| |
Entecavir (ETV) vs. Adefovir (ADV): HBV DNA Reduction In Chronically Infected HBeAg(+) Nucleoside-Naive Adults In A 12-Week Viral Kinetics Study
|
| |
| |
Reported by Jules Levin
Presented at the NIH Management of Hepatitis B Conference, April 6 2006
R Wilber1, H Brett-Smith1, J Zhu1, K Mencarini1, R Colonno1, A Cross1, JC Lopez-Talavera1 for the Entecavir Viral Kinetics Group
1. Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, CT, USA
Abstract
Background: Elevated HBV DNA is associated with increased risk of disease progression and complications, such as hepatocellular carcinoma. Antiviral strategies should aim at rapidly and extensively reducing viral load and replication. To date, there are no direct comparisons of the antiviral activity of ETV vs. ADV. We compared the early antiviral activity and viral kinetic profiles of ETV vs. ADV in HBeAg+ nucleoside-naive adults chronically infected with HBV.
Methods: This was a randomized, open label, comparative study of ETV 0.5 mg QD vs. ADV 10 mg QD. Serum HBV DNA was assessed daily from Day 1 to 14 and then at weeks 3, 4, 6, 8, 10 and 12. Two biphasic models were fit separately to each treatment: (1) 3-parameter spline model to estimate slopes and (2) 4-parameter exponential decay model to estimate effectiveness and free virus half-life. Significance was based on 2-sided t-tests.
Results:
Mean baseline HBV DNA was 10.45 (ETV) and 9.89 (ADV) log10
copies/mL. Mean change (log10 copies/mL) of HBV DNA at Week 12 was -6.23 for ETV (n=33) and -4.42 for ADV (n=32) patients (p<0.0001).
All ETV patients experienced a minimum reduction of at least 3.88 log10 copies/mL compared to 0.95 log10 copies/mL for ADV patients.
There was a higher variability of viral load reduction for ADV compared to ETV. Treatment resulted in a biphasic decline in both arms with a first-phase rapid decline lasting 10 days, yielding a calculated circulating virus half life of 14 hr (ETV) and 26 hr (ADV).
A significant difference between the arms favoring ETV was reached as early as Day 10.
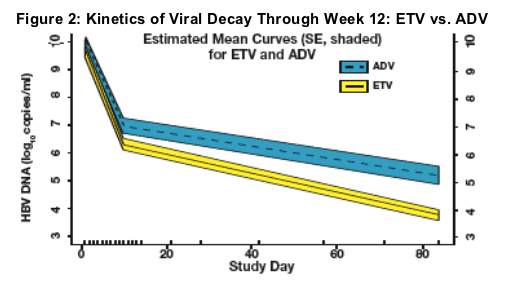
At 12 weeks, 52% (17/33) of ETV patients had HBV DNA below 10,000 copies/ml compared to 25% (8/32) for ADV patients.
Both antivirals were well tolerated and had comparable safety profiles
throughout the study.
Conclusions: ETV showed superior antiviral activity compared to ADV by rapidly and extensively reducing HBV DNA in nucleoside naive, HBeAg(+) patients.
RESULTS
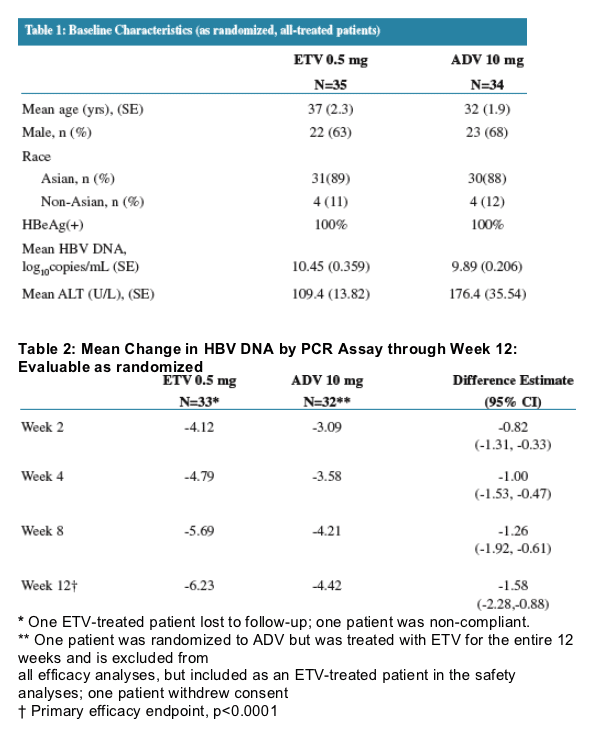
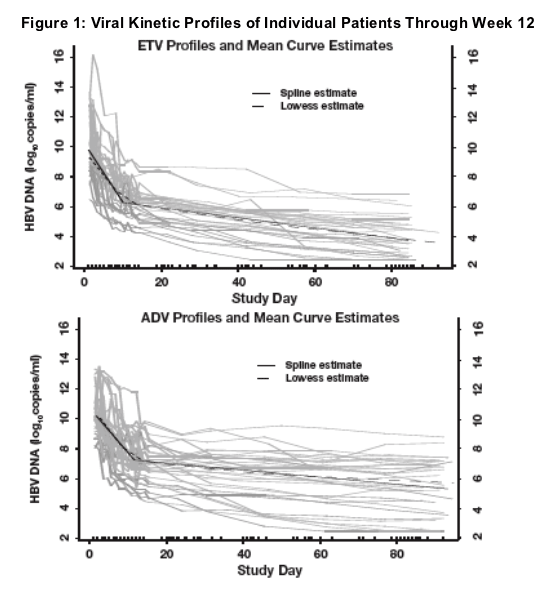
- Treatment resulted in a biphasic decline in both treatment arms, with a first phase rapid decline lasting 10 days
- 95% confidence intervals of the estimated spline differences between ETV and ADV indicate ETV significantly decreases HBV DNA (log10 copies/mL) at Day 10 and thereafter, compared to ADV
- The parameter estimates for free virus half-life were 14 hours for ETV and 26 hours for ADV
- At Week 12, 52% (17/33) of ETV-treated patients achieved HBV DNA less than 104 copies/mL, versus 25% (8/32) of ADV-treated patients (figure 3)
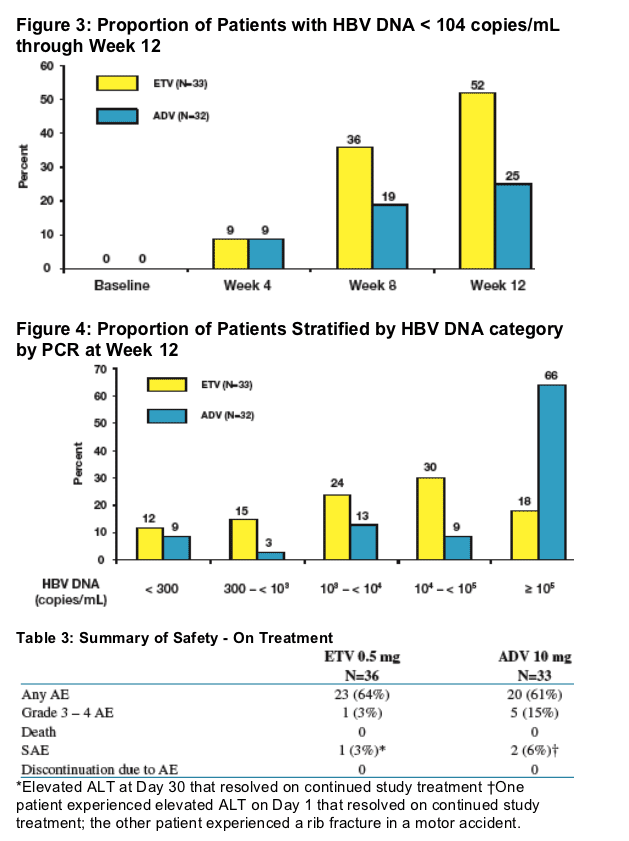
Conclusions
- ETV 0.5 mg was superior to ADV 10 mg for the primary efficacy endpoint of mean change from baseline in HBV DNA at Week 12
- Both ETV and ADV demonstrated biphasic viral kinetics, with a first phase of rapid decline lasting 10 days
- A significant difference between the treatment arms in favor of ETV was reached as early as Day 10. There was a higher variability of viral load reduction for ADV compared to ETV
- 66% of ADV-treated patients demonstrated HBV DNA >105 copies/mL at Week 12, compared to 18% of ETV-treated patients
- The safety profile of ETV was comparable to that of ADV
References
1. Chang TT, Gish RG, de Man RA, et al. A comparison of entecavir and lamivudine for HBeAg-positive chronic hepatitis B. N Engl J Med 2006;354:1001-1010.
2. Lai CL, Shouval D, Lok AS, et al. Entecavir versus lamivudine for patients with
HBeAg-negative chronic hepatitis B. N Engl J Med 2006; 354:1011-1020
3. Neumann AU, Lam NP, Dahari H, et al. Hepatitis C viral dynamics in vivo and the antiviral efficacy of interferon-alpha therapy. Science 1998; 282(5386): 103-107
|
|
| |
| |
|
|
|