| |
Combination therapy improves survival rates in AIDS-related lymphoma
|
| |
| |
Feb 27, 2006, 18:02, Reviewed by: Dr. Rashmi Yadav
"The present study showed that our risk-adapted strategy for concomitant administration of HAART with CHOP is effective and safe."
By John Wiley & Sons, Inc. , Combining aggressive HIV therapy and chemotherapy significantly improves the survival rates of HIV-positive men and women treated for lymphoma, according to a new study.
Published in the April 1, 2006 issue of CANCER, a peer-reviewed journal of the American Cancer Society, the study reveals that combination therapy showed the greatest benefit for HIV patients suffering from aggressive malignant non-Hodgkin's lymphoma. This benefit was most pronounced in HIV patients without severely impaired immune functions. These so-called "standard risk" patients responded as well to therapy and survived as long as lymphoma patients without HIV.
Lymphomas are cancers of the immune system's white blood cells. They are treated with chemotherapy, often consisting of a multi-drug regimen using cyclophosphamide, doxorubicin, vincristine and prednisone (CHOP). People with HIV, a virus that depletes integral, specialized white cells called CD-4 cells, are at increased risk of developing lymphomas, particularly aggressive, fast-growing non-Hodgkin type lymphomas. These are called "AIDS-related lymphomas" (ARL) and generally have a poorer prognosis than non-HIV-related lymphomas. Highly active antiretroviral therapy (HAART) revolutionized care of HIV-positive men and women. It not only improves laboratory indicators, such as increased CD-4 cells and reduced viral loads, but also significantly improves survival and delays the onset of AIDS and AIDS-related cancers, including lymphomas.
With the lack of study data to show the efficacy of maintaining HIV-positive patients on HAART while they are treated with chemotherapy for ARL, oncologists are hesitant to expose HIV patients to hypothetical drug toxicities related to combining the therapies. Researchers led by Rudolf Weiss, M.D., of Specialist Practice for Hematology, Oncology and Infectious Diseases in Bremen, Germany, treated 72 HIV-patients with ARL divided into high-risk and standard-risk cohorts with combined CHOP and HAART to evaluate the safety and efficacy of the combined regimen.
The investigators found combined therapy improved survival rates for patients with ARL and standard level of risk to rates comparable to those in non-HIV patients with lymphoma treated with CHOP and superior to previously published rates achieved by CHOP alone. For standard-risk ARL patients 79 percent achieved complete remission, and after 47 months of follow-up and study's end, more than 50 percent of patients survived. Moreover, only 40 percent reported moderate drug toxicity. For high-risk ARL patients, only 29 percent achieved complete remission and median survival was only 7.2 months. Sixty-nine percent reported moderate toxicity.
"The present study showed that our risk-adapted strategy for concomitant administration of HAART with CHOP is effective and safe," the authors concluded.
- The study is published in the April 1, 2006 issue of CANCER, a peer-reviewed journal of the American Cancer Society.
Acquired immunodeficiency syndrome-related lymphoma
Simultaneous treatment with combined cyclophosphamide, doxorubicin, vincristine, and prednisone chemotherapy and highly active antiretroviral therapy is safe and improves survival - Results of the German Multicenter Trial
Cancer
Early View (Articles online in advance of print)
Published Online: 27 Feb 2006
Rudolf Weiss, M.D. 1 *, Paris Mitrou, M.D. 2, Keikawus Arasteh, M.D. 3, Dirk Schuermann, M.D. 4, Marcus Hentrich, M.D. 5, Ulrich Duehrsen, M.D. 6, Hinrich Sudeck, M.D. 7, Ingo G. H. Schmidt-Wolf, M.D. 8, Ioannis Anagnostopoulos, M.D. 9, Dieter Huhn, M.D. 101Private Practice for Hematology, Oncology and Infectious Diseases, Bremen, Germany
2Department of Hematology, Johann Wolfgang Goethe University Hospital, Frankfurt, Germany
3Department of Infectious Diseases, Auguste Viktoria Hospital, Berlin, Germany
4Department of Infectious Diseases, Charite Hospital, Humboldt University, Berlin, Germany
5Department of Hematology and Oncology, Harlaching Hospital, Munich, Germany
6Department of Hematology and Oncology, University of Essen Hospital, Essen, Germany
7Bernhard Nocht Institute for Tropical Medicine, Hamburg, Germany
8Department of Hematology and Oncology, University of Bonn Hospital, Bonn, Germany
9Department of Pathology, Benjamin Franklin Branch, Charite Hospital, Humboldt University, Berlin, Germany
10Department of Hematology and Oncology, Charite Hospital, Humboldt University, Berlin, Germany
Article Text
Highly active antiretroviral therapy (HAART) has changed the biology and clinical presentation of acquired immunodeficiency syndrome (AIDS)-related lymphoma (ARL).[1][2] Although the incidence of AIDS-related primary central nervous system (CNS) lymphoma has decreased significantly, the decline in ARL has been less pronounced or absent.[3-13] Since the introduction of HAART, CD4 counts at diagnosis have become higher, and the survival of patients with systemic ARL has increased; hence, the prognosis of systemic ARL has improved.[14][15] Because of to the decreasing incidence of Kaposi sarcoma and opportunistic infections, an increasing proportion of mortality and morbidity in human immunodeficiency virus (HIV)-infected patients is related to ARL.[3][16][17] Treatment options for patients with ARL recently were reviewed.[18-21]
Because it has been found that HAART improves the survival and quality of life of these patients, it would appear to be detrimental to withhold HAART during chemotherapy. Nevertheless, primary reduction of the intensity of chemotherapy may not be appropriate because of the aggressive nature of ARL. Furthermore, combining HAART with chemotherapy may lead to an increase in adverse drug interactions and toxicity, thereby compromising remission and survival.
This uncertainty has led to the development of different treatment approaches. Some investigators consider chemotherapy with concomitant HAART to be effective and tolerable,[22-24] whereas others prefer to suspend antiretroviral therapy during chemotherapy to avoid excessive toxicity.[25] Consequently, the question of whether combining HAART with chemotherapy outweighs the potential risk of increased toxicity remains controversial.
Therefore, the German AIDS-Related Lymphoma Study Group initiated a prospective, multicenter Phase II trial to address this question. The objective of the current study was to assess the efficacy of combined cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) plus concomitant HAART and to determine whether this treatment modality leads to increased toxicity.
Abstract
BACKGROUND
Highly active antiretroviral therapy (HAART) has improved the survival of patients with acquired immunodeficiency syndrome-related lymphoma (ARL). The German ARL Study Group investigated whether HAART administered concomitantly with cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) chemotherapy compromised the course of immune parameters during and after chemotherapy and exerted a positive effect on remission and survival.
METHODS
From 1997 to 2001, 72 patients with ARL were stratified prospectively into a standard-risk group (n = 48 patients) and a high-risk group (n = 24 patients) with either 0-1 or 2-3 of the following risk factors: CD4 < 50/uL, prior opportunistic infection, and/or a World Health Organization performance status >/= 3. Patients in the high-risk group received >/= 75% of the CHOP regimen.
RESULTS
In the standard-risk group (CD4 = 223/L; age-adjusted International Prognostic Index [aaIPI], 38% >/= 2), the complete remission (CR) rate was 79%, and median survival was not reached after a median 47 months of follow-up. CD4 counts did not change from baseline to 4 weeks after the end of chemotherapy (206/uL). In the high-risk group (CD4 = 34/uL; aaIPI, 88% >/= 2), the CR rate was 29%, and the median survival was 7.2 months (3 patients survived for > 3 yrs). Toxicity was moderate: Leukopenia Grade 3 or 4 occurred in 100 of 249 chemotherapy cycles (40%) in the standard-risk group and in 70 of 102 cycles (69%) in the high-risk group.
CONCLUSIONS
Based on the aaIPI, the survival of patients in the standard-risk group was very similar to that achieved by nonhuman immunodeficiency virus-infected patients who had aggressive lymphomas. Concurrent CHOP plus HAART can be administered in an outpatient setting. Thus, the authors recommend using this modality as first-line therapy for patients with ARL. Cancer 2006. 2006 American Cancer Society.
Patients
HIV-positive patients, as determined by Western blot analysis, who were diagnosed with malignant lymphoma were documented by the German AIDS-Related Lymphoma Study Group. Biopsy materials were reviewed and diagnoses were confirmed by one of three German lymphoma reference centers. Patients who were eligible for this trial had aggressive B-cell lymphomas (the Revised European-American Lymphoma [REAL] classification).[26] Patients whose organ function was adequate unless impaired by non-Hodgkin lymphoma (NHL) were treated as specified below. Patients with infections that prevented treatment or primary CNS lymphomas were ineligible. Participants were enrolled consecutively between 1997 and 2001. Each participating center's local ethics committee approved the trial protocol, and all patients gave their written informed consent.
According to these criteria, 157 patients with lymphoproliferative diseases were registered on the study. Seventy-seven of those patients were excluded (see below), and 8 patients were lost to follow-up before chemotherapy was started. Of the 72 patients who were treated according to protocol, 48 patients were classified as standard risk, and 24 patients were classified as high risk (see Fig. 1).
Exclusion criteria included refractory, active infections and opportunistic infections; serious cardiovascular, pulmonary, psychiatric, or metabolic disease; inadequate renal or hepatic function; treatment with chemotherapy or cytokines within the preceding 4 weeks with the exception of granulocyte-colony stimulating factor (G-CSF), granulocyte-macrophage-colony stimulating factor, or erythropoetin; pregnancy; life expectancy < 4 weeks; primary CNS involvement, and Ann Arbor Stage IA lymphoma (not Stage IE) located above the diaphragm. Patients who fulfilled these criteria were excluded without follow-up. Patients with Stage IA lymphomas above the diaphragm and patients with primary CNS lymphoma received radiation therapy only and were documented off-study. This treatment was standard practice in Germany at the time the study was planned.[27-30]
Patients who were included in the prospective treatment protocol were stratified into two subgroups. High-risk patients had 2 or 3 of the following risk factors: CD4 lymphocytes < 50/L at the time of lymphoma diagnosis, a World Health Organization (WHO) performance status 3, or a previous AIDS-defining opportunistic infection. Standard-risk patients had none or only one of those risk factors.
The histologic diagnosis was made according the REAL classification.[26] One of three German lymphoma reference centers (Prof. Dr. H. Stein, Berlin and Prof. Dr. M.-L. Hansmann, Frankfurt; Prof. Dr. H.-K. Mueller-Hermelink, Wuerzburg) reviewed the biopsy material and confirmed the diagnosis.
The stage of the disease at the time of diagnosis was determined by computed tomographic scans of the chest, abdomen, and pelvis; unilateral bone marrow aspiration and biopsy; and lumbar puncture, including quantitative and qualitative cytologic evaluations for the presence of lymphoma cells. Lymphomas were staged according to the Ann Arbor classification. Additional tests at baseline included blood cell counts, blood chemistry, CD3, CD4, CD8 lymphocyte counts, quantitative serum immunoglobulins, viral load, and urinanalysis.
Treatment
Patients in the standard-risk group received CHOP on the following schedule: intravenous cyclophosphamide 750 mg/m2, intravenous doxorubicin 50 mg/m2, and intravenous vincristine 1.4 mg/m2 (maximum dose, 2.0 mg) all given on Day 1 of each cycle together with oral prednisolone 100 mg/m2 on Days 1-5. Each cycle was repeated at Day 22. Patients in the high-risk group received >/= 75% of the CHOP regimen in the first cycle, but dose escalation to 100% was permissible based on the neutrophil count and the occurrence of infectious complications. The objective of this strategy was to avoid excessive toxicity in this vulnerable risk group.
Both standard-risk patients and high-risk patients received six cycles of CHOP. In both groups, antiretroviral therapy was continued or started with induction therapy in all but six patients. Three of those patients had viral loads < 50,000 copies/uL, and 3 patients refused parallel therapy after they had been included in the study. Two of the latter patients (all in the high-risk group) did not achieve complete remission (CR).
Intrathecal methotrexate (15 mg) was given to each patient initially and subsequently was continued with each chemotherapy cycle in patients who had Stage IV disease or who had lactate dehydrogenase (LDH) levels > 800 U/L (normal range, 80-240 U/L). Patients who had Grade 3 or 4 granulocytopenia or prolongation of the treatment interval received G-CSF beginning on Day 4 of the subsequent cycle. Pentamidine inhalation or trimethoprim-sulfamethoxazole (TMP/SMZ) prophylaxis was used to prevent Pneumocystis carinii pneumonia. Standard-risk patients received maintenance therapy with interferon-a. This part of the study was omitted later to guarantee full-dose antiretroviral therapy.
Evaluation
Treatment response was assessed after Cycles 2, 4 and 6. Posttreatment follow-up examinations were performed bimonthly. CR was defined as the complete disappearance of all known lymphoma manifestations that lasted >/= 4 weeks. Partial remission (PR) was defined as a reduction > 50% in of the sum of the products of the greatest cross-sectional dimensions of all known measurable lesions. Nonresponders were patients who attained less than PR or who experienced progressive disease (PD).
WHO criteria were used to assess toxicity. The age-adjusted International Prognostic Index score (aaIPI) was used to ensure the comparability of our results with results from other studies.[31-33]
Statistical Analysis
In an intention-to-treat analysis, survival was calculated as the time from diagnosis of malignant lymphoma until the date of death or of last patient contact. Survival functions and median survival were estimated with the Kaplan-Meier method and were compared by using the log-rank test and a Cox proportional hazard model. Factors that influenced remission rates or recurrences were included in a logistic regression analysis. The criterion for inclusion in the multivariate model was a level of significance of P
DISCUSSION
In the current study, all patients with lymphoproliferative diseases diagnosed in the participating clinics were documented and subsequently evaluated for eligibility in the study. Screening was carried out by the study center to avoid patient selection by the treating physician. Ultimately, 72 of 157 documented patients qualified for inclusion in the trial. Of the 72 patients who were treated according to protocol, 48 were defined as standard-risk patients (0-1 risk factor), and 24 were defined as high-risk (2-3 risk factors). The risk factors were as follows: CD4 count < 50/uL, prior opportunistic infections, and/or a performance status > 2. These criteria, as demonstrated in a previous study,[34] can differentiate sharply between a standard-risk group of patients who have a good chance of long-lasting remission and a tolerable number of toxic or infectious events and a high-risk group of patients who will not tolerate even standard chemotherapy. These results are well in line with a recently published prognostic score for patients with ARL.[35]
In the high-risk group, HAART plus CHOP induced CR in 7 of 24 (29%) patients. The median survival was 7.2 months, and 9 patients had either stable disease or PD. The high rate of progression also led to premature termination of therapy, as is reflected by the lower number of chemotherapy cycles in this group. The prognosis for high-risk patients generally is dismal: The median survival was only 4.5 months in the earlier trial,[34] and it was 7.2 months in the current trial. Nevertheless, 3 of our high-risk patients have survived for > 3 years.
In the standard-risk group, HAART plus CHOP produced a considerable rate of CRs (79%), and the median survival had not been reached after a median 47 months of follow-up. This result is superior to that achieved in the previous Phase II study[34] that was carried out in the pre-HAART era (21.1 mos). The data presented here demonstrate that, in standard-risk patients, CHOP plus HAART can achieve results comparable to those achieved in non-HIV-infected patients.[36]
Regarding toxicity, combined CHOP and HAART was assessed as tolerable. In standard-risk patients, the median length of the first 4 chemotherapy cycles was 21 days; Grade 3 and 4 leukopenia occurred in 100 of 249 chemotherapy cycles (40%), and G-CSF was administered in 65 of 249 cycles (26%). In high-risk patients (102 chemotherapy cycles), Grade 3 and 4 leukopenia occurred in 70 cycles (69%), and G-CSF was given in 54 cycles (53%). One patient died of neutropenia, which was caused by an infection of unknown origin.
The single, main cause of death in both risk groups was malignant lymphoma, as evidenced by the fact that fatal PD occurred in 10 of 48 standard-risk patients and in 17 of 24 high-risk patients. This contrasts with our previous findings, which demonstrated a high rate of deaths related to infections, and to opportunistic infections in particular.[34]
These results suggest that concomitant HAART contributed considerably to improving the response rates and survival of patients in the standard-risk group who were treated in the current study. In addition, their immune parameters recovered shortly after the completion of chemotherapy, and their viral load decreased during the treatment cycles.
The favorable changes in CD4-lymphocytes counts and HIV viral loads are in accordance with previously published results on chemotherapy in combination with HAART.[22] Inversely, the HIV viral load increases when HAART is withheld during chemotherapy.[25] The importance of a functioning immune system was illustrated by Stebbing et al., who demonstrated that successful HAART and the subsequent improvement of immune parameters lowered the incidence of malignant lymphomas.[37] With the exception of one study,[25] neither low-dose nor intensified chemotherapy improved the response or survival rates in patients with ARL compared with standard CHOP. This is true for both the pre-HAART era and the HAART era.[22-24][34][38-42] Little and colleagues achieved excellent response and survival results with an infusional regimen. In their study, suspension of HAART during chemotherapy and 50% reduction of the initial cyclophosphamide dose in patients with low CD4 lymphocytes did not affect outcomes negatively and appeared to be associated with less toxisity.[25] In the current study, we achieved comparable results in our group of standard-risk patients, who had very similar baseline patient characteristics, by using standard CHOP in an outpatient setting.
Future studies will be needed to determine whether treatment modifications can improve patient outcomes and further reduce the toxicity of CHOP plus HAART, particularly in high-risk patients. The treatment of non-HIV-infected patients with aggressive lymphomas was improved by adding monoclonal antibodies to CHOP[43][44]; similarly, combining anti-CD20 monoclonal antibody to CHOP plus HAART may improve outcomes further among patients with ARL. However, the problem of the long-lasting deterioration of B-lymphocyte function must be addressed adequately. Three different trials with chemoimmunotherapy resulted in significantly divergent response, survival, and toxicity results; and those differences could not be attributed to differences in baseline patient characteristics.[42][45][46] However, short follow-up in some reports makes it difficult to compare or interpret the data. In a recent publication,[47] the pooled results of 3 Phase II trials (rituximab with cyclophosphamide, doxorubicin, and vepeside) showed promising remission and survival data; however, compared with cyclophosphamide, doxorubicin, and vepeside, a high rate of treatment-related toxicity was observed.
In conclusion, the current results show that our risk-adapted strategy for concomitant administration of HAART with CHOP is effective and safe. In standard-risk patients, the treatment achieved median survival rates that were comparable to those obtained in non-HIV-infected patients who had aggressive lymphomas. Future studies should focus on dose-intense CHOP-like regimens or on the potential of chemoimmunotherapy for further improving outcomes in patients with ARL. Another advantage of CHOP plus HAART is that it can be administered in an outpatient setting, thus saving treatment resources. Therefore, we recommend the use of CHOP plus HAART as first-line therapy for patients with ARL.
RESULTS
Patients Characteristics
Patient characteristics are shown in Table 1. Standard-risk patients exhibited a better performance status and better aaIPI scores; no prior opportunistic infections, higher lymphocyte counts, including T-cell subsets; and lower viral loads. The standard-risk and high-risk groups did not differ with respect to age, lymphoma stage, extranodal manifestations, presence of B-symptoms, LDH levels, or immunoglobulin G levels. Histologic subgroups were distributed evenly between the two risk groups.
Average CD4 count was 175 for all patients & 223 for High Risk patients.
Dose Intensity
A dose intensity of 97% was achieved in the standard-risk group. In the high-risk group, 17 of 24 patients (71%) received 100% of the CHOP regimen in the first cycle, and the overall CHOP dose intensity was 91%.
Response to Treatment
The median follow-up was 26.2 months for the overall group and 47 months for the standard-risk group. The number of patients who achieved CR was 79% in the standard-risk group versus 29% in the high-risk group (Table 2, Fig. 2). The median survival was 26.1 months for the overall group and 7.2 months for the high-risk group. The median survival for the standard-risk group had not yet been reached after a median follow- up of 47 months. Currently, 60% of patients in the standard-risk group are alive after 3 years of follow-up, and 33% are alive after 5 years. The aaIPI-based overall survival and remission data are shown in Table 3 and in Figures 3-5.
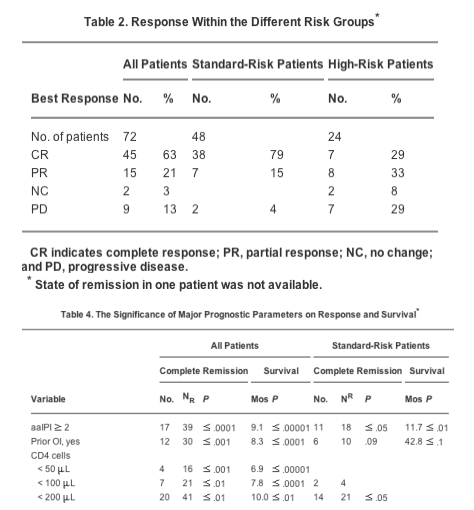
In the standard-risk-group, aaIPI scores >/= 2, prior opportunistic infections, CD4 lymphocytes, and elevated LDH levels had a negative impact on CR. aaIPI scores >/= 2, prior opportunistic infections, bone marrow involvement, thrombocytopenia, and elevated LDH levels predicted shorter survival.
Factors that were significant at the .1 level were included in a multivariate analysis (Cox model/logistic regression), which yielded the following results: For the overall group, CD4 lymphocytes < 100/uL (P 300 U/L (normal range, 80-240 U/I; P /= 2 (P /= 2 (P
In the standard-risk group, CD4 lymphocytes < 200/uL (P 300 U/L (P
Immune Parameters and Viral Load
Lymphocyte counts for the standard-risk group at the start of chemotherapy, during chemotherapy, and 4 weeks after the completion of chemotherapy are shown in Figure 6. Lymphocyte counts decreased significantly during treatment; but CD3, CD4, and CD8 cell counts assessed 4 weeks after the completion of chemotherapy did not differ significantly from baseline counts. HIV viral loads decreased during chemotherapy, and counts measured during treatment were significantly lower that those measured at baseline.
Toxicity
Grade 3 and 4 leukopenia occurred in 48% of all chemotherapy cycles in the overall group. In the standard-risk group, Grade 3 or 4 leukopenia occurred in 40% of cycles, and G-CSF was administered in 26% of cycles. In high-risk patients, Grade 3 or 4 leukopenia occurred in 69% of the chemotherapy cycles, and G-CSF was given in 53% of cycles. Neutropenic fever occurred in 28 cycles (11%) in the standard-risk group and in 31 cycles (30%) in the high-risk group.
Grade 3 and 4 peripheral neurologic disorders were seen in 8 patients in the standard-risk group and in 2 patients in the high-risk group (Table 5). The first 4 chemotherapy cycles lasted a median 21, 22, 21, and 21 days, respectively, in the standard-risk group and slightly longer (21, 23, 24, and 24 days) in the high-risk group.
Independent of the risk group, progression and recurrence of lymphoma were the most frequent causes of death. Thirty-eight patients died during the course of this study (17 standard-risk patients, 21 high-risk patients). Regarding the 17 deaths in the standard-risk group, 10 patients died of PD, 1 patient died during PR (cirrhosis, 4.9 mos; cytomegalovirus pneumonia, 6.1 mos), 4 patients died during CR (sepsis, 8.7 mos; cirrhosis, 26.2 mos; heroin overdose, 42.8 mos; myocardial infarction, 64.7 mos), and 1 patient died on Day 27 because of sepsis during neutropenia.
Regarding the 21 deaths in the high-risk group, 17 patients died of PD, and 1 patient died of wasting syndrome (5.2 mos). The other 3 patients died during CR (cirrhosis, 6.2 mos; sepsis, 10 mos; myocardial infarction, 16.8 mos).
|
|
| |
| |
|
|
|