| |
Hepatic steatosis in HIV- HCV coinfected patients: analysis of risk factors
|
| |
| |
.....To our knowledge, this is the largest study of steatosis in HIV infected patients with chronic hepatitis C. Steatosis was present in 241 (61%) of cases, a prevalence similar to that observed in HCV monoinfected patients (30- 70%)....we found a correlation between steatosis and both antiretroviral therapy in general and NRTI- based treatment (but not PI- based treatment), but only in univariate analysis (see discussion below)...
AIDS: Volume 20(4) 28 February 2006 p 525- 531
Bani- Sadr, Firouzea; Carrat, Fabricea; Bedossa, Pierreb; Piroth, Lionelc; Cacoub, Patriced; Perronne, Christiane; Degott, Claudeb; Pol, Stanislasf; ANRS HC02 - Ribavic Study team
From the aGroupe Hospitalier Universitaire Est, Universite Paris 6, INSERM V707, Paris, France
bGroupe Hospitalier Universitaire Nord, Universite Paris 7, Paris, France
cCentre Hospitalier Universitaire, Dijon, France
dGroupe Hospitalier Universitaire Est, Universite Paris 6, Paris, France
eCentre Hospitalier Universitaire Raymond Poincare, Universite de Versailles, Garches, France
fGroupe Hospitalier Universitaire Ouest, Universite Paris 5, Universite Paris, France.
Hepatic steatosis, i.e., the accumulation of lipids (mainly triglycerides) in the hepatocyte cytoplasm, is a frequent histological finding during chronic hepatitis C virus (HCV) infection [1- 3]. The prevalence in HCV monoinfected patients is between 30 and 70% [1- 4]. Steatosis is associated with host factors (overweight, diabetes, hyperlipidaemia and excess alcohol intake), viral factors such as HCV genotype 3 and fibrosis [2,4- 6]. In HIV monoinfected patients, hepatic steatosis is a recognized complication of nucleoside reverse transcriptase inhibitor (NRTI) and protease inhibitor (PI) therapy [7- 9]. Consequently, there may be significant interplay among factors influencing steatosis in HIV- HCV coinfected patients. As steatosis accelerates fibrosis, we examined the prevalence and severity of steatosis, and the possible interactions among steatosis, host factors, viral factors, and HIV treatment in a large population of HIV- HCV coinfected patients.
Abstract
Objective: To evaluate the prevalence and severity of steatosis and possible interactions between steatosis, host factors, viral factors, and treatment for HIV infection in HIV- hepatitis C virus (HCV) coinfected patients.
Methods: Steatosis was assessed among 395 HIV- HCV coinfected patients who were enrolled in the ANRS trial HC02 Ribavic and for whom histological data were available. Steatosis was graded as follows: 0 (none); 1 (< 30% hepatocytes containing fat); 2 (30- 70%); 3 (> 70%).
Results:
Steatosis was present in 241 patients (61%), of whom 149 (38%) had grade 1, 64 (16%) grade 2 and 28 (7%) grade 3.
In multivariate analysis, the following five independent risk factors were associated with steatosis:
- - HCV genotype 3 [odds ratio (OR), 3.02; 95% confidence interval (CI), 1.91- 4.79; P < 0.0001],
- - the mean METAVIR fibrosis score (OR, 1.43; 95% CI, 1.11- 1.84; P = 0.0053), the body mass index (BMI; OR, 1.13; 95% CI, 1.05- 1.21; P = 0.0013), HCV viral load (OR. 1.65; 95% CI, 1.22- 2.23; P = 0.0012) and
- - ferritin (OR, 1.13; 95% CI, 1.06- 1.21; P < 0.0003).
As HCV genotype 3 was a risk factor for steatosis, further exploratory analyses were stratified according to the HCV genotype (1 and 3). Factors independently associated with steatosis were BMI and HCV viral load in patients with HCV genotype 3 infection and the mean METAVIR fibrosis score, the BMI and ferritin in patients with HCV genotype 1 infection.
Conclusion: Steatosis is particularly frequent in HIV- HCV coinfected patients, who appear to have the same risk factors for steatosis as HCV monoinfected patients. None of the characteristics of HIV infection, including antiretroviral therapy, was independently associated with steatosis.
Methods
This study involved 395 HIV- HCV coinfected patients who were enrolled in the ANRS trial HC02 Ribavic and for whom histological data were available. The results of this trial have been reported in detail elsewhere [10]. Briefly, 412 HIV- HCV coinfected patients who had never received interferon or ribavirin were randomly assigned to receive either weekly subcutaneous injections of 1.5 mg/kg peginterferon alfa- 2b (ViraferonPeg , Schering- Plough, Kenilworth, New Jersey, USA) plus daily ribavirin (Rebetol , Schering- Plough) (800 mg), or thrice- weekly subcutaneous injections of 3 MU of interferon alfa- 2b plus daily ribavirin (Rebetol , Schering- Plough) (800 mg) for 48 weeks. Patients were eligible for the trial if they had detectable serum HCV RNA, interpretable results of liver biopsy performed within the previous 18 months and showing at least mild activity or fibrosis, a CD4 cell count > 200/mm, stable HIV RNA load (< 1 log10 variation in the previous 3 months) at randomization, and stable or no antiretroviral treatment during the previous 3 months. The main ineligibility criteria were active narcotic consumption and/or self- reported daily alcohol intake exceeding 40 g (women) or 50 g (men) within 3 months before entry to the study; decompensated cirrhosis; and positive HBs antigenaemia.
Biochemical and haematological tests were done in local laboratories. HCV RNA tests, viral genotyping and pathological evaluation of biopsy specimens were performed in central laboratories.
HCV RNA was detected with a PCR assay (Amplicor 2.0 HCV Monitor; Roche Diagnostics Systems, Basel, Switzerland) with a detection limit of 50 IU (100 copies)/ml. HCV RNA levels were measured with a branched- chain DNA assay (bDNA3.0; Bayer Diagnostics, Tarrytown, New York, USA) with a detection limit of 615 IU (3200 copies)/ml. HCV genotyping was performed by sequence analysis of the 5�� untranslated region.
Two experienced pathologists blinded to the clinical and laboratory findings assessed all biopsies on standard stained sections. The mean delay between liver biopsy and entry to the study was 7.15 ± 7.08 months. Steatosis was graded as follows: 0 (none); 1 (< 30% hepatocytes containing fat); 2 (30- 70%); 3 (> 70%). Hepatic necroinflammation and fibrosis were graded with the METAVIR scoring system (scores ranging from 0 to 3 for necroinflammatory activity, and 0 to 4 for fibrosis) [11].
The following data were collected at enrolment: age, sex, birth place (France, North Africa, Sub- Saharan Africa, Asia, other), body mass index (BMI), antiretroviral drugs, duration of HIV and HCV infection (defined as the date of first transfusion or the date of first intravenous drug use), the METAVIR scores, biochemical test results, liver enzyme activities, transferrin saturation, serum ferritin, fasting blood glucose, CD4 lymphocyte counts, plasma HIV RNA load and plasma HCV RNA load. Diabetes and lipodystrophy were collected in the history of the patients reported by the physician at inclusion.
The Cochran- Armitage test for trends was used to identify links between steatosis and qualitative variables, and Spearman's rank correlation test was used to test for associations with quantitative variables. Other comparisons used the Chi- squared test for qualitative variables and the Mann- Whitney test for quantitative variables. A proportional- odds cumulative logistic regression model was constructed with steatosis as the response variable, in three ordered levels. Explanatory variables with P < 0.20 in univariate analysis were included in the multivariate models and selected by using a stepwise procedure. All statistical tests were two- sided, with a type I error of 5%.
Results
The mean size of liver biopsy was 18.1 mm (± 8.1). Steatosis was present in 241 (61%) of the 395 HIV- HCV coinfected patients, of whom 149 (38%) had grade 1, 64 (16%) grade 2, and 28 (7%) grade 3. In view of the relatively small number of patients with grade 2 and 3 steatosis, these patients' data were pooled for statistical analysis.
Most patients were male (73.4%), white (75.7%) and the mean age was 39.7 (± 5.4) years. Intravenous drug use was the risk factor for HCV transmission in 80% of cases. The mean BMI was 22.3 ± 2.9 kg/m2. Five patients were obese (BMI > 30 kg/m2). The distribution of HCV genotypes was as follows: genotype 1, 190 cases (48.1%); genotype 2, 12 cases (3%); genotype 3, 138 cases (34.9%); genotype 4, 51 cases (12.9%). In total, 297 patients (75%) were receiving antiretroviral therapy at the time of liver biopsy.
Factors associated with grade of steatosis TOP
Demographic and laboratory data are summarized in Table 1 and Table 2 according to grade of steatosis. In univariate analysis, patients with grade 1 or grade 2/3 steatosis were significantly older (39.8 ± 5.4 and 40.6 ± 5.7 years, respectively) than patients with no steatosis (39.0 ± 5.3 years; P = 0.03). A highly significant positive correlation (P < 0.0001) was found between the BMI and the grade of steatosis. The mean BMI (± SD) of patients with no steatosis was 21 ± 2.8 kg/m2, compared to 22.3 ± 2.8 kg/m2 and 23.5 ± 3.1 kg/m2, respectively, in patients with grade 1 and grade 2/3 steatosis. Only 12 patients had diabetes, and the possible impact of this factor on steatosis could not therefore be evaluated. Four diabetic patients had grade 1 steatosis and five had grade 2/3 steatosis. No correlation was found between steatosis and sex or ethnicity.
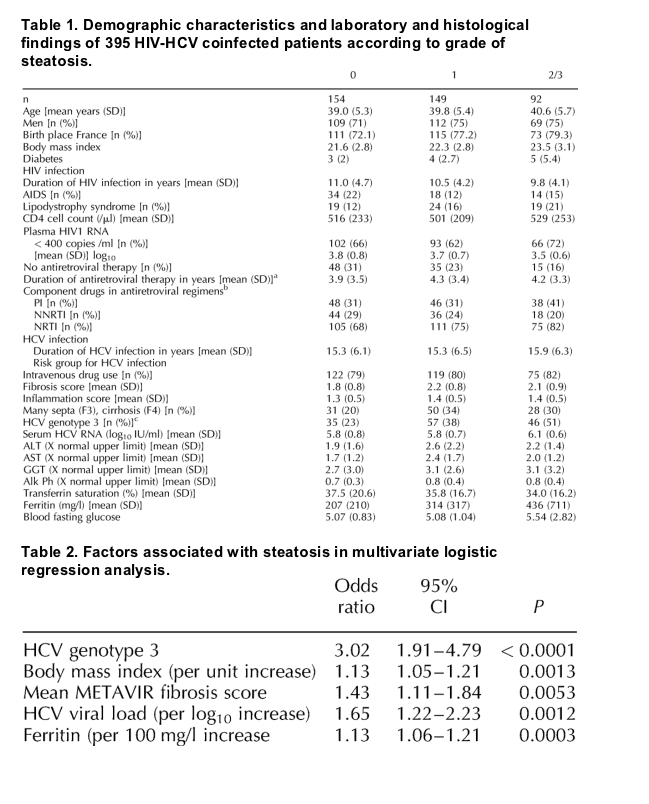
The mean duration of HIV infection was significantly shorter (9.8 ± 4.1 years) among patients with grade 2/3 steatosis than in patients with grade 1 steatosis (10.5 ± 4.2 years) and patients without steatosis (11.0 ± 4.7 years) (P = 0.007). In total, 297 patients (75%) were receiving antiretroviral therapy before or at the time of liver biopsy. Patients with steatosis grade 2/3 were more likely to be receiving antiretroviral therapy than were patients with grade 1 steatosis (84% versus 77%) and patients without steatosis (69%) (P = 0.008). NRTI- based treatment was more frequent among patients with steatosis grade 2/3 than among patients with grade 1 or grade 0 steatosis (82%, 75% and 68%, respectively; P = 0.02). No particular NRTI was associated with steatosis. No correlation was observed between steatosis and AIDS status, the CD4 cell count, HIV viral load, or PI or non- nucleoside reverse transcriptase inhibitor (NNRTI)- containing regimens. Lipodystrophy was present in 15.7% overall, i.e., 21% in patients with grade 2/3 steatosis, 15% in patients with grade 1 steatosis, and 11% in patients with no steatosis (P = 0.08).
Steatosis was more prevalent in patients with genotype 3 infection than in patients infected by other genotypes (genotype 3, 74.6%; genotype 1, 51.6%, genotype 2, 41.6%; genotype 4, 64.7%) and was also significantly more severe in patients with genotype 3 infection (P < 0.001). Higher HCV viral load was also correlated with the severity of steatosis (r = 0.14; P < 0.006).
The severity of steatosis was significantly associated with the severity of fibrosis (r, 0.14; P = 0.004): the mean METAVIR fibrosis score was 2.1 ± 0.9, 2.2 ± 0.8 and 1.8 ± 0.8, in patients with grade 2/3 steatosis, patients with grade 1 steatosis, and patients without steatosis, respectively. Severe liver fibrosis (METAVIR score F3 or F4) was also more frequent in patients with grade 2/3 steatosis and patients with grade 1 steatosis (30% and 34%, respectively) than in patients without steatosis (20%; P = 0.04). The grade of steatosis correlated with serum levels of alanine aminotransferase (r, 0.15; P = 0.0024), aspartate aminotransferase (r, 0.18; P = 0.0002) and ferritin (r, 0.2; P = 0.0001). In particular, patients with steatosis grade 2/3 had higher ferritin levels (436 ± 711 mg/l) than patients with grade 1 steatosis (314 ± 317 mg/l) and patients without steatosis (207 ± 210 mg/l) while transferrin saturation was not associated with steatosis. No correlation was observed between steatosis and the duration of HCV infection, the risk group for HCV infection, the METAVIR necroinflammation score, the serum level of gamma glutamyl transferase, alkaline phosphatase or the fasting blood glucose.
Multivariate analysis identified five independent risk factors associated with steatosis, namely HCV genotype 3 (P < 0.0001), the mean METAVIR fibrosis score (P = 0.0053), the BMI (P = 0.0013), HCV viral load (P = 0.0012) and ferritin (P < 0.0003). As HCV genotype 3 was a risk factor for steatosis, further exploratory analyses were stratified according to the HCV genotype (1 and 3). In patients with HCV genotype 3 infection, BMI [odds ratio (OR), 1.17; 95% confidence interval (CI), 1.03- 1.32; P = 0.015] and HCV viral load (OR, 2.81; 95% CI, 1.73- 4.57; P < 0.0001; Fig. 1) were independently associated with steatosis. In patients with HCV genotype 1 infection, the mean METAVIR fibrosis score (OR, 1.92; 95% CI, 1.31- 2.81; P = 0.0009), BMI (OR, 1.15; 95% CI, 1.03- 1.29; P = 0.015) and ferritin (OR, 1.15; 95% CI, 1.06- 1.26; P = 0.0011) were independently associated with steatosis.
Discussion
To our knowledge, this is the largest study of steatosis in HIV infected patients with chronic hepatitis C. Steatosis was present in 241 (61%) of cases, a prevalence similar to that observed in HCV monoinfected patients (30- 70%) [2,4,12,13].
In multivariate analysis, steatosis was associated with BMI, HCV genotype 3, HCV viral load, METAVIR fibrosis score and ferritin levels. The correlation between BMI and steatosis was not dependent on the HCV genotype in our dually infected patients, in agreement with another study in HIV- HCV coinfected patients [14], whereas this association is usually found only with genotype 1 in HCV monoinfected patients [2,5,15]. BMI also correlated with the duration of HIV infection (r, - 0.18; P = 0.0004), explaining the counter intuitive and artefactual relationship between the duration of HIV infection and steatosis in univariate analysis. Steatosis usually occurs in patients with a BMI > 25 kg/m2 and can be improved by weight loss [2,3,16]. In our study, the mean BMI of patients with grade 2 or 3 steatosis was higher than that of patients with grade 1 steatosis and patients without steatosis, but remained below the World Health Organization thresholds for overweight and obesity (≥ 25 and > 30 kg/m2, respectively). In HCV monoinfected patients, visceral obesity rather than total fat mass seems to play an important role in the development of steatosis [2]. We did not measure visceral fat in our patients, but it is noteworthy that most were receiving antiretroviral therapy, which can cause lipodystrophy [17]. However, lipodystrophy did not correlate with steatosis, but this may due to the low prevalence of lipodystrophy in our population.
Hepatic steatosis is a well established complication of some NRTI and PI treatment [7- 9]. However, we found a correlation between steatosis and both antiretroviral therapy in general and NRTI- based treatment (but not PI- based treatment), but only in univariate analysis. In multivariate analysis, due to adjustment on the fibrosis score, antiretroviral therapy was not independently associated with steatosis (P = 0.11). The proportion of METAVIR scores F3 or F4 was 17% in patients not receiving antiretroviral therapy versus 31% in those receiving the antiretroviral therapy before or at the time of liver biopsy (P = 0.009). There are two not- mutually exclusive explanations for these observations: the reasons underlying antiretroviral therapy are also linked with fibrosis and steatosis; and the antiretroviral therapy is involved in a causal manner to a higher degree of fibrosis and steatosis. Although our cross- sectional design does not formally permit us to choose between these two explanations, we believe that the first explanation is more likely as the duration of antiretroviral therapy in treated patients (n = 297) was not associated with steatosis or fibrosis (P = 0.16 and P = 0.27, respectively), excluding a dose- response relationship and arguing against causality. Finally, our results are in line with those of others studies which failed to demonstrate a link between antiretroviral therapy and steatosis [14,18]. In contrast, Sulkowski et al. found that steatosis was associated with stavudine use [19]. Although 42% of patients of our study were treated with stavudine during a mean time of 5.5 ± 2.7 years, no particular NRTI was associated with steatosis.
As in HCV monoinfected patients, the prevalence of steatosis differed significantly according to the HCV genotype, i.e., patients with genotype 3 infection had a higher prevalence (74.6%) than patients infected by genotype 1 (41.6%). HCV viral load was also associated with the severity of steatosis. In HCV monoinfected patients, HCV viral load has only been linked to steatosis in patients infected by genotype 3 [2,6]. Our finding could be the result of the higher HCV viral loads observed in HIV- HCV coinfected patients and reinforce the link between the production of viral proteins, including the HCV capsid proteins and steatosis [20,21]. However, our multivariate analysis stratified according to the HCV genotype (1 and 3) also showed that steatosis correlated with HCV viral load in genotype 3 infected patients (OR, 2.85; 95% CI, 1.79- 4.54; P < 0.0001) but not in genotype 1 infected patients. In HIV- seronegative patients infected by HCV genotype 3, steatosis frequently improves during successful anti- HCV therapy, suggesting a possible direct pro- steatotic role of this genotype [22]. We have previously shown, on the same patients, that steatosis significantly improves in patients infected by HCV genotype 3 who have a sustained virologic response to anti- HCV therapy (- 13%, P < 0.001), but not in patients infected by HCV genotype 1 [10].
In HCV monoinfected patients, the onset and progression of steatosis are strong independent predictors of both the severity and the progression of fibrosis [2- 5,23- 25]. In our study, the severity of steatosis was related to a higher hepatic fibrosis score, independently of the HCV genotype and of factors (e.g., age and CD4 cell count) that can influence the rate of HCV disease progression in HIV- HCV coinfected patients [14,26]. However, as in HCV monoinfected patients, in multivariate analysis stratified by genotype 1 and 3, steatosis was associated with fibrosis in genotype 1 infected patients (OR, 1.83; 95% CI, 1.29- 2.60; P = 0.0007) but not in genotype 3 infected patients [4,6,15].
Increased ferritin levels in the presence of normal transferrin saturation have been reported in non- infected HIV patients with steatosis but usually did not reflect iron overload [27- 29]. However, recent reports found that hyperferritinemia is an independent risk factor for faster liver progression in HCV monoinfected patients [25,28]. We showed too a correlation between steatosis and increased ferritin levels (OR, 1.13; 95% CI, 1.06- 1.21; P < 0.0003) but in multivariate analysis stratified by genotype 1 and 3, this correlation persists only in genotype 1 infected patients (OR, 1.15; 95% CI, 1.06- 1.26; P = 0.0011), suggesting a role for hyperferritinemia in fibrosis progression.
As the liver biopsies in this study were all performed to determine whether anti- HCV therapy was indicated, our findings may not be representative of the general HIV- HCV coinfected population. In HIV- seronegative populations, a synergistic interaction between steatosis and even low alcohol consumption is a major determinant of liver fibrosis severity [30]. Indeed, alcohol is believed to impair mitochondrial B- oxidation of fatty acids by causing oxidative damage to mitochondrial enzymes [31]. This risk factor was not evaluable as all our patients were requested to limit their daily alcohol consumption during the 3 months preceding enrolment. Additionally, several studies have shown that the frequency of steatosis varies significantly with ethnicity [32,33]. No such correlation was found in our study but this may be due to the fact that most of our patients were native of France. Our findings contrast with those of a study in HIV- HCV coinfected patients in which age was found to be the only independent factor associated with steatosis, whereas male sex, age, BMI, genotype 3a and histological fibrosis correlated to steatosis in HCV monoinfected patients [18]. In this study, the distribution of HCV genotypes was genotype 1 (80% versus 48% in our study) and genotype 3 (11% versus 35% in our study) and 33% of patients had no fibrosis (versus 0% in our study). Patients were also older (47 ± 7 versus 39.7 ± 5.4 years). These differences together with the number of patients (92 versus 395 in our study) may explain the different findings.
Finally, it is noteworthy that steatosis quantification was performed on routinely stained biopsies. The staining allows an accurate estimation of macrovacuolar steatosis, the most common histopathological feature of HCV- induced steatosis, but might underestimate microvesicular steatosis, a histological pattern of steatosis often related to mitochondrial toxicity. Such assessment is only possible on frozen sections with special staining which were not available in this study.
In conclusion, this large study showed a high prevalence of steatosis in HIV- HCV coinfected patients. The same risk factors for steatosis were identified in these patients as in HCV monoinfected patients. Steatosis was associated with HCV genotype 3 and correlated with HCV viral load. In patients with genotype 1 infection steatosis was associated with more severe fibrosis but not with HCV viral load. The BMI was also an independent risk factor for steatosis in patients with both genotype 1 and genotype 3 infection. In contrast, steatosis was not related to characteristics of HIV infection, including antiretroviral therapy.
References
1.
Mihm S, Fayyazi A, Hartmann H, Ramadori G.
Analysis of histopathological manifestations of chronic hepatitis C virus infection with respect to virus genotype.
Hepatology 1997; 25:735- 739.
2.
Adinolfi LE, Gambardella M, Andreana A, Tripodi MF, Utili R, Ruggiero G.
Steatosis accelerates the progression of liver damage of chronic hepatitis C patients and correlates with specific HCV genotype and visceral obesity.
Hepatology 2001; 33:1358- 1364.
3.
Hourigan LF, Macdonald GA, Purdie D, Whitehall VH, Shorthouse C, Clouston A, Powell EE.
Fibrosis in chronic hepatitis C correlates significantly with body mass index and steatosis.
Hepatology 1999; 29:1215- 1219.
4.
Patton HM, Patel K, Behling C, Bylund D, Blatt LM, Vallee M, et al.
The impact of steatosis on disease progression and early and sustained treatment response in chronic hepatitis C patients.
J Hepatol 2004; 40:484- 490.
5.
Westin J, Nordlinder H, Lagging M, Norkrans G, Wejstal R.
Steatosis accelerates fibrosis development over time in hepatitis C virus genotype 3 infected patients.
J Hepatol 2002; 37:837- 842.
6.
Hezode C, Roudot- Thoraval F, Zafrani ES, Dhumeaux D, Pawlotsky JM.
Different mechanisms of steatosis in hepatitis C virus genotypes 1 and 3 infections.
J Viral Hepat 2004; 11:455- 458.
7.
Lonergan JT, Behling C, Pfander H, Hassanein TI, Mathews WC.
patients receiving nucleoside analogue combination regimens.
Clin Infect Dis 2000; 31:162- 166.
8.
Walker UA, Bauerle J, Laguno M, Murillas J, Mauss S, Schmutz G, et al.
Depletion of mitochondrial DNA in liver under antiretroviral therapy with didanosine, stavudine, or zalcitabine.
Hepatology 2004; 39:311- 317.
9.
Riddle TM, Kuhel DG, Woollett LA, Fichtenbaum CJ, Hui DY.
HIV protease inhibitor induces fatty acid and sterol biosynthesis in liver and adipose tissues due to the accumulation of activated sterol regulatory element- binding proteins in the nucleus.
J Biol Chem 2001; 276:37514- 37519.
10.
Carrat F, Bani- Sadr F, Pol S, Rosenthal E, Lunel- Fabiani F, Benzekri A, et al.
Pegylated interferon alfa- 2b versus standard interferon alfa- 2b, plus ribavirin for chronic hepatitis C in HIV- infected patients.
Randomized controlled trial.
JAMA 2004; 292:2839- 2848.
11.
Bedossa P, Poynard T.
An algorithm for the grading of activity in chronic hepatitis C.
The METAVIR Cooperative Study Group.
Hepatology 1996; 24:289- 293.
12.
Clouston AD, Jonsson JR, Purdie DM, Macdonald GA, Pandeya N, Shorthouse C, Powell EE.
Steatosis and chronic hepatitis C: analysis of fibrosis and stellate cell activation.
J Hepatol 2001; 34:314- 320.
13.
Ramesh S, Sanyal AJ.
Hepatitis C and nonalcoholic fatty liver disease.
Semin Liver Dis 2004; 24:399- 413.
14.
Marks KM, Petrovic LM, Talal AH, Murray MP, Gulick RM, Glesby MJ.
Histological findings and clinical characteristics associated with hepatic steatosis in patients coinfected with HIV and hepatitis C virus.
J Infect Dis 2005; 192:1943- 1949.
15.
Sharma P, Balan V, Hernandez J, Rosati M, Williams J, Rodriguez- Luna H, et al.
Hepatic steatosis in hepatitis C virus genotype 3 infection: does it correlate with body mass index, fibrosis, and HCV risk factors? Dig Dis Sci 2004; 49:25- 29.
16.
Hickman IJ, Clouston AD, Macdonald GA, Purdie DM, Prins JB, Ash S, et al.
Effect of weight reduction on liver histology and biochemistry in patients with chronic hepatitis C.
Gut 2002; 51:89- 94.
17.
Tien PC, Grunfeld C.
The fatty liver in AIDS.
Semin Gastrointest Dis 2002; 13:47- 54.
18.
Monto A, Dove LM, Bostrom A, Kakar S, Tien PC, Wright TL.
Hepatic steatosis in HIV/hepatitis C coinfection: prevalence and significance compared with hepatitis C monoinfection.
Hepatology 2005; 42:310- 316.
19.
Sulkowski MS, Mehta SH, Torbenson M, Afdhal NH, Mirel L, Moore RD, Thomas DL.
Hepatic steatosis and antiretroviral drug use among adults coinfected with HIV and hepatitis C virus.
AIDS 2005; 19:585- 592.
20.
Soriano V, Puoti M, Bonacini M, Brook G, Cargnel A, Rockstroh J, et al.
Care of patients with chronic hepatitis B and HIV co- infection: recommendations from an HIV- HBV International Panel.
AIDS 2005; 19:221- 240.
21.
Moriya K, Yotsuyanagi H, Shintani Y, Fujie H, Ishibashi K, Matsuura Y, et al.
Hepatitis C virus core protein induces hepatic steatosis in transgenic mice.
J Gen Virol 1997; 78:1527- 1531.
22.
Castera L, Hezode C, Roudot- Thoraval F, Lonjon I, Zafrani ES, Pawlotsky JM, Dhumeaux D.
Effect of antiviral treatment on evolution of liver steatosis in patients with chronic hepatitis C: indirect evidence of a role of hepatitis C virus genotype 3 in steatosis.
Gut 2004; 53:420- 424.
23.
Fartoux L, Chazouilleres O, Wendum D, Poupon R, Serfaty L.
Impact of steatosis on progression of fibrosis in patients with mild hepatitis C.
Hepatology 2005; 41:82- 87.
24.
Ortiz V, Berenguer M, Rayon JM, Carrasco D, Berenguer J.
Contribution of obesity to hepatitis C- related fibrosis progression.
Am J Gastroenterol 2002; 97:2408- 2414.
25.
Fernandez- Rodriguez CM, Gutierrez ML, Serrano PL, Lledo JL, Santander C, Fernandez TP, et al.
Factors influencing the rate of fibrosis progression in chronic hepatitis C.
Dig Dis Sci 2004; 49:1971- 1976.
26.
Martin- Carbonero L, Benhamou Y, Puoti M, Berenguer J, Mallolas J, Quereda C, et al.
Incidence and predictors of severe liver fibrosis in human immunodeficiency virus- infected patients with chronic hepatitis C: a European collaborative study.
Clin Infect Dis 2004; 38:128- 133.
27.
Angulo P, Keach JC, Batts KP, Lindor KD.
Independent predictors of liver fibrosis in patients with nonalcoholic steatohepatitis.
Hepatology 1999; 30:1356- 1362.
28.
Bugianesi E, Manzini P, D'Antico S, Vanni E, Longo F, Leone N, et al.
Relative contribution of iron burden, HFE mutations, and insulin resistance to fibrosis in nonalcoholic fatty liver.
Hepatology 2004; 39:179- 187.
29.
Fargion S, Mattioli M, Fracanzani AL, Sampietro M, Tavazzi D, Fociani P, et al.
Hyperferritinemia, iron overload, and multiple metabolic alterations identify patients at risk for nonalcoholic steatohepatitis.
Am J Gastroenterol 2001; 96:2448- 2455.
30.
Serfaty L, Poujol- Robert A, Carbonell N, Chazouilleres O, Poupon RE, Poupon R.
Effect of the interaction between steatosis and alcohol intake on liver fibrosis progression in chronic hepatitis C.
Am J Gastroenterol 2002; 97:1807- 1812.
31.
Pessayre D, Mansouri A, Fromenty B.
Nonalcoholic steatosis and steatohepatitis.
V.
Mitochondrial dysfunction in steatohepatitis.
Am J Physiol Gastrointest Liver Physiol 2002; 282:G193- G199.
32.
Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, et al.
Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity.
Hepatology 2004; 40:1387- 1395.
33.
Weston SR, Leyden W, Murphy R, Bass NM, Bell BP, Manos MM, Terrault NA.
Racial and ethnic distribution of nonalcoholic fatty liver in persons with newly diagnosed chronic liver disease. Hepatology 2005; 41:372- 379.
|
|
| |
| |
|
|
|