management of dual infection by HIV and hepatitis B virus (HBV), which have similar modes of transmission. Immunodeficient patients spontaneously clear serum HBV e antigen (HBeAg) significantly less frequently than HIV-seronegative patients [1]. This might contribute to the higher prevalence of chronic carriage of HBV surface antigen (HBsAg; approximately 5-10% according to geographical origin of patients) among HIV-infected patients than in other subjects (< 5%) [2]. The longer life expectancy of HIV-infected persons treated with HAART will likely lead to a higher frequency of complications of chronic HBV infection, such as fibrosis and cirrhosis. End-stage liver disease has recently emerged as an important cause of death among HIV-infected patients [3], and chronic HBV infection has been identified as a cause of the excess of liver disease-related deaths among these patients [4]. Determinants of fibrosis progression have been extensively studied in the context of coinfection with HIV and hepatitis C virus (HCV) [5,6] but not in HIV/HBV coinfection. Likewise, viral determinants of liver fibrosis and their implication in hepatitis B outcome and treatment, such as the HBV genotype and polymerase or pre-core mutations, have been investigated in patients infected by HBV alone [7-10] but not in patients infected by both HBV and HIV. The present cross-sectional study sought to describe the main general and virological features of HIV/HBV coinfection, and to study the impact of HBV genomic variability on liver fibrosis as part of a prospective multicentre cohort of HIV/HBV-coinfected patients.
Patients and study design
In 2002, seven French HIV/AIDS outpatient clinics from four university hospitals agreed to participate in a 3 year prospective cohort study of liver fibrosis progression in HIV-infected patients with chronic HBV infection. Patients were invited to join the study if they had both HIV (detected by two enzyme-linked immunosorbent assays and confirmed by a full Western blot pattern) and HBV (HBsAg seropositivity) infection. The study was approved by the Pitie-Salpetriere Hospital Ethics Committee (Paris, France) and written informed consent was obtained from all the patients. In order to assess the biases affecting the selection of the patients enrolled in the study, comparison was performed with HBV-uninfected HIV-infected patients included in the French Hospital Database on HIV (FHDH) [11].
Results
General data
Between 1 May 2002 and 1 May 2003, 326 patients recruited in the seven participating centres who were dually infected by HIV and HBV were eligible for a 3 year clinical and biological follow-up study. Preinclusion testing showed that 308 patients were still seropositive for HbsAg. The baseline characteristics of these 308 patients are shown in Table 1. In this group, 281 patients had already been treated with antiretroviral agents and 249 were taking antiretroviral agents at enrolment. The most frequent combinations were one protease inhibitor plus two or three nucleoside reverse transcriptase inhibitors (NRTI; 83 patients); one non nucleoside reverse transcriptase inhibitor (NNRTI) plus two or three NRTI (70 patients); three NRTI (48 patients); and one, two or three NRTI plus one NNRTI plus one protease inhibitor (22 patients).
Table 1 also compares the study patients with the HIV-infected population included in the FHDH database. Male homosexuals and patients from World Health Organization (WHO) zone 3 countries (usually in West Africa) were significantly more frequent in the study population than in the FHDH population. The known duration of HIV infection was also longer in coinfected patients (9.8 versus 8.2 years; P < 0.01) and more patients were receiving HAART at inclusion (81.5 versus 69%; P < 0.005), explaining the higher percentage of patients with controlled HIV infection (54.2 versus 47.7% of patients with HIV RNA < 50 copies/ml; P < 0.05).
Analysis of HBV antigens showed that 161 (52.3%) and 136 (44.2%) patients were seropositive for HBeAg and anti-HBeAg antibodies (HBeAb), respectively, while 11 patients were positive for neither HBeAg nor HBeAb. A coinfection by HCV was noted in 18 patients, with the detection of HCV RNA in 50%. Coinfection with HDV occurred in 14 patients, and 10 patients carried both HDV and HCV. Prior to inclusion, 268 patients had been treated for HBV infection, and 229 patients were still taking anti-HBV therapy. The mean duration of treatment was 30.3 months.
HBV genome analysis showed that genotype A strains predominated and that genotype G was present in 25 patients (12.1% of all genotypes). Patients born in WHO zone 1 countries were mostly infected by genotype A (113/138), genotype G (19/25) or genotype D (12/19), while 22/23 patients originating from WHO zone 3 countries were infected by genotype E. One patient of French origin was infected by the rare genotype B.
Description of the population with liver biopsy and complete HBV genome sequencing
Among the 308 patients included in the follow-up study, HBV genome analysis was performed in 206 and liver biopsy in 134 (Fig. 1). The reasons for the lack of viral genome data were HBV DNA PCR negativity at inclusion in 94 and an undetermined nucleotide sequence reading in eight. As a result, 104 patients were eligible for this study of risk factors for liver fibrosis.
These 104 patients were compared with the overall study population (Table 2). Patients with biopsy data were more likely to be males born in a Western country (mainly France) and to have a long duration of HIV infection. As expected, given the indications for liver biopsy, these patients had a higher HBV load (HBV DNA 5.5 versus 3.8 log copies/ml; P < 0.0001) and were more frequently seropositive for HBeAg (80.8 versus 37.7%; P < 0.0001). They also had higher alanine aminotransferase levels (82.1 versus 51.7 IU/ml; P < 0.0001) and were more likely to have received interferon therapy (30.8 versus 16.2%, P < 0.005). Neither the distribution of HBV genotypes and mutations nor coinfection with other hepatotrophic viruses differed between the two groups.
Table 3 shows the clinical and biological features of the 104 patients providing biopsies according to the HBV genotype. Patients with HBV genotype G infection were more likely than other patients to have the viral stop codon mutation at codon 28 of the pre-core region (81.6% versus 9.6% in genotype A; P < 0.0001). The frequency of YMDD mutations (indicating lamivudine exposure) was not influenced by the HBV genotype.
Methods
Patients and study design (see above)
Biochemical and virological data collection
Biochemical tests and serological tests for HBV, HCV and hepatitis D virus (HDV) were performed at enrolment. HBV load was quantified prospectively with a commercial polymerase chain reaction (PCR) assay (PCR-Amplicor, Roche Diagnostic Systems, Meylan, France; detection limit 200 copies/ml). If HBV load was above the working range of the assay, the sample was diluted and retested. When antibodies to HCV or HDV were detected, serum HCV and/or HDV RNA was quantified by PCR when appropriate. HIV load was measured with the branched-DNA technique (b-DNA Quantiplex 3.0 Bayer Diagnostics, Cergy Pontoise, France; detection limit 50 copies/ml). CD4 and CD8 cells were counted by flow cytometry. HBV polymerase gene mutations, pre-core gene region mutations and HBV genotypes were determined by using DNA chips, after amplifying the whole HBV genome in a two-step PCR reaction, as previously described for HIV [12]. The main lamivudine-resistance mutations in the gene for viral polymerase (V173L, L180M, M204V, M204I) are detected by this method, together with the stop codon mutation at codon 28 of the pre-core region, and the HBV genotype.
Histological data collection
Liver biopsies performed within 18 months before or after inclusion are referred to as baseline liver biopsies. Liver biopsy was proposed on the basis of the peer-reviewed literature and consensus statements on HBV-monoinfected patients. Liver paraffin-embedded biopsies were stained with haematoxylin-phloxin-saffran and picrosirius red and read by four pathologists who were unaware of the clinical and biological data, except for the diagnosis of chronic HBV infection. Histological fibrosis and activity were scored with the METAVIR classification [13]. Liver fibrosis was quantified as absent/mild (METAVIR F0-F1) or extensive (METAVIR F2-F3-F4). A generalized estimating equation [14] was used to test the clustering effect possibly caused by readers' heterogeneity in the final logistic regression model.
Statistical analysis
The results were obtained by a cross-sectional analysis of baseline data. The frequencies of qualitative data and means of quantitative data are expressed as absolute numbers and percentages or SE values, respectively. Comparisons with the FHDH population were based on Pearson's x2-square test for categorical variables and Student's t-test for continuous variables. The level of significance was set to P = 0.05.
Associations between the degree of liver fibrosis and candidate variables were expressed as odds ratios (OR) and 95% confidence intervals (CI). Dummy variables were created for qualitative variables of more than two classes. The OR values for quantitative variables (for an increase of one unit in the variable) were calculated by simple regression analysis. A logistic regression model was built by entering all variables for which the test of association with liver fibrosis stage > F1 led to P < 0.10 in univariate analysis. Because the numbers contained in cells built with variables such as HBV genotype and HDV serology were small, exact logistic regression was used to determine regression coefficients and their CI values. Data were computed with SPSS software release 11.5.0 (2002; SPSS, Chicago, Illinois, USA)) and regression analyses were performed with LogXact release 5 (Cytel Software, Cambridge, Massachusetts USA).
|
|
| |
| |
|
|
|