| |
HBV DNA Early Response Predicts IFN Nonresponse
|
| |
| |
Quantitative HBV DNA levels as an early predictor of nonresponse in chronic HBe-antigen positive hepatitis B patients treated with interferon-alpha
Journal of Viral Hepatitis
Volume 13 Page 96 - February 2006
A. van der Eijk1, H. G. M. Niesters2, B. E. Hansen3, R. A. Heijtink2, H. L. A. Janssen1, S. W. Schalm1 and R. A. de Man1
Departments of 1Gastroenterology & Hepatology; 2Virology and 3Epidemiology & Biostatistics, Erasmus MC, Rotterdam, The Netherlands
Summary. To reduce unnecessary exposure to treatment, physicians must decide at an early stage whether continuation of treatment has a reasonable chance of success for the individual patient. The objectives of our study were to evaluate the previously described quantitative hepatitis B e antigen (HBeAg) measurements vs quantitative hepatitis B virus (HBV) DNA measurements for prediction of nonresponse and response in interferon (IFN)-a treated HBeAg positive chronic HBV patients.
Serum HBV DNA and HBeAg levels were assessed at baseline and weeks 8 and 12. For each test (HBV DNA level at baseline, HBV DNA decrease between baseline and weeks 8 and 12, or the combination of these two, as well as HBeAg level at baseline, HBeAg decrease between baseline and weeks 8 and 12, and the combination of these two), we calculated the positive predictive value, negative predictive value, sensitivity and specificity.
Monitoring with quantitative HBV DNA levels (area under ROC 0.87) was superior to monitoring with quantitative HBeAg levels (0.76, P < 0.05). Step-wise logistic regression identified HBV DNA at baseline and decrease in HBV DNA from baseline to week 12, as independent predictors of response.
The overall test performance of predicting nonresponse (predictive value 100%) was best for log HBV DNA testing at week 12 compared with testing at week 8 due to a better prediction of sustained response (46%vs 38%) and lower misidentification of nonresponse (39%vs 54%).
This study showed that quantitative HBV DNA testing at baseline in combination with a decrease between baseline and week 12 has a high predictive value for identifying patients who have virtually no chance of reaching a sustained response with IFN therapy.
RESULTS
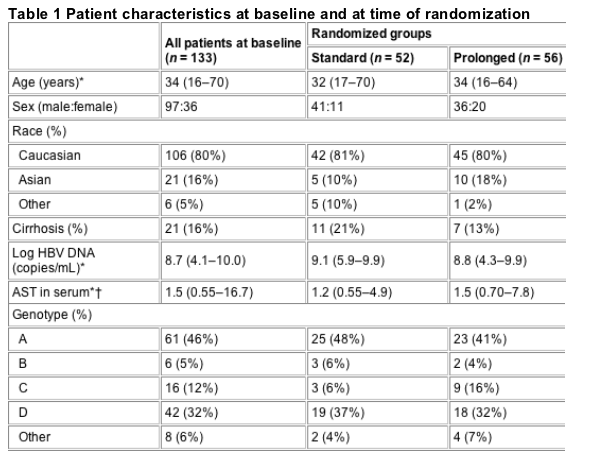
Demographic and clinical characteristics of the patients are given in Table 1.
Correlation between sustained response and pretreatment serum HBV DNA levels, HBeAg levels and ALT levels
The SR had significantly lower pretreatment HBV DNA (P < 0.001) and HBeAg levels (P < 0.001) and significantly higher pretreatment ALT levels (P < 0.05) compared with NR.
Changes in serum HBV DNA levels in sustained responders vs nonresponders
Retrospectively, SR had a significantly larger mean [standard deviation (SD)] log decline in HBV DNA levels, 1.0 (SD 0.79), 1.7 (SD 0.98) and 2.5 (SD 1.38) at weeks 4, 8 and 12 respectively compared with NR, who showed a mean log HBV DNA decrease of 0.5 (SD 0.62), 0.8 (SD 0.89) and 1.0 (SD 1.27), at the respective time points (P < 0.001 for the change from baseline to 4, 8 and 12 weeks). Median log HBV DNA level at time of seroconversion to anti-HBe was 4.2 (range 2.8-7.6).
Correlation between decrease in HBV DNA and genotype
Of 133 patients 61 (46%) had genotype A, 42 (32%) genotype D, 16 (12%) genotype C and 6 (5%) genotype B. The study population consisted of 80% Caucasians, 16% Asians and 5% other. Caucasians were predominantly infected with genotype A (54%) or D (38%), whilst Asians by B (24%) or C (57%). Chronic HBV patients infected with genotype A or D had a higher baseline serum HBV DNA [mean log HBV DNA level 8.6 (SD 1.0)] than chronic HBV patients infected with genotype B or C [mean log HBV DNA level 7.7 (SD 1.2), P < 0.01]. The decrease in log HBV DNA between baseline and week 16 for the different genotypes was 1.6 (SD 1.7) for genotype A, 1.2 (SD 1.4) for genotype D, 2.2 (SD 2.1) for genotype B and 2.3 (SD 1.8) for genotype C. If we compare the decrease in log HBV DNA in the first 16 weeks between genotype A and D [1.5 (SD 1.6)] and between B and C [2.3 (SD 1.8)] a significantly stronger decline in log HBV DNA could be found for genotype B and C (P < 0.005).
Quantitative HBeAg measurements vs quantitative HBV DNA measurements
The objectives of our study were first to evaluate quantitative HBeAg measurements vs quantitative HBV DNA measurements for the prediction of response and nonresponse in IFN-a treated patients. ROC curves were used to compare the discriminative value between the two diagnostics tests. Figure 2 shows the ROC curves for HBV DNA testing at baseline in combination with the HBV DNA decrease between baseline and week 12, and HBeAg testing at baseline in combination with the HBeAg decrease between baseline and week 12. ROC curves show the relationships per test between the chance of correctly identifying an eventual SR vs the chance of a false positive result. An optimal test would approach 100% sensitivity, 0% false positivity, while a test without discriminative value would only reach 100% sensitivity, at 100% false positivity. Differences between curves were evaluated by comparing the area under the ROC curves. The area under the ROC curve was significantly higher for HBV DNA testing at baseline in combination with a HBV DNA decrease between baseline and week 12 (0.87), compared with HBeAg testing at baseline in combination with a HBeAg decrease between baseline and week 12 (0.76 P < 0.05).
Prediction of response and nonresponse at week 12
The second objective of our study was to evaluate the predictive value of precise quantitative HBV DNA measurements for the response and nonresponse of IFN-a treated patients.
The question was whether, as in chronic hepatitis C, quantitative measurements of HBV DNA could predict outcome of response. Stepwise logistic regression analysis identified HBV DNA at baseline and a decrease in HBV DNA as independent predictors of response. We therefore used the combination of these variables to develop a prediction model.
A ROC curve is a graph of the pairs of true positive (= sensitivity) and false positive rates (= 1 - specificity) that correspond to each possible cut-off for the diagnostic test result. Using the outcomes of the quantitative HBV DNA tests as end criteria for early treatment, the clinically most relevant goals are a high predictive value for nonresponse and no exclusion of potential SR. We therefore selected the cut-off that maximized the true positive rate (= sensitivity of 100%) and used the corresponding HBV DNA value at baseline and HBV DNA decrease between baseline and week 12 to calculate the positive and the negative predictive values. Table 2 shows the results of this analysis. If patients had a log HBV DNA level at baseline above 8 in combination with a log decrease <1.0 log between baseline and week 12 the chance of nonresponse was 100%. If this test was used as a limiting criterion, no SR would have been missed. If we compare this with a log HBV DNA at baseline above 9 in combination with 2.5 log decrease between baseline and week 12, the performance of the first test was better. This is due to a better prediction of SR (46%vs 38%) and lesser misidentification of NR (39%vs 54%).
If a log HBV DNA level at baseline below 8 in combination with a HBV DNA decrease between baseline and week 8 or 12 was used, then a sensitivity of 100% in identifying NR could not be accomplished, and therefore treatment with IFN-a could be initiated in this group of patients.
Prediction of non-response at week 8
We investigate whether testing for HBV DNA before week 12 might lead to even greater benefits. The decrease in viral load 8 weeks after the first dose of IFN-a correlates with the decrease after 12 weeks of treatment. (R = 0.85; P < 0.001). Figure 3 shows the ROC curves for log HBV DNA at baseline in combination with the HBV DNA decrease between baseline and week 12 vs log HBV DNA at baseline in combination with the log HBV DNA decrease between baseline and week 8. The areas under the ROC curves were not significantly different between the two tests (0.86 vs 0.85, P = 0.60).
We calculated the positive and negative predictive value, using the values of log HBV DNA level at baseline and log HBV DNA decrease between baseline and week 8, which corresponded with the cut-off that maximized the true positive rate (= sensitivity of 100%) at week 8 (Table 3). If we use the limit of log HBV DNA level at baseline above 8 in combination with a log decrease <1 log between baseline and week 8, two out of 25 (7%) SR undergoing treatment would have been missed. The overall test performance of predicting nonresponse was best for log HBV DNA testing at week 12 compared with testing at week 8 due to a better prediction of SR (46%vs 38%) and lesser misidentification of NR (39%vs 54%).
The predictive value of nonresponse does not change if we correct for duration of therapy. But we can improve the identification of NR by letting the cut off value depend on duration of therapy. For patients with standard therapy with a baseline log HBV DNA level >8 in combination with a viral load decrease between baseline and week 12 of <2 log, the predictive value of nonresponse was 100% and 33% of NR could not be identified. Prolonging therapy for 16 weeks in patients with standard therapy with a log HBV DNA baseline level >8 and viral load decrease between baseline and week 12 of >1 but below 2 logs would lead to a response in 60% (3 of 5) of these patients.
Discussion
To reduce unnecessary exposure to treatment physicians must decide at an early stage whether continuation of treatment has a reasonable chance of success in a patient. The objectives of our study were to assess quantitative HBeAg measurements vs quantitative HBV DNA measurements as predictors of nonresponse and response in IFN-a treated HBeAg positive patients.
Predictive factors for end of treatment response in chronic HBV patients treated with IFN-a have been studied in the past. In a randomized-controlled trial of IFN-a, with or without prednisone priming, Perrillo et al. [2] found that baseline serum HBV DNA level was the most important independent predictor of response (P = 0.003). Approximately 50% of the patients with baseline HBV DNA levels under 100 pg/mL (solution-hybridization assay; Abbott laboratories) responded to treatment with 5 million units of IFN-a compared to only 7% of patients with HBV DNA levels at baseline above 200 pg/mL. A study investigating the post-treatment durability of HBeAg seroconversion following lamivudine, IFN monotherapy or IFN-lamivudine combination therapy also identified pretreatment HBV DNA levels as the major predictor of SR [16]. They also found a significant predictive value of pretreatment ALT level for the durability of HBeAg seroconversion (higher baseline ALT - lower chance of relapse).
Midtreatment HBV DNA levels showed a significant correlation (P < 0.001) with response in Chinese adults with chronic HBV infection [17]. Response was achieved in 53% of patients who had a HBV DNA level below 0.7 Meq/mL (branched DNA assay) at midtreatment, but in only 17% of those who remained HBV DNA positive. In a large prospective, randomized-controlled trial investigating the efficacy of treatment prolongation for an additional 16 weeks in those patients who did not respond with HBeAg seroconversion during the standard 16-week IFN-a course, a low level of HBV DNA (<10 pg/mL) at randomization was found to be the only independent predictor of response (52%vs 0%P < 0.001), while a low HBV DNA level at entry tended towards significance (P = 0.07) [3].
Early monitoring of HBeAg levels during therapy with IFN-a showed that changes in the HBeAg level from the start of therapy to week 8 were significantly related with response at the end of follow up [6]. The present study has shown that quantitative HBV DNA measurements by validated real-time PCR can be used for early identification of NR to IFN-a and that quantitative HBV DNA measurements by this technique can be used for early identification of NR to IFN-a. Monitoring with sensitive quantitative HBV DNA measurements is superior to monitoring with quantitative HBeAg levels. Stepwise logistic regression identified HBV DNA at baseline and a decrease in HBV DNA levels as independent predictors of response; quantitative HBeAg levels did not add to the prognostic value. This is in contrast to earlier published studies where HBeAg levels in pretreatment serum and the decrease in HBeAg level from the start of therapy to weeks 4 and 8 related significantly to response to therapy at week 16 [6]. However, this study was performed using a HBV DNA assay based on hybridization in solution (Genostics; Abbott Laboratories), which has a minimum detection limit of only 107-108 HBV copies/mL.
The overall test performance of predicting nonresponse was best for quantitative HBV DNA testing at baseline in combination with a decrease between baseline and week 12, compared with quantitative HBV DNA testing at baseline in combination with a decrease between baseline and week 8. We therefore suggest that a quantitative HBV DNA test at 12 weeks can be used as a management tool for the decision whether to continue treatment or to adjust it.
This study confirmed the significant relationship between lower pretreatment HBV DNA levels and HBeAg levels, and higher pretreatment ALT levels, associated with SR. This study shows that quantitative HBV DNA testing at baseline in combination with the decrease between baseline and week 12 has a high predictive value in identifying patients who have virtually no chance of reaching a SR with IFN-a therapy. Future research should explore whether this concept also holds for prolonged therapy (≥1 year), for PEG-interferon monotherapy and for patients who receive combination therapy of a nucleoside or nucleotide analogue and (PEG-) IFN.
Introduction
Interferon-alpha (IFN-a) is a powerful immune stimulatory drug used for the treatment of chronic hepatitis B patients. IFN-a treatment increases the spontaneous response rate and leads to loss of hepatitis B e antigen (HBeAg) in 15-40% of patients [1,2]. The most important predictive factors known for response to IFN-a are low baseline hepatitis B virus (HBV) DNA levels and elevated alanine aminotransferase (ALT) levels [2-4]. The most important adverse effects of treatment with IFN-a are flu-like syndrome with fever and myalgia, fatigue, asthenia, anorexia, depression and disturbed concentration [3].
To reduce unnecessary exposure to treatment and its potential side effects as well as to reduce costs, parameters must be found that indicate at an early stage whether continuation of treatment has a reasonable chance of success for the individual patient.
We initiated a large-scale prospective, randomized-controlled trial to investigate the efficacy of prolonging treatment for an additional 16 weeks for those patients who did not respond with HBeAg seroconversion during a standard 16-week IFN-a treatment course [3]. A low level of HBV DNA (<10 pg/mL) at randomization was found to be the only independent predictor of response (52%vs 0%P < 0.001) for prolonged therapy, while a low HBV DNA level at entry tended towards significance (P = 0.07). However, the HBV DNA assay used at that time, i.e. hybridization in solution (Genostics; Abbott Laboratories, Chicago, IL, USA), has a lower limit of detection of 107-108 copies/mL. Meanwhile, more sensitive quantitative virology measurements have become available [5]. The precise quantitative measurement of HBV DNA levels could further optimize response prediction by indicating likely response in the event of continuously decreasing serum HBV DNA.
Quantitative assessment of HBeAg in pretreatment serum and during therapy is considered an option in the literature, as an alternative to monitoring chronic HBV patients during therapy and has been used for the prediction in response in patients treated with IFN-a [6,7]. As validated real-time-based quantitative polymerase chain reaction assays for the measurement of HBV DNA in serum have become available, it is as yet not determined which test should be used to monitor patients during treatment.
Early quantitative hepatitis C virus RNA measurements have been shown to predict sustained virologic response in chronic hepatitis C patients [8,9]. The current National Institute of Health Consensus recommendation is testing at 12 weeks of treatment with peginterferon-alpha and ribavirin. Chronic hepatitis C patients who fail to achieve a drop of 2 logs in viral load at week 12 should not be treated further [10]. The question arose whether quantitative HBV DNA measurements at week 12 could be used to predict nonresponse of chronic hepatitis B patients treated with IFN-clinically our most relevant goals are a high predictive value for nonresponse without exclusion of potential sustained responders.
The objectives of our study were first to evaluate quantitative HBeAg measurements vs quantitative HBV DNA measurements for prediction of nonresponse and response in IFN-a treated patients. Secondly, we determined the value of precise quantitative HBV DNA measurements for predicting nonresponse and response in IFN-a treated patients.
Patients and methods
Patients
For this study, data from patients treated in a large randomized-controlled trial performed in 16 European centres (EUROHEP) were used [3]. In this trial, a standard 16-week treatment protocol (10 million units, three times a week) was compared with prolonged treatment for 32 weeks (10 million units, three times a week). All patients who were still HBeAg-positive at week 16 were randomized to undergo either no further treatment or prolonged treatment. For the present study, 29 patients were excluded for discontinuation of therapy in an early phase (n = 12) and insufficient serum (n = 17). A total of 133 patients participated eventually in our study (Fig. 1).
Virologic measurements
The HBV DNA assessments were performed routinely every 4 weeks during treatment and every 4-8 weeks during the follow-up period until week 52. If a week 0 sample was not available for assessment of HBV DNA at baseline, a sample from week -4 (total 23) or week -8 (total 8) was used. Quantitative HBeAg measurements were carried out using the AxSYM HBe 2.0 Quantitative test (Abbott Laboratories), as described previously [11]. All HBV markers were assessed centrally.
Isolation of HBV DNA was performed using the MagnaPure LC isolation station (Roche Applied Science, Penzberg, Germany) with a modified protocol with proteinase K digestion initially [5]. The HBV DNA TaqMan assay, calibrated according to EUROHEP HBV DNA standards [12], was used for the quantitative measurement of HBV DNA in serum [5].
The HBV genotyping was performed with a line probe assay (INNO-LiPA HBV DR; Innogenetics N.V., Ghent, Belgium). The INNO LiPA HBV DR assay was performed essentially as described previously [13]. In case of a missing sample at baseline a sample taken at a timepoint close to baseline was used for genotyping.
Response criteria
For the definition of sustained response (SR) at week 52 we used the definition of virologic response recommended by the National Institute of Health workshop on chronic hepatitis B [14]. Sustained virologic response is defined as a loss of HBeAg, as indicated by AxSYM (<0.27 PEI U/mL; Abbott laboratories), together with a decrease in HBV DNA <105 copies/mL at week 52. Patients not fulfilling these criteria were considered nonresponders (NR). The early virologic on-treatment responses were evaluated to determine their ability to predict response and nonresponse at week 52.
Statistical analyses
For each test (HBV DNA level at baseline, HBV DNA decrease between baseline and week 8 and week 12, and the combination of these two, HBeAg level at baseline, HBeAg decrease between baseline and week 8 and 12, and the combination of these two), we calculated the positive predictive value (% SR if the test is normal), its negative predictive value (% NR if test is abnormal), its sensitivity (% SR identified by test) and its specificity (% NR identified by test) using the 2 X 2 method. Because of its clinical relevance, we also calculated the reverse forms of the sensitivity and specificity i.e. the fraction of all SR not identified by the test and the fraction of NR not identified by the test. For all tests the areas under the receiver operating characteristic (ROC) curves were calculated and compared according to the method described by Delong et al. [15]. Backward stepwise logistic regression was used to determine the best set of independent predictors of nonresponse.
Chi-square testing was used for categorical variables. Statistical analysis of differences between groups was performed with the Mann-Whitney U-test. The Pearson correlation coefficient was calculated for log values of HBV DNA at 8 and 12 weeks. A P-value of <0.05 was considered significant. All calculations were performed with SPSS software (SPSS Inc, Chicago, IL, USA).
|
|
| |
| |
|
|
|