| |
Tenofovir in HBV/HIV Coinfection, added to Lamivudine
|
| |
| |
"Anti-hepatitis B virus efficacy of tenofovir disoproxil fumarate in HIV-infected patients"
Hepatology, March 2006 Yves Benhamou 1 *, Herve Fleury 2, Pascale Trimoulet 2, Isabelle Pellegrin 2, Renaud Urbinelli 3, Christine Katlama 4, Willy Rozenbaum 5, Gwenael Le Teuff 3, Aldo Trylesinski 6, Christophe Piketty 6, TECOVIR Study Group 1Service d'Hepato-gastroenterologie, Hopital Pitie Salpetriere, Paris, France 2Service de Virologie, CHU Bordeaux, Bordeaux, France 3Service de Virologie, Cenbiotech, Dijon, France 4Service des Maladies Infectieuses, Hopital Pitie Salpetriere, Paris, France 5Service des Maladies Infectieuses, Hopital Tenon, Paris, France 6Service d'Immunologie Clinique, Hopital Europeen Georges Pompidou, Paris, France Potential conflict of interest: Dr. Benhamou is a consultant and has received research grants from Gilead. Funded by: Gilead
Abstract
Tenofovir disoproxil fumarate (TDF) has shown in vitro activity against both HIV and hepatitis B virus (HBV). We retrospectively evaluated the efficacy of TDF (300 mg/d), administered as a part of anti-retroviral therapy, in a large cohort of HIV/HBV-coinfected patients.
Sixty-five HIV/HBV-coinfected patients who received TDF for at least 6 months with serum HBV DNA levels above 2.3 log10 copies/mL at TDF initiation and who had stored serum samples before and during TDF therapy were included. Serum HBV DNA was measured on stored samples. The median follow-up period was 12 (Q1-Q3: 8-17) months.
Serum hepatitis B e antigen (HBeAg) was positive in 54 patients (83.1%). Fifty-two patients (80.0%) were receiving lamivudine (LAM) (150 mg twice a day), and 68.8% had documented LAM resistance at baseline.
Among HBeAg-positive patients, the median reduction from baseline (8.17; Q1-Q3 = 7.30-8.30 log10 copies/mL) of serum HBV DNA was 4.56 log10 copies/mL (Q1-Q3 = 3.33-5.55) (P < .0001).
In HBeAg-negative patients, serum HBV DNA decline from baseline (4.83; Q1-Q3 = 2.69-6.40 log10 copies/mL) was 2.53 log10 copies/mL (Q1-Q3 = 0.39-4.10).
At the end of the study, HBV DNA became undetectable in 29.6% and 81.6% of the HBeAg-positive and HBeAg -negative patients, respectively. Serum HBeAg became negative in 4 patients, 2 of whom acquired serum hepatitis B e antibody.
In conclusion, this retrospective analysis demonstrates the efficacy of TDF against wild-type, presumed precore mutants and LAM-resistant HBV when used as a part of anti-retroviral therapy in HIV-coinfected patients.
DISCUSSION
The results of this study of 65 HIV/HBV-coinfected patients strongly suggest the efficacy of TDF in combination with LAM for the control of HBV replication in both HBeAg-positive and HBeAg-negative patients mostly exhibiting mutations within the YMDD motif associated with LAM resistance. The median reduction in serum HBV DNA after approximately 12 months of TDF therapy was 4.56 log10 copies/mL in HBeAg-positive patients and 2.53 log10 copies/mL in HBeAg-negative patients. HBV DNA became undetectable in 29.6% and 81.6% of the HBeAg-positive and HBeAg-negative patients, respectively. Durability of the virological response on TDF therapy was documented in 100% and 66.6% of the HBeAg-positive and HBeAg-negative patients, respectively. In the current study, the proportion of patients with LAM resistance to HBV was higher than previously reported in HIV/HBV-coinfected patients (70% after a median period of 2.7 yr of LAM monotherapy).[14][37] This high-resistance profile was not observed with TDF. Only 2 patients experienced a rebound from nadir in serum HBV DNA during TDF therapy. Interestingly, mutations in the DNA reverse transcriptase were found in only 1 patient after 19 months of LAM and TDF combination therapy. The M204V mutation - which confers HBV LAM resistance - was not an emergent mutation in this patient, because it was identified at baseline. The A181V and N236T mutations associated with HBV resistance to ADV monotherapy, a nucleotide close to TDF structure, in HBV-infected patients were not identified in the 2 HIV/HBV-coinfected patients with serum HBV DNA rebound.[36] Similarly, the A194T mutation, which may confer HBV resistance to TDF in HIV/HBV-coinfected patients,[34] was not found in our 2 patients who experienced a breakthrough. A181V, N236T, and A194T mutations were not observed in the 4 patients who failed to respond to TDF therapy. Furthermore, no other changes in addition to LAM-, ADV-, and TDF-associated mutations at the conserved site of the HBV polymerase were identified before and during TDF therapy in any of these six patients. It is likely that both absence of HBV virological response and HBV DNA breakthrough were related to noncompliance to TDF therapy in these 6 patients.
Among HBeAg-positive patients, plasma HIV RNA of less than 2.87 log10copies/mL was the only independent factor significantly associated with serum HBV DNA negativity during TDF therapy. The control of HIV replication which is associated with more effective immune functions may have contributed to a better control of HBV replication. A lower plasma HIV RNA level may reflect a good adherence to antiretroviral therapy - including TDF - and subsequently a better inhibition of HBV replication. Indeed, concomitant therapy with LAM and baseline serum HBV DNA (<8.17 log10 copies/mL) may also influence the virological response of TDF. However, this association was not significant. It may be due to a lack of statistical power, as suggested by the wide boundaries for the confidence limit interval. Mutations within the YMDD motif of the DNA polymerase result not only in a decrease of viral susceptibility to LAM but also in a lower replication efficiency compared with wild-type strains.[38] In the current study, LAM was maintained throughout TDF therapy in all the patients receiving LAM at baseline. Thus, addition of TDF to LAM therapy may be more effective in reducing serum HBV DNA than TDF monotherapy.
The rate of serum HBeAg loss among the HBeAg-positive patients (7.4%) was within the previously observed range in HIV/HBV-coinfected patients receiving LAM and ADV.[19][39] Suppression of HBV replication was associated with normalization of ALT in 41.9% of the patients, which may be associated with an improvement in necroinflammatory liver lesions. Accordingly, no liver decompensation was observed during the study period in any of the patients, including patients with documented cirrhosis.
TDF was also effective against HIV replication. Although a large proportion of patients had a controlled HIV replication at TDF initiation, a significantly higher proportion of patients exhibited HIV RNA below the level of quantification (<2.6 log10 copies/mL) at the end of the study compared with baseline.
Our study has some limitations, mainly due to its retrospective design. TDF may have been initiated not only for LAM-resistant HBV but also for the failure of anti-retroviral therapy, as indicated by the heterogeneous HBV DNA levels at baseline. The large range of baseline serum HBV DNA may have contributed to an overestimation of TDF anti-HBV activity. However, 78.5% of the 65 included patients had a high baseline serum HBV DNA (5 log10 copies/mL). In addition, accurate evaluation of TDF anti-HBV activity has been limited by the nonstandardized interval collection of serum for HBV DNA measurements across participating centers. Because the median follow-up was short, particularly in HBeAg-negative patients, we have limited efficacy analyses for patients with at least 6 months of TDF therapy. Longer treatment periods may have shown different results, particularly regarding the emergence of TDF resistance. Finally, the effect of HBV DNA suppression on liver lesions was not assessed in this study. However, recent studies in HBV-infected patients have shown a strong correlation between long-term HBV DNA suppression and a decreased risk of liver decompensation, hepatocellular carcinoma, and liver-related mortality.[40-42] Prospective studies with longer study periods are needed to assess the durability of the response, emergence of resistance, HBeAg seroconversion, and histological improvement of TDF therapy in HIV/HBV-coinfected patients.
Long-term use of LAM monotherapy for HBV is limited in HIV/HBV-coinfected patients because of the high rate of HBV resistance.[9][12][14][15][18][37][43] Until recently, treatment options for LAM-resistant HBV were limited. ADV 10 mg/d over 48 weeks has demonstrated efficacy and safety in CHB treatment-naive patients and in patients who have developed HBV resistance to LAM.[44-47] In HIV-infected patients with LAM-resistant HBV, 48-week treatment with ADV resulted in a significant reduction in serum HBV DNA and serum ALT level without emergence of mutations.[19] Virological and biochemical responses were sustained over a course of 3 years of therapy.[48] However, the 10-mg daily dose of ADV recommended for CHB treatment is not active against HIV infection. The efficacy and safety of TDF in HIV infection has been demonstrated in both anti-retroviral-naive and reverse transcriptase inhibitor-experienced patients.[21-23] TDF has been recently approved in combination with other anti-retroviral agents for the treatment of HIV infection in naive and experienced patients at a dose of 300 mg once daily. Thus, TDF therapy appears to be the best option for the treatment of HBV in HIV-coinfected patients. However, whether TDF should be used as monotherapy or in combination with LAM in HIV/HBV-coinfected patients remains to be studied.
In conclusion, the present study suggests that TDF 300 mg/d, given as a part of antiretroviral therapy, is effective against wild-type, presumed precore mutant and LAM-resistant HBV strains in HIV-coinfected patients. However, prospective studies with TDF are needed to assess long-term antiviral efficacy, durability of HBV DNA suppression, HBeAg loss and seroconversion rates, the potential for emergence of resistance, and long-term safety when combined with either LAM or emtricitabine as first-line therapy in HIV/HBV-coinfected patients.
RESULTS
One hundred nineteen HIV/HBsAg-positive patients who received TDF were identified. Five patients were excluded because of missing clinical information in 2 patients and no available stored serum in 3 patients. Of the 114 remaining patients, 76 had a follow-up of 6 months or more. HBeAg-positive/HBeAb-negative (HBeAg-positive) and HBeAg negative/HBeAb positive (HBeAg-negative) serum samples were found in 55 (72.4%) and 21 (27.6%) patients, respectively.
Serum HBV DNA (>2.3 log10copies/mL) was detectable at baseline in 54 (98.2%) and 11 (52.4%) of the HBeAg-positive and HBeAg-negative patients, respectively. These 65 patients represented the basis of the analyses of anti-HBV activity of TDF in this study. The main baseline characteristics of included patients according to HBe serological status are summarized in Table 1. Identification of HBV LAM resistance-associated mutations at baseline and HBV genotypes are shown in Table 2. There were no statistically significant differences between included patients with a follow-up of 6 months or more and 114 HIV/HBsAg-positive patients initially identified with regard to the baseline characteristics (data not shown). Cirrhosis was documented in 7 of 28 patients who had a liver biopsy (25.0%). Two patients with cirrhosis experienced episodes of liver decompensation before initiation of TDF. None of the patients with cirrhosis experienced liver decompensation throughout the study period.
TDF therapy was generally well tolerated; only 1 case of renal tubulopathy was recorded.
Response to TDF Therapy.
LAM therapy was maintained in the anti-retroviral regimen throughout the study period in all the patients who were on LAM at TDF initiation. The proportion of patients with 3 or more serum HBV DNA determinations was 86.8%; a median of 4 serum HBV DNA measurements per patient were performed during the study period. None of the included patients had serum HBsAg loss at the end of the study period.
HBeAg-Positive Patients.
Serum HBeAg loss was observed in 4 (7.4%) of the HBeAg-positive patients (n = 54) and was associated with acquisition of serum HBeAb in 2 patients (3.7%).
Fifty of the 54 (93%) HBeAg-positive patients had a decline in HBV DNA serum level of at least 1 log10 copies/mL. The median reduction of serum HBV DNA at the end of follow-up was 4.56 log10 copies/mL (Q1-Q3 = 3.33-5.55) (P < .0001). Median changes in serum HBV DNA of HBeAg-positive patients are shown in Fig. 1. Time to reach serum HBV DNA undetectability (<2.3 log10 copies/mL) is presented in Fig. 2. Sixteen (29.6%) HBeAg-positive patients became HBV DNA undetectable during TDF therapy, and 13 of these patients had subsequent HBV DNA measurements. Serum HBV DNA remained undetectable in all 13 patients for a median period of 6 months (Q1-Q3 = 3-9).
Serum HBV DNA did not substantially change from baseline in 3 (5.6%) HBeAg-positive patients, and 1 patient (1.9%) had an increase from baseline in serum HBV DNA of 2.48 log10 copies/mL. Genotyping analysis of the serum samples of these 4 patients did not reveal ADV or potent TDF-related HBV resistance mutations (A181V, N236T, A194T) at baseline and during TDF therapy.
During the study period, 2 HBeAg-positive patients experienced a rebound in serum HBV DNA of at least 1 log10 copies/mL from nadir at month 10 and 19 of TDF therapy. The reverse transcriptase sequences of serum samples of these 2 patients at baseline and at the time of breakthrough were analyzed. Mutations of resistance to LAM (V173L, L180M, M204V/I) and ADV (A181V, N236T) were sought.[13][35][36] None of the reported DNA polymerase mutations associated with LAM or ADV resistance to HBV was identified in 1 patient. In the other patient, an M204V mutation was the only mutation found at baseline and at month 19 of TDF and LAM combination therapy. TDF was discontinued at month 20 because of a renal tubulopathy that occurred in the context of Castelman disease and non-Hodgkin's lymphoma. The patient was hospitalized with renal tubule disorder reported as a Fanconi-like syndrome without renal failure. All antiviral treatments including TDF were stopped. No renal biopsy was performed. This subject recovered few weeks after TDF withdrawal. In addition, A194T mutation was not identified in either of these two patients.
The median serum ALT levels at TDF initiation were 70 (Q1-Q3 = 41-95) IU/L in HBeAg-positive patients (Table 1). At baseline, 38 (70.4%) patients had elevated serum ALT levels; these levels had returned to normal in 16 (42.1%) patients by the end of follow-up (12 months; Q1-Q3 = 8-17).
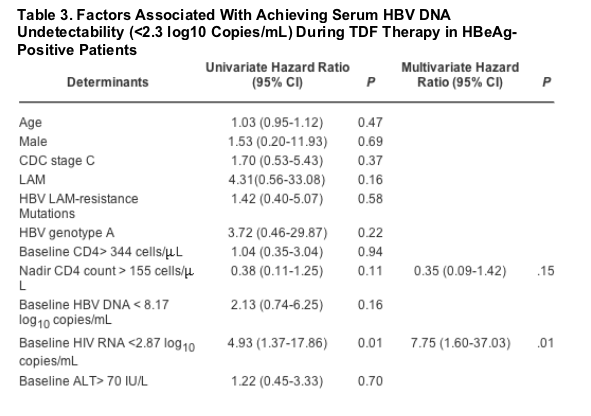
Studied factors associated with achieving an undetectable serum HBV DNA (< 2.3 log10 copies/mL) in HBeAg-positive patients with detectable serum HBV DNA at baseline are shown in Table 3. In a multivariate Cox regression model, only a baseline HIV RNA of <2.87 log10 copies/mL remained independently associated with a shorter period to reach serum HBV DNA undetectability (hazard ratio = 7.75 [95% CI 1.60-37.03]; P = .01). Concomitant LAM therapy and baseline serum HBV DNA lower than 8.17 log10 copies/mL were also associated with serum HBV DNA negativation, but this relationship did not reach statistical significance in multivariate analysis.
HBeAg-Negative Patients.
Median serum HBV DNA decline among the 11 HBeAg-negative patients (n = 11) was 2.53 (Q1-Q3 = 0.39-4.10) log10 copies/mL after a median period of 10 (Q1-Q3 = 7-17) months of TDF therapy. Nine of 11 (81.6%) HBeAg-negative patients became serum HBV DNA undetectable during TDF therapy. Serum HBV DNA remained negative in 6 of these 9 patients who had subsequent measurements for a median period of 6 months. None of the HBeAg-negative patients experienced a rebound in serum HBV DNA during the study period. ALT serum level normalized in 2 of the 5 HBeAg-negative patients with elevated serum ALT at baseline.
HIV RNA and CD4 Cell Count Changes.
In the overall studied population (n = 65), the median baseline CD4 cell count and plasma HIV RNA were 342 X 106 cells/L (Q1-Q3 = 201-489) and 2.86 log10 copies/mL (Q1-Q3 = 2.60-5.13), respectively. At the end of follow-up, a significantly higher proportion of patients exhibited a viral load below 2.6 log10 copies/mL compared with baseline (67.2% vs. 46.8% ; P = .01).
BACKGROUND
There are approximately 350 million hepatitis B carriers and over 40 million HIV-infected individuals worldwide.[1] Because HIV and hepatitis B virus (HBV) share similar routes of transmission, prevalence of hepatitis B surface antigen (HBsAg) is more than 5-fold higher among HIV-infected patients compared with the general population.[2-4] Individuals with chronic hepatitis B (CHB) are at increased risk of disease progression to cirrhosis and hepatocellular carcinoma.[5] After HBV infection, both development and persistence of CHB are greater in patients with previous HIV infection.[6][7] Underlying HIV infection is associated with a higher risk of cirrhosis and a substantial increased in liver-related mortality compared with HBV-monoinfected individuals.[2][8]
Lamivudine (LAM) inhibits HBV replication in both HIV and non-HIV-infected CHB patients.[9-11] However, emergence of HBV resistance to LAM has been widely observed in both HBV-infected and HBV/HIV-coinfected populations.[12-15] HBV resistance to LAM in HIV patients has been reported with an incidence of 50% and 90% after 2 and 4 years of therapy, respectively.[14] LAM resistance is related to mutations located at the YMDD motif of HBV DNA polymerase in all cases.[15] The long-term clinical consequences of HBV resistance in HIV-positive patients remain to be determined. However, as observed in HBV-infected patients, cases of CHB exacerbation and liver failure have been reported in HIV/HBV-coinfected individuals who develop LAM-resistant HBV.[16-18]
Until recently, treatment options for LAM-resistant HBV were limited. Adefovir dipivoxil (ADV) 10 mg/d has been shown to significantly decrease serum HBV DNA and alanine aminotransferase (ALT) levels and to improve liver histology of HIV-infected patients with LAM-resistant HBV coinfection.[19] However, ADV at 10 mg/d is not active against HIV.[20] Tenofovir disoproxil fumarate (TDF) has in vitro activity against both HIV and HBV. TDF is licensed for the treatment of HIV-1 in both treatment-experienced and treatment-naive patients. Numerous studies have demonstrated the use of TDF in the treatment of HIV infection.[21-23] However, TDF activity against HBV in patients with HIV coinfection has been poorly studied. TDF anti-HBV efficacy in HIV-coinfected patients has been reported in small studies with short durations of therapy.[24-32]
The aim of the present study was to analyze TDF efficacy and factors associated with HBV virological responses in a large, retrospective, multicenter cohort study of HBsAg-positive/HIV-coinfected patients who received TDF 300 mg/d as a part of an anti-retroviral regimen.
Patients
Eighteen infectious diseases and/or internal medicine departments in France participated in the study. From January 2001 to January 2003, HIV-infected patients with documented HBsAg seropositivity who had received TDF as a part of anti-retroviral therapy for at least 6 months, with detectable HBV DNA at TDF initiation, and had frozen stored serum samples (-80 C) available at TDF initiation and during TDF therapy were included in the study. Institutional approval was obtained from the French Ministry of Research and the National Healthcare Information Council.
For all included patients, the following data were retrospectively recorded: demographic characteristics; antiretroviral therapies at TDF initiation; HCV coinfection assessed via polymerase chain reaction (Cobas Amplicor HCV 2.0; Roche, Meylan, France); Delta virus infection assessed via Delta antigen or immunoglobulin M anti-Delta-positive serology; absolute numbers of CD4 T lymphocytes measured on whole blood by flow cytometry; plasma HIV RNA levels measured via locally available techniques (lower limit of detection: 2.6 log10 copies/mL); and results of liver biopsy assessed via the METAVIR scoring system.[33] CD4 cell count, plasma HIV RNA, and ALT serum level were recorded at baseline and at the end of follow-up.
All available stored sera were collected to determine serum hepatitis B e antigen (HBeAg) and hepatitis B e antibody (HBeAb), HBV DNA serum levels, and HBV genotyping.
|
|
| |
| |
|
|
|