| |
Entecavir for Treatment of Lamivudine-Refractory, HBeAg-Positive Chronic Hepatitis B
|
| |
| |
Gastroenterology June 2006
Volume 130, Issue 7, Pages 2039-2049
Data presented in part at the 55th Annual Meeting of the American Association for the Study of Liver Diseases 2004 (Abstract 1152), the 14th Biennial Meeting of the Asia Pacific Association for the Study of the Liver 2004 (Abstract 46), the Therapies for Viral Hepatitis Workshop 2004 (Abstract 42), the 1st Annual Winter Meeting of the Canadian Association for the Study of the Liver 2005 (Abstract 91), and the 13th World Congress of Gastroenterology 2005 (Abstract R.0780).
Morris Sherman, Cihan Yurdaydin, Jose Sollano, Marcelo Silva, Yun-Fan LiawÅa, Janusz Cianciara#, Anna Boron-Kaczmarska, Paul Martin, Zachary Goodman, Richard Colonno, Anne Cross, Gail Denisky, Bruce Kreter, Robert Hindes
Department of Medicine, Toronto General Hospital, Toronto, Ontario, Canada
Ankara University Medical School, Ankara, Turkey
University of Santo Tomas, Manila, Philippines
Hospital Universitario Austral, Pilar, Argentina
Åa Chang Gung Memorial Hospital and University, Taipei, Taiwan
# Medical University of Warsaw, Warsaw, Poland
Department of Infectious Diseases, Pomeranian Medical University, Szczecin, Poland
Mount Sinai School of Medicine, New York, New York
Armed Forces Institute of Pathology, Washington, DC, USA
Bristol-Myers Squibb Pharmaceutical Research Institute, Princeton, New Jersey, and Wallingford, Connecticut
ABSTRACT
Background & Aims: Lamivudine treatment is associated with frequent development of resistant hepatitis B virus (HBV) and loss of treatment benefit. In preclinical and phase II studies, entecavir demonstrated potent antiviral activity against lamivudine-resistant HBV.
Methods: In this phase III, double-blind trial, hepatitis B e antigen-positive patients who were refractory to lamivudine therapy (persistent viremia or documented YMDD mutations while receiving lamivudine) were randomized to switch to entecavir 1 mg daily (n = 141) or continue lamivudine 100 mg daily (n = 145) for a minimum of 52 weeks. Two coprimary end points were assessed at 48 weeks: histologic improvement and a composite end point (HBV branched DNA <0.7 MEq/mL and alanine aminotransferase [ALT] <1.25 times the upper limit of normal).
Results:
Histologic improvement occurred in 55% (68/124) of entecavir-treated vs 28% (32/116) of lamivudine-treated patients (P < .0001).
More patients on entecavir than lamivudine achieved the composite end point: 55% (77/141) vs 4% (6/145), respectively (P < .0001).
Mean change from baseline in HBV DNA was -5.11 log10 copies/mL for entecavir-treated patients and -0.48 log10 copies/mL for lamivudine-treated patients (P < .0001).
Virologic rebound because of entecavir resistance substitutions occurred in 2 of 141 of entecavir-treated patients, and genotypic evidence of resistance was detected in 10 patients.
The safety profile of entecavir was comparable to lamivudine with fewer ALT flares on treatment.
Conclusions: In patients with lamivudine-refractory chronic hepatitis B, switching to entecavir provides superior histologic improvement, viral load reduction, and ALT normalization compared with continuing lamivudine, with a comparable adverse event profile.
Background
Chronic hepatitis B virus (HBV) infection has a worldwide prevalence of over 400 million people, which represents more than 5% of the global population.1 These patients are at increased risk for development of hepatic cirrhosis, liver failure, and hepatocellular carcinoma (HCC), and over 1 million people die each year from these HBV-associated complications.2 A recently reported cohort study has shown that, among chronic hepatitis B patients, elevated HBV DNA levels are associated with the development of cirrhosis3 and of HCC.4 A principal goal of therapy in patients with chronic hepatitis B infection is to arrest and reverse progression of liver damage by suppressing HBV replication.5
Lamivudine was the first nucleoside analogue to be introduced as therapy for chronic hepatitis B infection and remained the only available nucleoside antiviral for several years. A major limitation of lamivudine therapy is the emergence of drug-resistant HBV from substitutions at M204I/V (within the tyrosine-methionine-aspartate-aspartate YMDD motif of the viral DNA polymerase) that are frequently accompanied by an L180M substitution. Genotypic evidence of lamivudine resistance precedes virologic rebounds by several months. Although lamivudine-resistant strains of HBV emerged in up to 24% of patients after 1 year and as many as 70% of patients after 4 years of treatment, the clinical implications of lamivudine resistance were not immediately evident.6, 7
Early studies found that patients with lamivudine-resistant HBV who continued to receive lamivudine maintained lower serum HBV DNA and alanine aminotransferase (ALT) levels than baseline and continued to experience hepatitis B e antigen (HBeAg) seroconversion (albeit at a lower rate than patients with wild-type HBV).8, 9, 10 In addition, discontinuation of lamivudine in patients with lamivudine-resistant HBV was associated with hepatic flares.11 Thus, continuing lamivudine therapy was recommended and generally accepted, despite emergence of resistance substitutions.12, 13 It has subsequently been shown that, although lamivudine-resistant mutants are replication impaired, additional compensatory mutations are selected during continued lamivudine exposure that can restore replication fitness.14, 15, 16
Accumulated evidence now points to the negative consequences of continuing lamivudine after the development of resistance. Increases in viral replication and serum ALT levels,17, 18, 19, 20, 21 flares of hepatic disease,11, 19, 22 reversal of previous histologic improvement,8, 23 and decreased rates of HBeAg seroconversion9, 10 may occur. Liaw et al recently found no benefit to continuing lamivudine therapy compared with stopping treatment in patients with lamivudine-resistant HBV.24 Among chronic hepatitis B patients with compensated cirrhosis, lamivudine resistance has been associated with hepatic decompensation and the development of HCC.25, 26
When this study began, the only nucleoside treatment option for patients who had developed lamivudine resistance was continuation of lamivudine. Treatment guidelines at the time recommended continuing lamivudine to avoid reappearance of wild-type virus and to maintain viral load and ALT below baseline levels.12, 13 A double-blind, phase II dose-ranging study of entecavir in HBeAg-positive and -negative lamivudine-refractory patients found that a 1 mg dose of entecavir for 48 weeks provided a reduction in HBV DNA of greater than 5 log10 copies/mL, with a safety profile comparable with that of lamivudine.27 Therefore, in this phase III trial in lamivudine-refractory patients, switching patients to entecavir 1 mg daily was compared with continuing lamivudine 100 mg daily for impact on liver histology, suppression of viral load, serologic response, and normalization of ALT.
Results
Study Population
Of 420 patients enrolled and screened, 293 were randomized (Figure 1). Patients were not randomized for the following reasons: 108 did not meet inclusion criteria; 18 withdrew consent; and 1 completed screening after randomization closed. Two hundred eighty-six patients received at least 1 dose of blinded study drug (entecavir 141, lamivudine 145 patients). The 2 treatment groups were well-balanced for demographics and disease characteristics at baseline (Table 1). Overall, 54% of treated patients had received interferon prior to study entry. Five (4%) patients in the entecavir group and 3 (2%) patients in the lamivudine group were positive for HBeAg during screening and negative for HBeAg at baseline. Of the 11 entecavir-treated and 4 lamivudine-treated patients with seroconversion at week 48, 2 entecavir-treated and 1 lamivudine-treated patient seroconverted in the period between screening and randomization; these patients continued in the study. Of the 286 treated subjects, 281 (98%) had a baseline liver biopsy, and 240 patients (84%) had evaluable baseline liver biopsies.
Figure 1. Study design. Response at week 48 was defined as undetectable HBV DNA by bDNA assay (<0.7 MEq/mL) and loss of HBeAg. Virologic-only response at week 48 was defined as undetectable HBV DNA by bDNA assay, with persistent HBeAg positivity. Nonresponse was defined as HBV DNA ≥0.7 MEq/mL by bDNA assay at week 48. Patients achieving the protocol-defined response were to discontinue therapy and be followed for 24 weeks off treatment. Patients achieving a virologic-only response were to continue blinded study medication until week 96 or until a response was achieved, whichever occurred first. Patients with nonresponse were to discontinue blinded therapy and begin marketed anti-HBV therapy as recommended by their physician or enroll in an entecavir rollover protocol.
ALT was 123-131
Knodel infalammartory score: 6.5 on both arms.
Knodell fibrosis score: 1.7-1.8
Bridging fibrosis: 23% in ETV arm, 31% in LAM arm.
Cirrhosis: 10% in ETV arm, 6% in LAM.
Prior IFN: 52-55%
Most randomized patients (88%) completed the first year of dosing. During the first year, fewer patients in the entecavir group (<1%) than in the lamivudine group (5%) discontinued treatment because of adverse events. Five lamivudine-treated patients (3%) and 2 entecavir-treated patients (1%) withdrew consent during the first year. A greater proportion of patients in the entecavir group than in the lamivudine group continued to the second year of treatment (62% vs 19%, respectively), primarily because of achieving a virologic-only response at week 48.
Histologic Response
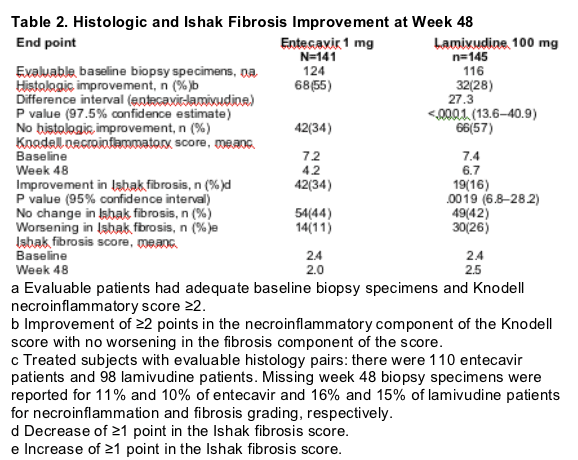
Compliance with week 48 biopsies was high; 251 (88%) had a week 48 biopsy. The observed rate of missing biopsies was slightly higher in the lamivudine group (16%) vs the entecavir group (12%). This difference was largely due to the greater number of discontinuations because of adverse events in the lamivudine group. Entecavir was superior to lamivudine for the proportion of patients demonstrating histologic improvement at week 48, with 55% and 28%, respectively, achieving this coprimary end point (P < .0001) (Table 2). Entecavir was also superior to lamivudine for the proportion of patients demonstrating improvement in the Ishak fibrosis score (34% vs 16%, respectively, P = .0019). Fewer patients in the entecavir group (11%) than in the lamivudine group (26%) experienced worsening of fibrosis, defined as a ≥1-point increase in the Ishak fibrosis score compared with baseline.
Response for the Composite End Point
Fifty-five percent of entecavir patients achieved the composite end point (HBV DNA <0.7 MEq/mL and ALT <1.25 X ULN) by week 48 compared with 4% of lamivudine patients (P < .0001) (Table 3).

Virologic Response
The mean change from baseline in HBV DNA by PCR assay was -5.11 log10 copies/mL for entecavir and -0.48 log10 copies/mL for lamivudine at week 48 (P < .0001; Table 3). As shown in Figure 2, entecavir-treated patients experienced marked suppression of HBV DNA by week 24, with additional viral load reduction between weeks 24 and 48. Entecavir was also superior to lamivudine for the proportion of patients achieving HBV DNA <300 copies/mL at week 48 (19% vs 1%, respectively, P < .0001; Table 3). Among patients with PCR measurements during treatment, 1 entecavir-treated patient and 94 lamivudine-treated patients had less than a 1.0 log10 decline in HBV DNA.
Serologic Response
Loss of HBeAg occurred more frequently among entecavir- than among lamivudine-treated patients (10% vs 3%, respectively; P < .0278). HBeAg seroconversion occurred in 8% of entecavir-treated and 3% of lamivudine-treated patients (P = .06).
Biochemical Response
A significantly larger proportion of entecavir- than lamivudine-treated patients achieved ALT normalization (ALT ÅÖ1 X ULN) at week 48 (61% vs 15%, respectively, P < .0001; Table 3 and Figure 3).
Protocol-Defined Responses
Thirteen (9%) entecavir-treated patients and 1 (<1%) lamivudine-treated patient achieved a protocol-defined response (HBV DNA <0.7 MEq/mL and loss of HBeAg) at week 48 (P = .0008). Eighty (57%) entecavir-treated patients and 7 (5%) lamivudine-treated patients achieved a virologic-only response (HBV DNA <0.7 MEq/mL without HBeAg loss) at week 48. Nonresponse (HBV DNA ≥0.7 MEq/mL) occurred in 40 (28%) patients on entecavir and 121 (83%) patients on lamivudine. Of the responders, 5 of 13 (38%) entecavir-treated patients and 1 lamivudine-treated patient sustained their response (HBV DNA <0.7 MEq/mL and HBeAg loss) 24 weeks after treatment discontinuation.
Baseline Predictors of Response
An examination of baseline disease characteristics and treatment outcomes at 48 weeks identified several possible predictive factors for response. Observations were limited to the entecavir treatment group because of low response rates in the lamivudine population. As pretreatment ALT levels increased from <2 X ULN to >5 X ULN, the proportions of patients with histologic improvement and undetectable HBV DNA increased, whereas the proportion of patients with normalization of ALT decreased. Differences in baseline HBV DNA levels did not affect histologic improvement or ALT normalization.
Resistance
Among the 286 patients treated, over 85% had lamivudine-resistance substitutions (M204I/V ± L180M) present at baseline (Table 1). These lamivudine-resistant mutations result in an 8-fold reduction in sensitivity to entecavir in cell-based assays. However, intracellular entecavir triphosphate levels are predicted to exceed the level necessary to achieve viral inhibition.30 Virologic rebounds in entecavir-treated patients have only been observed in patients with lamivudine-resistant virus (M204V/I and/or L180M) that have an additional substitution at residues T184, S202, or M250.29, 31 Evidence for these substitutions was observed at baseline (before exposure to entecavir) in 18 (6%) lamivudine-refractory patients, and the substitutions appear to be secondary changes selected while receiving lamivudine therapy. Genotypic analysis of paired baseline and 48-week samples was performed from 134 of the 141 entecavir-treated patients; 7 patients were identified with substitutions at 1 or more of these entecavir-resistant residues at baseline. None of these 7 patients exhibited virologic rebound during the first year of therapy. Two other patients (1.4%) exhibited a confirmed virologic rebound (≥1 log10 increase from nadir by PCR assay) during the first year of treatment due to emergence of entecavir-resistant mutants.
Safety
Mean exposure to study therapy was 63 weeks for entecavir vs 52 weeks for lamivudine. The frequencies of adverse events and serious adverse events were similar between treatment groups (Table 4). The most frequent adverse events, occurring in ≥10% of patients in either treatment group, were upper respiratory tract infection, headache, fatigue, upper abdominal pain, cough, nausea, nasopharyngitis, and increased ALT. These events generally occurred with comparable frequency in the entecavir and lamivudine groups, with 2 exceptions: increased ALT occurred more frequently in the lamivudine group (10% vs 4% in the entecavir group), and upper respiratory tract infection occurred more frequently in the entecavir group (18% vs 11% in the lamivudine group). Most adverse events were mild to moderate in severity. Fewer entecavir-treated patients (1%) than lamivudine-treated patients (7%) discontinued study therapy because of adverse events.
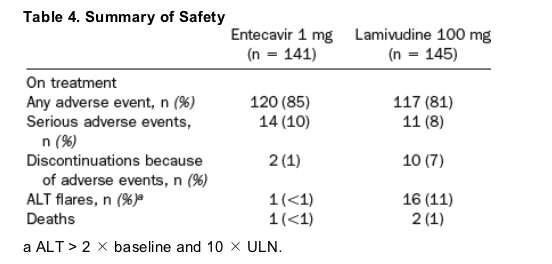
Fewer entecavir- than lamivudine-treated patients experienced an ALT flare (ALT >2 X baseline and 10 X ULN) during treatment (<1% vs 11%, respectively; Table 4). The single ALT flare in the entecavir group occurred on day 29 of treatment and was associated with a decline in HBV DNA to <0.7 MEq/mL that was maintained throughout the study. The flare was not associated with signs or symptoms of hepatic decompensation, and it resolved without treatment interruption after 57 days. In the lamivudine group, all flares were associated with stable or rising HBV DNA levels, and 1 flare was associated with hepatic decompensation and death. Five patients in the lamivudine group discontinued treatment because of ALT flares.
There were 3 deaths; none was judged related to the study medication by the investigator. One lamivudine-treated patient died of septic shock secondary to acute cholecystitis at week 44 of study therapy. One entecavir-treated patient, in whom splenic lymphoma was diagnosed during week 16 of study therapy, died of a gastrointestinal hemorrhage at week 96. One lamivudine-treated patient died of hepatic failure approximately 20 weeks after the last dose of study medication. This patient had discontinued lamivudine because of an ALT flare at week 52 and received no alternative anti-HBV therapy after treatment discontinuation (Table 4).
Discussion
To date, this is the largest randomized, controlled trial of nucleoside therapy for chronic hepatitis B in a lamivudine-resistant population and the only trial in this population to use liver histology as a primary end point. Patient eligibility for the study required the persistence or recurrence of viremia on long-term lamivudine treatment or documented lamivudine-resistance mutations. These entry criteria reflect the real-world situation wherein clinical failure is not always linked to documented viral mutations; however, over 85% of the lamivudine-refractory patients in this study also demonstrated lamivudine-resistance substitutions by genotypic analysis. In addition, 54% of patients in this trial had failed prior interferon therapy, thus representing a particularly treatment-refractory study population now receiving a third course of therapy.
The results of this study for the treatment of patients with lamivudine-resistant HBV with entecavir are encouraging. Entecavir demonstrated superior responses compared with lamivudine across histologic, virologic, serologic (HBeAg loss), and biochemical end points. The diminished efficacy demonstrated by continued lamivudine is consistent with literature suggesting its relative lack of benefit in patients with lamivudine-resistant chronic hepatitis B.24 In contrast, approximately 50% of patients treated with entecavir showed improvement in necroinflammation, and over 33% of patients experienced an improvement in fibrosis within 48 weeks of therapy. This is promising for patients who suffer ongoing liver damage because of infection with lamivudine-resistant HBV. Twenty-six percent of patients on lamivudine in this study (vs 11% on entecavir) experienced worsening of fibrosis. This is consistent with the results of recent studies that suggest that continued treatment with lamivudine after the emergence of lamivudine-resistant virus does not halt the progression of liver disease.23
Entecavir strongly suppressed HBV DNA levels in this trial. The mean HBV DNA reduction at 48 weeks was 5.11 log10 copies/mL for entecavir (vs 0.48 log10 copies/mL for lamivudine), and 19% of entecavir-treated patients vs 1% of lamivudine-treated patients achieved HBV DNA below the limit of detection by PCR of 300 copies/mL. Viral suppression by entecavir compares favorably with that reported in patients with lamivudine-resistant HBV receiving adefovir; Peters et al reported a mean HBV DNA reduction of 4.00 log10 copies/mL after 1 year of adefovir treatment.32
An alternative approach to treating lamivudine-resistant HBV is combination therapy. Although the therapeutic advantage of combining nucleosides(tides) has not been conclusively demonstrated, the combination of entecavir with a second nucleoside(tide), which does not select for the same pattern of resistance substitutions, might offer an advantage compared with sequential monotherapy. The viral load reduction seen with entecavir in the current trial was also comparable for viral load reduction with the combination of adefovir plus lamivudine for patients with lamivudine-resistant HBV.33
Lamivudine-treated patients in this trial, approximately half of whom had failed prior interferon therapy, experienced a markedly low HBeAg seroconversion rate (3% vs the spontaneous HBeAg seroconversion rate of 5%-10%),34 suggesting that this heavily pretreated population is particularly refractory to therapy. Entecavir-treated patients achieved seroconversion at a rate of 8% after 48 weeks; longer term therapy with entecavir may result in a higher proportion of patients achieving HBeAg seroconversion. The rate of sustained HBeAg loss off-treatment seen in this trial (5/13, 38%) may reflect the absence of a consolidation period of therapy following initial disappearance of HBeAg.5, 35
The results of this clinical trial confirm in vitro observations of the antiviral activity of entecavir. In vitro studies have shown that, with a 50% effective concentration (EC50) of 4 nmol/L, entecavir is over 300-fold more potent in cell culture than lamivudine for inhibition of wild-type HBV.36, 37 Although susceptibility decreases 8-fold in the presence of lamivudine-resistance substitutions L180M and M204V (EC50 = 0.026 É mol/L), intracellular concentrations of entecavir-triphosphate are predicted to exceed those needed for activity against lamivudine-resistant HBV.30
In this trial of lamivudine-refractory patients, virologic rebound from emergence of entecavir resistance was observed in 2 patients (<2%). The genotypic changes within the reverse transcriptase of HBV in these patients match the pattern after virologic rebound in 2 patients with prior lamivudine therapy who were subsequently treated with entecavir.29 Reduction of entecavir sensitivity and possible virologic failure require preexisting lamivudine-resistance substitutions and an additional substitution at residues T184, S202, or M250. In the absence of preexisting lamivudine-resistance mutations, these entecavir-associated substitutions cause minimal reduction of in vitro sensitivity to entecavir and have not been detected in clinical samples.29 Approximately 6% of the patients in this trial were found to have mutations associated with entecavir resistance at baseline, indicating that lamivudine independently selects for these primary entecavir-resistance changes. Apart from the patients with detectable entecavir-resistance changes at baseline, it is difficult to know precisely when these changes emerged. However, these resistant viruses are highly growth impaired and may take several months or years before they become the dominant viral population and cause virologic rebound. In any case, adefovir is active against entecavir-resistant HBV and could be considered as salvage therapy. In contrast, there was no evidence of genotypic or phenotypic resistance among the 310 samples from nucleoside-naive HBeAg-positive and -negative patients in phase III trials treated with entecavir for up to 96 weeks. Also, entecavir did not select for lamivudine-resistance substitutions in these nucleoside-naive populations.31, 38
Because the design of this study did not permit interruption of lamivudine treatment prior to randomization nor overlap of lamivudine and entecavir therapies, the consequences of switching directly to entecavir vs continuing and extending lamivudine treatment could be examined. Both extending lamivudine treatment and switching therapies can result in ALT flares. Among patients receiving long-term lamivudine treatment, rates of ALT flares are greater in patients with lamivudine-resistant HBV than in those with wild-type virus.22, 39 Switching or stopping therapy in this population can induce ALT flares; Chen et al found that 77% of patients stopping lamivudine therapy for lamivudine-resistant HBV exhibited ALT flares within 4 months vs 30% who continued lamivudine treatment.11 Peters et al have shown that lamivudine-resistant patients who switch from lamivudine to adefovir experience grade 3 ALT elevations (ALT >5-10 X ULN) at a rate of 37%, thus resulting in the recommendation to add adefovir to continued lamivudine.32 In the present study, the low rate (<1%) of ALT flares during entecavir treatment confirms findings from a phase II entecavir trial that the risk of ALT flare is low for patients with lamivudine-resistant virus who switch directly to, or are maintained on, entecavir.27
Based on its extensive use in both chronic hepatitis B and human immunodeficiency virus infections, lamivudine is established as a well-tolerated antiviral therapy. The comparable safety profiles of entecavir and lamivudine in this study provide evidence that entecavir is also well tolerated in patients with lamivudine-resistant chronic hepatitis B.
In summary, this large phase III trial demonstrated that 48 weeks of entecavir treatment resulted in clinically significant histologic, virologic, serologic, and biochemical benefits in patients with lamivudine-resistant chronic hepatitis B, with a safety profile comparable with that of lamivudine. Based on the results of this and a previous phase II trial27 in this difficult-to-treat population, even better outcomes may be achieved with a longer duration of entecavir therapy.
|
|
| |
| |
|
|
|