| |
SCH 503034 HCV Protease Inhibitor Plus PEG-IFN a-2b in HCV-1 PEG-IFN a-2b
Non-responders: Phase 1b Results
|
| |
| |
Reported by Jules Levin
These are the study results presented by S Zeuzem at AASLD Nov 2005 for the phase I 14 day study of the combination of SCH 503034 plus Peg-IFN a-2b. A larger phase II study is ongoing exploring several dosing regimens including a higher dose of 800 mg three times daily.
S Zeuzem1, C C Sarrazin1, F Wagner2, R Rouzier3, N Forestier1, s Gupta4, M Hussain, A Shah4, D Cutler4, J Zhang4
1Saarland University Hospital, Homburg, Germany
2PAREXEL International GmbH, berlin, germany
3Centre Cap, Montellier, france
4Schering-Plough Research Institute, Kenilworth, NJ, USA
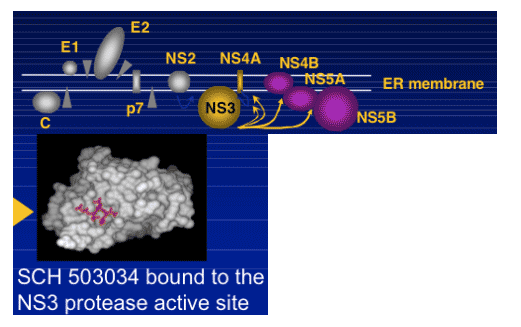
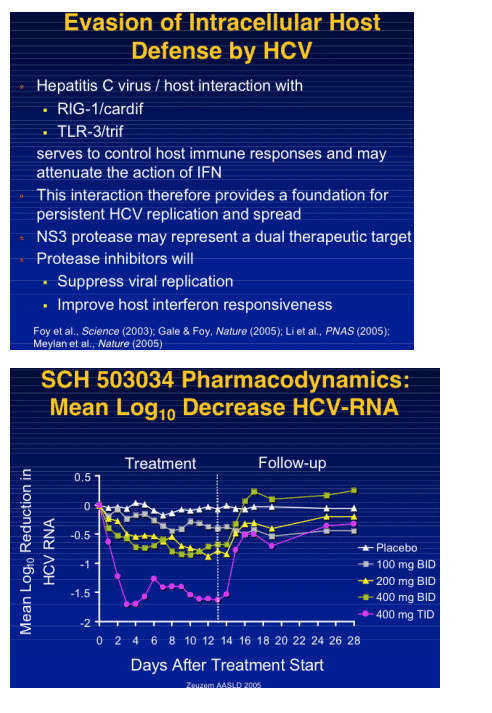

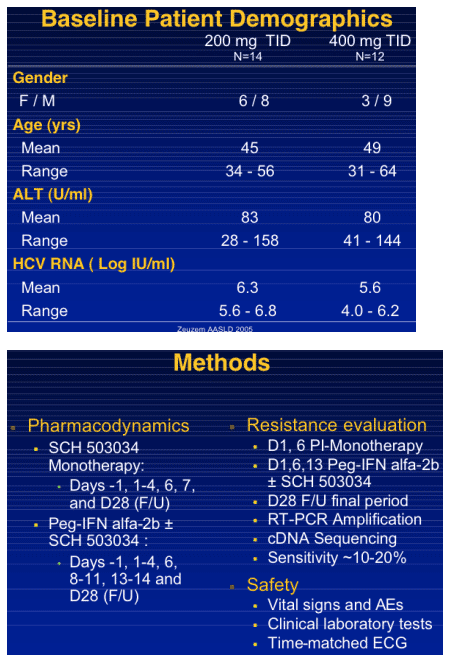
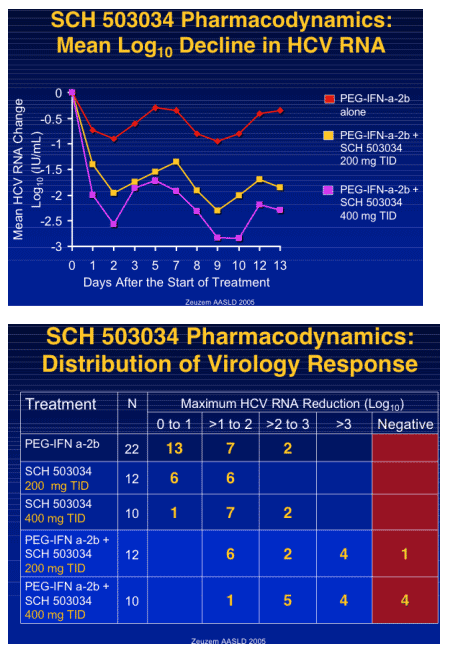
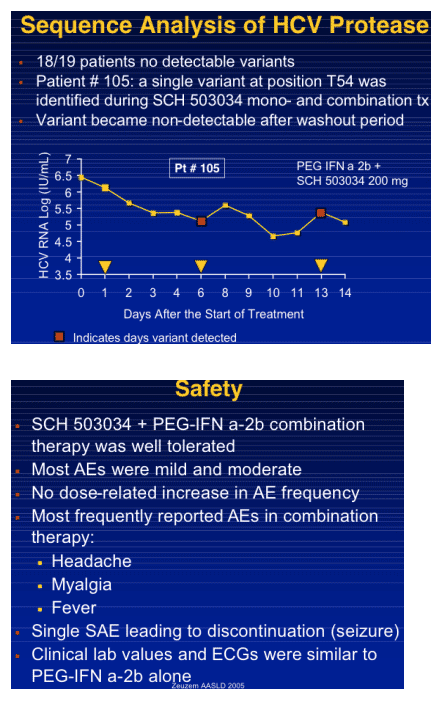
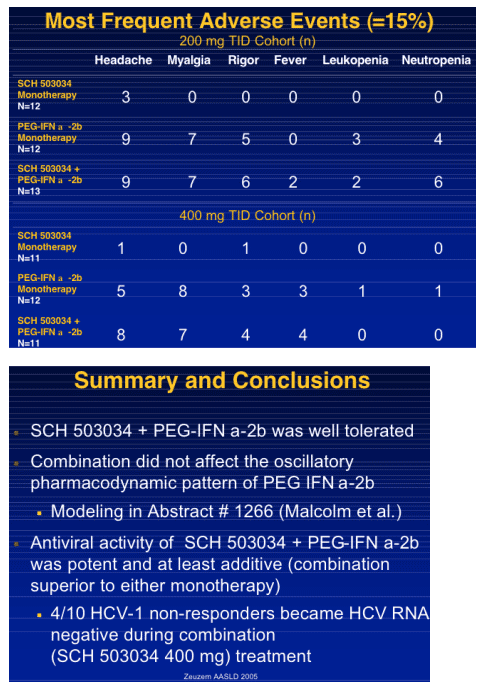
|
|
| |
| |
|
|
|