| |
Early monotherapy with pegylated interferon alpha-2b for acute hepatitis C infection: The HEP-NET acute-HCV-II study
|
| |
| |
Hepatology feb 2006
Johannes Wiegand 1, Peter Buggisch 2, Wulf Boecher 3, Stefan Zeuzem 4, Cornelia M. Gelbmann 5, Thomas Berg 6, Wolfgang Kauffmann 7, Birgit Kallinowski 8 , Markus Cornberg 1, Elmar Jaeckel 1, Heiner Wedemeyer 1, Michael P. Manns 1 * , German HEP-NET Acute HCV Study Group
1Department of Gastroenterology, Hepatology, and Endocrinology, Hannover Medical School, Hannover, Germany
2Universitatsklinikum Eppendorf, University of Hamburg, Hamburg, Germany
3I. Medizinische Klinik, University of Mainz, Mainz, Germany
4Medizinische Klinik II, Saarland University, Homburg, Germany
5Innere Medizin I, University of Regensburg, Regensburg, Germany
6Charite, Campus Virchow-Klinikum, Universitatsmedizin Berlin, Berlin, Germany
7Vivantes Klinikum Prenzlauer Berg, Berlin, Germany
8Medizinische Klinik IV, University of Heidelberg, Heidelberg, Germany
Potential conflict of interest: Dr. Zeuzem is a consultant for and has received unrestricted grants from Roche and Schering. Dr. Wiegand has received travel grants from Schering. Drs. Jaeckel and Comberg have received travel grants from Essex Pharma. Dr. Wedemeyer has received research and travel grants from Essex Pharma. Dr. Manns is a consultant for and has received research and travel grants from Schering. Dr. Buggisch is a consultant for, has received research and travel grants from, and has conducted clinical trials for Roche. He has also been a consultant and conducted clinical trials for Schering-Plough.
ABSTRACT
Early treatment of acute hepatitis C with interferon alpha-2b for 24 weeks prevents chronic infection in almost all patients. Because pegylated interferons have replaced conventional interferon in the therapy of chronic hepatitis C, the aim of this study was to analyze the efficacy of an early treatment of acute hepatitis C with peginterferon alfa- 2b. Between February 2001 and February 2004, 89 individuals with acute HCV infection were recruited at 53 different centers in Germany. Patients received 1.5 g/kg peginterferon alfa-2b for 24 weeks; treatment was initiated after a median of 76 days after infection (range 14-150). End-of-treatment response and sustained virological response were defined as undetectable HCV RNA at the end of therapy and after 24 weeks of follow-up, respectively. In the total study population, virological response was 82% at the end of treatment and 71% at the end of follow-up. Of 89 individuals, 65 (73%) were adherent to therapy, receiving 80% of the interferon dosage within 80% of the scheduled treatment duration. End-of-treatment and sustained virological response rates in this subpopulation were 94% and 89%, respectively. A maximum alanine aminotransferase level of more than 500 U/L prior to therapy was the only factor associated with successful treatment. In conclusion, in acute HCV infection, early treatment with peginterferon 2b leads to high virological response rates in individuals who are adherent to treatment. The high number of dropouts underlines the importance of thorough patient selection and close monitoring during therapy. Thus, future studies should identify factors predicting spontaneous viral clearance to avoid unnecessary therapy.
Article Text
Management of acute hepatitis C has become a controversial topic in recent years.[1][2] Acute infection with hepatitis C virus (HCV) leads to chronicity in 54% to 84% of patients.[3] Chronic hepatitis C can be cured with pegylated interferon alfa plus ribavirin in 54% to 63% of cases; however, this regimen is not successful in approximately half of patients infected with HCV genotype 1.[4-6] Prevention of a chronic course of hepatitis C seems to be a desirable therapeutic goal considering the high risk of chronicity, the substantial number of nonresponders in chronic HCV infection, and the potential risk to develop cirrhosis or hepatocellular carcinoma. In addition, in special patient groups such as health care personnel, the infection might have important legal and social consequences.
Detection of acute hepatitis C can be difficult, because infection is not often accompanied by relevant symptoms.[7] Furthermore, after the introduction of screening of blood products for HCV RNA, the main source of infection has shifted from transfusion to intravenous drug use.[8-10] For many reasons, drug addicts may not be eligible for clinical trials, which further limits the possibility to conduct prospective studies.
It still remains controversial if therapy should be initiated as early as possible or if the start of therapy can be delayed, because no prospectively controlled trials have been reported.[11-19] Both approaches have yielded sustained response rates of 80% to 98%,[11][12] but final recommendations about the optimal interferon dosage and treatment duration cannot be made yet. However, it has been shown that use of ribavirin combination is not needed in the treatment of acute HCV infection.[14] In Germany, the awareness of acute hepatitis C infection increased substantially after a pilot trial with an interferon alfa-2b monotherapy for 24 weeks showed that HCV could be eradicated in 98% of patients independently of HCV genotype.[11] Because the standard therapy of chronic hepatitis C has moved from conventional interferon alfa to pegylated interferon alfa the aim of the present study was to evaluate the efficacy of early treatment with peginterferon alfa-2b in patients with acute HCV infection. The study was conducted with the support of the German Competence Network for Viral Hepatitis (HEP-NET), an organization of hepatology experts that was founded to establish nationwide standards for the treatment of viral hepatitis, transfer knowledge between different institutions of the health care system, and coordinate scientific studies.[20]
Patients and Methods
The study was designed as a nationwide open, uncontrolled multicenter trial. HEP-NET served as coordinator and central information unit and distributed information brochures to hospitals, outpatient clinics, private practices, and patient-advocacy groups. Patients (18-65 years of age) were eligible if they were HCV RNA-positive (as assessed via polymerase chain reaction) and had elevated serum alanine aminotransferase (ALT) levels. Acute HCV infection was considered to be present if at least 1 of the following criteria was fulfilled: (1) known or suspected exposure to HCV within the preceding 4 months, (2) documented seroconversion (anti-HCV-negative to anti-HCV-positive), or (3) ALT 20 times the upper limit of normal. If ALT was elevated below 20 times the upper limit of normal, patients were approved by the central study unit only after extensive discussion with the investigator if the additional criteria mentioned in the manuscript were fulfilled, including a very likely event of HCV transmission within the preceding 4 months in combination with HCV seroconversion. In addition, any history of previous liver disease and any previous ALT elevation were ruled out. These criteria were strictly used throughout the entire study period. Thus, to the best of our knowledge, cases with potential chronic HCV infection were ruled out.
Exclusion criteria were: coinfection with hepatitis B virus or HIV; autoimmune diseases, especially autoimmune liver disease; leukocytopenia (<3,000 leukocytes/L); thrombocytopenia (<100,000 thombocytes/L), decompensated cirrhosis (Child-Pugh class B or C); decompensated renal (creatinin >2.0 mg/dL) or thyroid (TSH out of normal range) disease; psychiatric conditions such as severe depression; a history of seizures; poorly controlled diabetes mellitus or ophtalmological disease; pregnancy; or a history of organ transplantation or malignancy. Patients with ongoing abuse of alcohol or intravenous drugs were not eligible. In individual cases, if intravenous drug abuse was considered a single event or if the patient was participating in a withdrawal cure, inclusion in the study was possible.
To ensure uniform standards throughout the different sites, enrollment and treatment criteria were strictly controlled by the HEP-NET Study House and the investigators of the Hannover Medical School. Prior to enrollment of a patient, each local investigator had to contact the central study unit in Hannover, where the HCV infection criteria and the inclusion/exclusion criteria were checked. Thus, each patient had to be approved by the central study unit before inclusion. During the study period, the peripheral centers were frequently contacted to collect the data of each visit into the central database as early as possible. If patients were lost to follow-up, the investigators tried repeatedly to recontact the individuals via telephone or mail.
Patients received 1.5 g/kg/wk peginterferon alfa-2b subcutaneously for 24 weeks. Biochemical and hematological testings were performed locally. Viral genotypes were determined either locally or centrally in Hannover (INNO-LiPa HCV II Kit; Innogenetics, Heiden, Germany). HCV RNA was quantified before, during, and after treatment. The primary end point was defined as sustained virological response (HCV RNA-negative [as assessed via polymerase chain reaction] 24 weeks after the end of therapy) (Cobas Amplicor 3.0; Roche Diagnostics, Mannheim, Germany [detection limit 600 copies HCV RNA/mL]). Secondary end points were the absence of detectable levels of HCV RNA at the end of therapy and normalization of ALT levels.
Safety of the study drug was assessed via analysis of documented adverse events and serious adverse events. Adverse events were classified as mild (no dose adjustment necessary), moderate (dose adjustment possible) or severe (dose adjustment mandatory).
All patients provided written informed consent before enrolling in the study. The study was approved by the appropriate local ethics committees and the ethics committee of Hannover Medical School and was performed according to the Declaration of Helsinki of 1996.
Results
Baseline Characteristics of the Study Population.
Between February 2001 and February 2004, 89 patients with acute HCV infection were recruited at 53 different centers (university hospitals [n = 18], state hospitals [n = 26], private gastroenterologists and primary care physicians [n = 9]). Treatment was initiated in all 89 patients, and 70 patients (79%) were adherent to therapy. Adherence to therapy was defined as application of at least 80% of the peginterferon alfa-2b dosage and therapy for 80% of the scheduled treatment period. Complete follow-up data were available for 65 of the adherent individuals (93%). In 19 patients (21%) the criteria of therapy adherence were not fulfilled, because the patients prematurely stopped therapy due to side effects, were lost to follow-up during treatment, or were not treated according to protocol (Fig. 1). The reasons for protocol violations were reduced interferon dosage (1.0 g/kg) in two cases, interferon injection every other week in one patient, and noncompliance in one patient.
Suspected sources of infection were intravenous drug abuse and sexual transmission (each 22% of cases), medical procedures (19%; most common: surgery (11/17 individuals)), needle stick injuries (11%) and other potential transmission modes (acupuncture, blood brotherhood, bite injury, shared razor, tattooing, hair clippers; 8%). In 17% of patients no risk factor for infection could be determined.
Jaundice was observed in 62% of cases, 40% of patients reported fatigue, and 19% were asymptomatic. Additional demographic and clinical data of the study population are summarized in Table 1.
Efficacy of Antiviral Therapy.
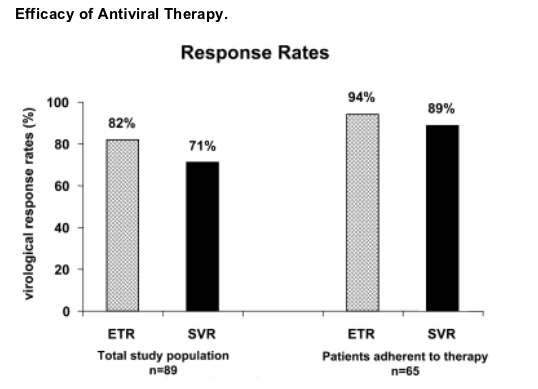
Serum levels of ALT were within the normal limits in the total study population in 73% of patients at the end of therapy and in 86% after 24 weeks of follow-up. In the subgroup of individuals adherent to therapy, ALT levels normalized in 75% and 91% of cases, respectively. Except for two patients with virological relapse, persistent elevations of ALT levels were only mild, not exceeding more than twice the upper limit of normal.
At the end of treatment, HCV RNA was undetectable in 82% of patients of the total study cohort and in 94% of individuals adherent to therapy (Fig. 2). After 24 weeks of follow-up, a sustained virological response was observed in 71% and 89% of cases, respectively. In addition to the 58 successfully treated patients in the therapy adherent subgroup, five other individuals became sustained virological responders who were either treated with pegylated interferon only every other week (n = 1) or for a shorter period of 8-16 weeks due to side effects (n = 4).
In the subgroup of patients adherent to therapy, we observed one patient with virological nonresponse, two patients with virological breakthrough, and 4 relapse cases (Table 2). These patients did not differ significantly from the sustained virological responders in age (>40 yr vs. <40 yr, P = 1.0; >30 yr vs. <30 yr, P = .4), sex (P = .2), HCV genotype (HCV genotype 1 and 4 vs. HCV genotype 2 and 3, P = 1.0), HCV RNA viral load (HCV RNA >850,000 IU/mL vs. HCV RNA <850,000 IU/mL, P = 1.0), or bilirubin levels at baseline (P = .2). Only maximum ALT levels of more than 500 U/L were significantly more frequent in patients with sustained treatment response (P = .025). Stepwise multivariate regression analysis confirmed the association of sustained virological response rates with ALT levels (P = .039). ALT levels in patients with HCV genotype 2 and 3 were not significantly higher than in patients with HCV genotype 1 and 4 (P = .15).
Compliance.
The difference of virological response rates between the total study population and the individuals adherent to therapy was mainly due to 13 cases who were lost to follow-up. The lost to follow-up rates did not differ between the different institutions (university hospitals, general hospitals, and private practices) that guided the patients during therapy (P = 0.3). Six of the 13 patients had previously used intravenous drugs. However, previous intravenous drug abuse was not associated with being lost to follow-up (P = 1.0). Patient sex did not correlate with therapy adherence (P = 0.8) or lost to follow-up rates (P = 1.0).
Safety.
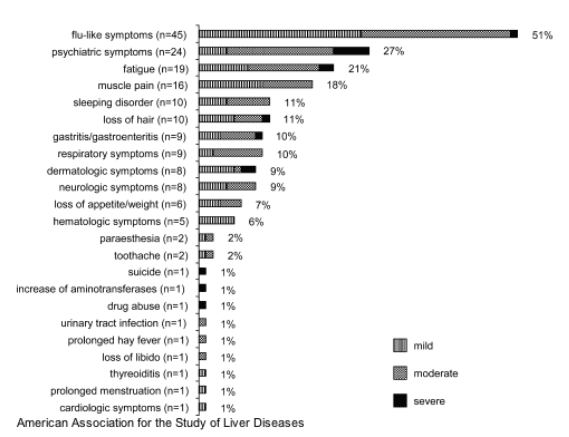
The overall tolerance of therapy was comparable to previous studies with peginterferon alfa-2b.[4][21] The spectrum of side effects and their reported intensity is shown in Fig. 3. Psychiatric symptoms were the most frequent side effects after flu-like symptoms and were responsible for discontinuing interferon treatment in 6 of 11 patients with premature stoppage of therapy. Patients who experienced psychiatric side effects tended to stop therapy prematurely more often than those who did not experience these effects (P = .06).
The most relevant serious adverse event was the suicide of one patient. This patient received psychiatric evaluation before therapy because of a suicide attempt 2 years earlier, after his wife had died. No obvious contraindications against interferon treatment were noticed. During therapy, the patient was seen weekly by the same physician without any signs of psychiatric alteration. However, the patient committed suicide during week 22 of therapy.
Two other serious adverse events occurred: 1 patient was in a deadly car accident, and another had a severe increase in ALT levels (2,369 U/L) during week 4 of therapy, requiring hospitalization and premature termination of therapy. The ALT increase was most likely due to patient noncompliance, because the patient interrupted therapy after week 2 or 3.
A notable side effect was the relapse of intravenous drug use at week 22 of therapy in one individual. Fortunately, drug consumption was successfully stopped after supportive psychological therapy. Reinfection with HCV did not occur.
Discussion
The aim of the present study was to evaluate the efficacy of an early therapy with peginterferon alfa-2b in patients with acute HCV infection. Compared with our previous nationwide German trial,[11] the number of patients and the number of recruiting centers was doubled within a similar study period. To our knowledge, this study cohort represents the largest ever described for a treatment trial of acute hepatitis C. The high recruitment rate is a result of the Jaeckel study and the support of the German Competence Network for Viral Hepatitis (HEP-NET), which markedly increased awareness of the disease. The more effective coordination reduced the median time from infection to the start of therapy from 89 to 76 days. However, the increase of the study population was associated with more frequent treatment failures, protocol violations, and patients lost to follow-up. Thus, the overall sustained virological response rates were lower than expected after the Jaeckel trial. Only in the patients adherent to therapy were the results almost in the same range as in the previous trial using conventional interferon alfa-2b. The only factor correlating with successful therapy was a high maximum ALT level before treatment. Whether the slightly higher sustained response rates in the Jaeckel trial using conventional interferon (98% vs. 89%) can be explained by the 4 weeks of daily induction dosing treatment applied in the first trial[11] remains to be investigated.
The high number of patients lost to follow-up highlights again that individuals with acute hepatitis C are difficult to treat. A recent report from Switzerland on intravenous drug users with acute hepatitis C demonstrated low adherence to therapy, especially in females and individuals with ongoing drug abuse.[18] Previous drug abuse and female sex did not significantly affect adherence in the present study. However, this study demonstrates that close monitoring both during therapy and follow-up is required - particularly for patients with acute hepatitis C - because our patients' selection criteria did not reliably prevent weak adherence, even though individuals with ongoing intravenous drug abuse were excluded.
The overall safety and tolerance of the medication did not differ from previous studies with peginterferon alfa-2b.[4][21] We observed a trend for psychiatric symptoms to be associated with premature termination of therapy. In addition, three serious adverse events occurred, including one suicide and one patient with a relapse of intravenous drug abuse. These observations clearly highlight the need of thorough patient selection and extensive information about potential side effects and about treatment alternatives, especially because other long-term side effects after interferon therapy seem to be possible in single cases.[22] Thus, therapy of acute HCV should be performed only by physicians experienced in handling the patients and the medication with its potential complications.
It should be noted that 10% to50% of patients with acute hepatitis C may clear the infection spontaneously.[2][7][23] Treating these individuals as early as possible may unneccessarily place them at risk for adverse events and may cause substantial therapeutic costs. Thus, overtreatment must be balanced against the risk of chronification. Unfortunately, there are no markers that reliably predict the natural course of the disease. ALT levels failed to predict the spontaneous outcome of acute HCV infection[24]; however, low ALT levels were associated with treatment failure in the present study and may therefore be considered before the initiation of therapy. An alternative to early treatment of acute HCV infection may be a delayed therapy to treat only those patients who do not clear the virus spontaneously. Gerlach et al.[12] treated only individuals who were still viremic 12 weeks after the onset of symptoms corresponding to 4-5 months of infection. The response rate in symptomatic patients was 91% in this study cohort. In other smaller patient cohorts, a delayed monotherapy either with conventional interferon or with pegylated interferon alfa lead to sustained virological response rates of 80% to 100%.[14][16][19][25] However, therapy should not be delayed for too long,[25] because response rates dropped to 40% in a Japanese study if treatment was initiated after 1 year.[13] In European patients, the risk for treatment failure may also be dependent on the time between initial presentation and treatment initiation.[15] Thus, there is increasing evidence that delayed therapy may be effective for many patients; however, randomized trials comparing immediate versus delayed therapy have not been published yet.
Future trials on acute HCV should also try to identify predictive factors for the natural course of the infection. The relevance of clinical symptoms and racial differences is unknown.[12-14][19][24] HCV genotyping should be performed in every patient to further individualize the indication before therapy. HCV genotype 1 and 4 are difficult to treat in chronic HCV but can be effectively cured in acute infection.[11][14][16] On the other hand, there are data indicating a higher rate of spontaneous viral clearance in acute HCV genotype 3 infection.[26] Considering the likelihood of successful therapy with pegylated interferon alfa plus ribavirin in individuals with genotype 3, a more reserved therapeutical approach may be reasonable in these patients. In certain settings, repeated HCV RNA testing might help to identify individuals who are able to clear the virus spontaneously,[24] although frequent HCV RNA testing may be difficult to apply in a routine clinical setting. Another criterion for the initiation of therapy is the social background of the patient. For social and legal purposes, it may be dangerous to delay therapy in infected health care workers - especially if there is no evidence of a late relapse after early interferon therapy, even years later.[22]
There is no evidence to combine ribavirin with interferon alfa in acute hepatitis C. The present study, in addition to several previous trials, report high rates of viral clearance using conventional interferon treatment or pegylated interferon alfa without ribavirin.[11][13][17] Moreover, the only randomized trial thus far comparing monotherapy and combination therapy in acute hepatitis C did not yield any significant difference in response rates between both approaches.[14] These data are supported by the recent study of Santantonio et al.,[19] who also used peginterferon alfa-2b, although the number of patients was limited and consisted mainly of individuals infected with HCV genotype 2 and 3.
In conclusion, the present study shows high rates of viral clearance in early therapy with peginterferon alfa-2b in patients with acute hepatitis C who are adherent to therapy. However, it also highlights limitations and risks of this therapeutical concept in routine clinical practice. Thus, future studies should aim to identify factors predicting spontaneous viral clearance to avoid unnecessary therapy.
The German HEP-NET Acute HCV Study Group
|
|
| |
| |
|
|
|