| |
HBV+HCV+HDV-dynamic diseases when in combination
|
| |
| |
"Longitudinal evaluation reveals a complex spectrum of virological profiles in hepatitis B virus/hepatitis C virus-coinfected patients"
Hepatology
Jan 2006
.....in HDV-neg patients.,..considering that patients presenting with phases of viral reactivation and high viremia levels alternate with phases of apparent viral suppression, and a decreased amount of circulating virions must be classified as carriers of active infection, the virological behavior at the end of the follow-up led us to make diagnoses differing from those made when only a single time point had been evaluated in approximately one third of the patients.....in HDV pos patients.....Fifteen of the 30 patients included in this group showed persistently inactive HBV and HCV infections, while the other 15 had active HBV (9 cases) or HCV (5 cases) or both (1 case).....
"..... HBV/HCV coinfection is associated with severe forms of chronic liver disease that generally have a serious prognosis and high risk of death, mostly because of their propensity to progress toward cirrhosis and to develop hepatocellular carcinoma.....very little is known from both the clinical/virological and therapeutic points of view..... there is no scientific evidence indicating the most appropriate follow-up for coinfected patients, and no guidelines have been established for their treatment..... we observed that approximately one third of the cases may present alternate phases of inhibition and recurrence of the activity of one or both the viruses, as revealed by broad changes over time of the amount of circulating HBV and/or HCV genomes. The exact identification of the viral infections primarily responsible for liver injury can only be reached via the longitudinal evaluation of the virological patterns.... HBV and HCV viremia longitudinal analyses also provided interesting information when HDV-positive cases were tested. First, we found that half of these patients had evidence of HBV and/or HCV active replication, and such individuals appear more frequently to have cirrhosis compared with those carrying constantly inactive HBV and HCV infections..... Both HBV and HCV showed wide fluctuations of the viremia levels in several HDV patients...."
Authors: Giovanni Raimondo 1 , Maurizia R. Brunetto 2, Patrizia Pontisso 3, Antonina Smedile 4, Anna Maria Maina 2, Carlo Saitta 1, Giovanni Squadrito 1, Natascia Tono 3, the Associazione Italiana Studio Fegato (AISF) Cooperative Group
1Unit of Clinical and Molecular Hepatology, Department of Internal Medicine, University of Messina, Messina, Italy
2Gastroenterology and Hepatology Division, Santa Chiara Hospital, Pisa, Italy
3Department of Clinical and Experimental Medicine, University of Padua, Padua, Italy
4Department of Gastroenterology, University of Turin, Turin, Italy
Hepatitis B virus (HBV) and hepatitis C virus (HCV) infections account for the majority of liver disease cases worldwide. They share modes of transmission, and their combined infection is a fairly frequent occurrence - particularly in areas where the two viruses are endemic and are found among subjects with a high risk of parenteral infection.[1-6] The classic form of chronic HBV/HCV infection - identified by the contemporaneous positivity of the HBV surface antigen (HBsAg) and the antibody to HCV (anti-HCV) - occurs in a sizable proportion of chronic hepatitis patients,[1][3][5][7-9] and is generally considered a condition favoring the progression of liver fibrosis and the establishment of cirrhosis.[10-12] It also represents one of the most important risk factors for the development of hepatocellular carcinoma,[13-16] even more significant than that observed in the case of single HBV or HCV infection.[17][18] Despite its considerable clinical importance, scant information is currently available on the treatment of the HBV/HCV coinfected population,[19] although the few available studies in the field agree that this form of chronic viral hepatitis is difficult to cure.[20-24] In this context, it is noteworthy to focus attention on the several reports suggesting that HBV and HCV interact in the case of coinfection (reviewed by Alberti et al.[5] and Liaw[11]). In particular, in vitro studies indicate that HCV is capable of suppressing HBV activity and that this inhibitory effect is essentially mediated by the HCV core protein.[25-28] A certain number of in vivo studies also indicate a possible interplay between the two viruses, in some cases confirming a prevalent role of HCV,[2][29-32] while other reports suggest a reciprocal interference or even a dominant effect of HBV.[1][3][6][33][34] This confusing information about HBV and HCV behavior in the case of dual infection has been generated primarily by cross-sectional studies, whereas comprehensive longitudinal evaluation of HBV and HCV activities in this condition has not yet been performed.
To clarify the virological profiles that may occur in the case of HBV/HCV combined infection, it is essential to better understand the biological and clinical features of this peculiar condition and to define the best therapeutic strategies for its treatment. This Italian multicenter study investigated the virological characteristics of chronic HBsAg/ anti-HCV positive patients through the longitudinal evaluation of an adequate number of coinfected individuals. Clinical aspects that could correlate with the different virological patterns were identified.
Abstract
Hepatitis B virus (HBV) and hepatitis C virus (HCV) coinfection is often associated with severe forms of liver disease. However, comprehensive studies are lacking, and scant information is available regarding the virological behavior over time in coinfected patients. This study enrolled 133 untreated HBV/HCV-positive patients (male/female = 102/31; median age 51 years [range: 22-83 years]) who were longitudinally followed up for 1 year with bimonthly evaluation of HBV/HCV viremia levels and liver biochemistry. Thirty of these patients had triple infection with hepatitis Delta virus (HDV), while 103 patients were HDV-negative. In the HDV-negative group, active infection with both HBV and HCV was revealed in 24 cases, inactive infection by both viruses was seen in 15 cases, active HBV/inactive HCV was seen in 15 cases, and inactive HBV/active HCV was seen in 49 cases. However, 32 subjects (31%) presented dynamic virological profiles characterized by fluctuation of HBV and/or HCV viremia levels that at different time points were over or under the cutoff limits.
Consequently, a correct diagnosis could be performed in these subjects only by serially repeating the virological tests 1 year apart. Similarly, 15 of the 30 HDV-positive subjects showed active HBV and/or HCV infection, with fluctuating virological patterns in 8 cases.
In conclusion, this study showed that the virological patterns in HBV/HCV coinfection are widely divergent and have dynamic profiles. A careful longitudinal evaluation of the viremia levels of both viruses is essential for making a correct diagnosis and tailoring the appropriate therapeutic schedule in coinfected patients.
Discussion
HBV/HCV coinfection is associated with severe forms of chronic liver disease that generally have a serious prognosis and high risk of death, mostly because of their propensity to progress toward cirrhosis and to develop hepatocellular carcinoma.[6][7][10-18] In spite of the considerable clinical impact, many aspects concerning this dual infection have been insufficiently explored thus far, and in particular very little is known from both the clinical/virological and therapeutic points of view. In fact, there is no scientific evidence indicating the most appropriate follow-up for coinfected patients, and no guidelines have been established for their treatment. A key step for a better characterization of this category of patients is the behavior of the viruses implied in the coinfection. As all the available information on the virological patterns of the concurrent HBV/HCV infection comes from cross-sectional investigations, we designed a novel study. It combined the examination of the largest series of chronically coinfected individuals with the longitudinal evaluation of their HBV and HCV viremia levels performed at several time points during a 1-year follow-up. This approach allowed us to demonstrate a wide and complex spectrum of virological profiles in HBsAg/anti-HCV-positive individuals. In fact, we observed that approximately one third of the cases may present alternate phases of inhibition and recurrence of the activity of one or both the viruses, as revealed by broad changes over time of the amount of circulating HBV and/or HCV genomes. The exact identification of the viral infections primarily responsible for liver injury can only be reached via the longitudinal evaluation of the virological patterns.
Several studies have suggested an interplay between the two viruses in cases of dual infection with a prevalent negative influence of HCV on HBV activity.[2][25-32] Although the investigation of such intervirus interaction was not the aim of our study, the longitudinal profile of each virus in all the cases with fluctuating patterns appeared to be totally independent of the viremia levels of the other. In this context, one should consider that the classic anti-HBe-positive B hepatitis is often characterized by phases of low levels of HBV replication interspersed with episodes of viral reactivation,[39-42] and the vast majority of our cases were anti-HBe-positive. Similarly, in cases of single infection, HCV may also show alternating phases of active and suppressed replication.[43-45] Consequently, we might speculate that at least in a portion of HBV/HCV-coinfected cases, the behavior of each virus is independent of the contemporary presence of the other. Thus each virus might exert its own pathogenetic role, deriving a synergistic (and accumulating) effect in terms of liver damage that may explain the high grade of disease severity frequently observed in case of coinfection. This hypothesis is in agreement with previous data showing the contemporary presence of the typical histological patterns of each individual infection in cases of HBV and HCV coexistence.[46] It appears to be confirmed by the significant association between abnormal ALT values and double active infection that we revealed in this study. In fact, in cases with active infections showing fluctuating virological profiles, the viremic changes do not appear to correlate with parallel modifications of liver biochemistry. However, as the peaks of viremia may precede the rise in ALT values by several days, we cannot exclude the possibility that flares of the ALT levels were missed in some cases due to the bimonthly schedule of serum sampling used in this study. In addition to the ALT values and the obvious association between HBeAg positivity and active HBV infection, the only other statistical significance was detected in the sex distribution, with males equally distributed in all the different virological categories while female sex appeared to be more frequently associated with the inactive HBV/active HCV status. Again, these results appear to reflect the typical behavior of the single infections and likely depend on the well-known lower frequency of development of chronic hepatitis B in women compared with men.
HBV and HCV viremia longitudinal analyses also provided interesting information when HDV-positive cases were tested. First, we found that half of these patients had evidence of HBV and/or HCV active replication, and such individuals appear more frequently to have cirrhosis compared with those carrying constantly inactive HBV and HCV infections. In this context, although the common belief is that HDV suppresses HBV and tends to also play a dominant role in the case of HCV coinfection,[12][32] it is relevant to recall some studies of the early 1990s that reported that HDV patients with persistence of active HBV replication are associated with the more severe and quickly evolving forms of liver disease.[47][48] Both HBV and HCV showed wide fluctuations of the viremia levels in several HDV patients. Interestingly, serum HDV RNA levels also seemed to change over time in some cases, confirming the importance of longitudinal evaluation of virological patterns in cases of multiple viral hepatitis infections.
In conclusion, this study demonstrates that HBV/HCV coinfection is not a stable condition, but it may present dynamic and possibly evolving profiles. This is also the case in additional HDV infection. Consequently, HBsAg/anti-HCV-positive individuals must be carefully monitored over time to precisely define their virological status. Considering that HBV/HCV coinfection is highly prevalent in many areas of the world, and in view of the severity of the associated liver diseases, it appears particularly urgent to design and perform therapeutic trials for the definition of the best approaches for the treatment of these patients - trials that must take into account the peculiar behavior of viral activity in these subjects. At present, for coinfected patients who cannot wait for the indications of future trials, the therapeutic schedule should be tailored to each individual patient on the basis of his/her own virological profile.
Results
A total of 133 subjects (132 Italians, 1 Albanian) were included in the study (Table 1); 103 of these patients had no evidence of HDV infection, whereas 30 were anti-HDV-positive (Table 2). Of the 103 anti-HDV-negative individuals, 77 were male and 26 were female (age mean ± SD: 54.1 ± 13.1 years). Sixty-eight patients had been exposed, and 35 had no history of exposure to risk factors for parenterally transmitted viruses. Forty patients had received previous interferon treatment (finished in all the cases from 1 to 10 years before enrollment in the study), while 63 had never been treated with any antiviral therapy. Sixty-nine patients had chronic hepatitis, and 34 had cirrhosis (the diagnosis was performed via liver biopsy in 71 cases and via clinical/biochemical/ultrasonographic evaluation in 32 cases). Eleven patients were HBeAg-positive, and 92 were anti-HBe-positive. Finally, among the 73 subjects who were found to be HCV RNA-positive, the viral genotype was available for 56 of them, 39 of whom were infected by genotype 1 and 17 by a non-1 genotype (13 with genotype 2, 4 with genotype 3).
Of the 30 anti-HDV-positive individuals, 25 were male and 5 were female (age mean ± SD: 45.9 ± 11.6 years), and 15 had no known risk factors for parenterally transmitted viruses. Eleven patients had previously been treated with interferon (from 1 to 13 years before this study), while 19 had never received antiviral therapy. Thirteen patients had chronic hepatitis and 17 had cirrhosis (the diagnosis was performed via liver biopsy in 21 cases and via clinical/biochemical/ultrasonographic evaluation in 9 cases). One patient was HBeAg-positive and 29 were anti-HBe-positive. Among the 6 HCV RNA-positive individuals, the viral genotype was available in 4 cases, with evidence of genotype 1 in 2 cases and genotypes 2 and 4 in 1 case each. Statistical analyses showed that HDV positivity was significantly associated with younger age and cirrhosis compared with the counterpart (P = .01 and P = .02, respectively) (Table 2).
We examined a total number of 758 serum samples for HBV DNA and HCV RNA quantifications from the 133 subjects of the study population. The average number of samples per patient examined was 5.7 (range: 3-7). In detail, we analyzed a series of 3 sera in 12 cases, 4 sera in 11 cases, 5 sera in 30 cases, 6 sera in 32 cases, and 7 sera in 48 cases.
Anti-HDV-Negative Patients.
The evaluation of a single time point (e.g., the sole baseline sample from each patient) led to the identification of active infection of both HBV and HCV (as defined by HBV DNA levels >105 copies/mL and HCV RNA levels >600 IU/mL) in 12 cases, inactive infection by both viruses (HBV DNA <105 copies/mL, HCV RNA <600 IU/mL) in 20 cases, active HBV and inactive HCV in 14 cases, and inactive HBV and active HCV in 57 cases. However, when we considered all the serum samples longitudinally collected 1 year apart from each patient, we observed that 32 (31%) of the 103 cases had fluctuation patterns of HBV and/or HCV viremia levels that were above or below the cutoff limits at the various times evaluated (Fig. 1), whereas 71 cases (69%) had virological profiles stabilize over time (Table 3). Consequently, considering that patients presenting with phases of viral reactivation and high viremia levels alternate with phases of apparent viral suppression, and a decreased amount of circulating virions must be classified as carriers of active infection, the virological behavior at the end of the follow-up led us to make diagnoses differing from those made when only a single time point had been evaluated in approximately one third of the patients. In fact, there were 24 cases with HBV/HCV active infections; only 15 were confirmed as having both viruses inactive, 15 had active HBV and inactive HCV, and 49 cases had inactive HBV and active HCV. Detailing the results obtained, HBV DNA values were constantly below 105 copies/mL or were over that limit in 64 and 14 cases, respectively, while a fluctuation pattern was observed in 25 cases. Similarly, HCV RNA levels were persistently below or over 600 IU/mL in 30 and 60 cases, respectively, while a fluctuation profile was revealed in 13 cases. In particular, a fluctuation pattern of the HBV DNA alone was observed in 19 cases, of the HCV RNA alone in 7 cases, and of both of them in 6 cases.
Figure 1. Sequential HBV DNA, HCV RNA, and ALT values detected in three paradigmatic cases showing fluctuating virological profiles of HBV (case 1), HCV (case 2), or both viruses (case 3)
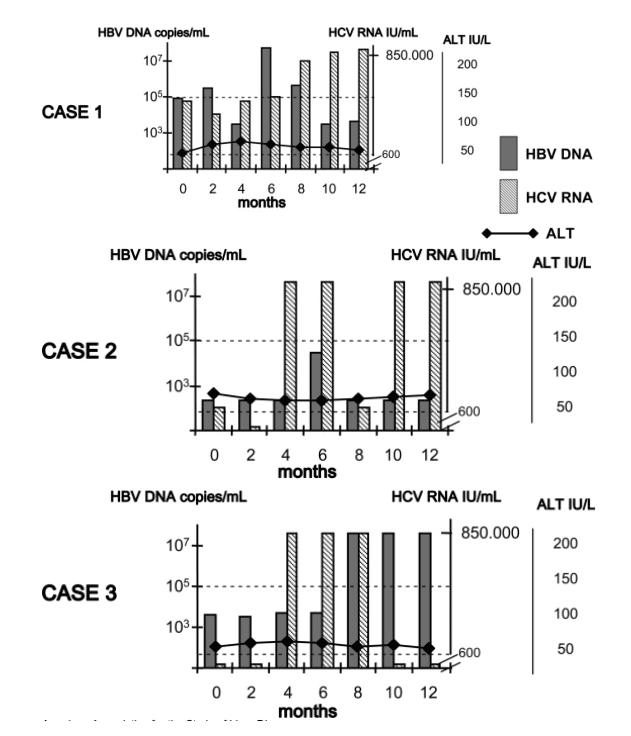
It is known that 105 copies/mL of HBV DNA is an arbitrary threshold for diagnosing the status of active HBV infection,35 so that a cutoff of 3 X 104 copies/mL has recently been proposed as a more reliable limit for distinguishing chronic hepatitis B patients from inactive carriers of the virus.[38] Of note, in the anti-HDV-negative group of our study, 4 of the subjects that we classified as persistently inactive HBV carriers showed HBV DNA values between 3 X 104 and 105 copies/mL (3 cases included in the inactive HBV/active HCV category and 1 case included in the inactive HBV/HCV category). Concerning the HCV RNA levels in the subjects categorized as actively HCV infected (a total of 73 cases), only 2 cases showed viral amounts lower than 100,000 IU/mL at all the time points examined (HBV was active in 1 case and inactive in the other), 11 cases had values persistently below 850,000 IU/mL (HBV was active in 4 cases and inactive in 7 cases), and 60 cases had HCV RNA levels almost constantly higher than 850,000 IU/mL (HBV was active in 20 cases and inactive in 40 cases). The viremia values over time of each individual virus appear to be totally independent of the virological profile of the other. In particular, the reactivation phases of a virus did not parallel with any peculiar modification of the viremia levels of the other.
The ALT values were normal in 21 patients, while 82 cases had abnormal ALT values (at all time points in 59 cases, alternating with normal values in the remaining 23), as detailed in Table 4. ALT flares (defined as described above) were revealed in 10 patients, 2 of whom had active HBV/active HCV, 1 of whom had both viruses inactive, 1 of whom had active HBV/inactive HCV, and 6 of whom had inactive HBV/active HCV. It should be noted that no case had ALT values higher than 7 times the normal levels at any time point. Finally, no episode of ALT flare occurred simultaneously with spikes of either HBV DNA or HCV RNA values.
Besides the obvious association between HBeAg-positive status and active HBV infection (P = .001), statistical significance was detected only for sex (P = .03, V-Cramer = .31, with female sex mostly related to inactive HBV/active HCV status and male sex quite uniformly distributed in the different categories) and for the ALT values that were related with the double-active infection in cases of abnormal values and with the dual inactive infection in cases of persistently normal levels (P < .001, V-Cramer = 0.35) (Table 4).
Anti-HDV-Positive Patients.
Fifteen of the 30 patients included in this group showed persistently inactive HBV and HCV infections, while the other 15 had active HBV (9 cases) or HCV (5 cases) or both (1 case). The comparison of the two subgroups of 15 cases each showed an association between cirrhosis and presence of active HBV and/or HCV, although statistical significance was not reached (P = .06). No other relevant correlation was found. Of these latter 15 cases, 4 had a constantly active profile and 5 had a fluctuating profile of HBV DNA levels, 3 had a constantly active profile and 2 had a fluctuating profile of HCV RNA values, while 1 case presented a fluctuating pattern of both viruses. It should be noted that 2 of the 15 patients categorized as inactive HBV/HCV showed levels of HBV DNA repeatedly comprised between 3 X 104 and 105 copies/mL. Of the 6 patients with active HCV infection, 1 had HCV RNA levels persistently below 100,000 IU/mL and inactive HBV infection, 4 had HCV RNA levels persistently below 850,000 IU/mL and inactive HBV, and 1 had HCV RNA values higher than 850,000 IU/mL with contemporary active HBV infection.
Serially collected serum samples from 21 of the 30 subjects belonging to this group were available to be tested for HDV RNA. The viral RNA was found in all the samples examined from 3 cases (1 with inactive HBV/HCV, 1 with active HBV/inactive HCV, 1 with inactive HBV/active HCV), whereas in 6 cases the HDV RNA was detected in no serum sample (4 with inactive HBV/HCV, 2 with active HBV/inactive HCV). The other 12 cases (6 with inactive HBV/HCV, 4 with active HBV/inactive HCV, 2 with inactive HBV/active HCV) showed alternate positivity for HDV RNA at the different time points examined. Interestingly, this kind of fluctuation pattern of HDV RNA positivity combined with a similar waving profile of the HBV DNA in the 4 actively HBV-infected patients and of the HCV RNA in 1 of the 2 cases with active HCV infection.
|
|
| |
| |
|
|
|