| |
Landmark Discovery of a Kaposi's Sarcoma-Associated Herpesvirus Receptor Provides New Perspectives on Disease Associated with HIV/AIDS
|
| |
| |
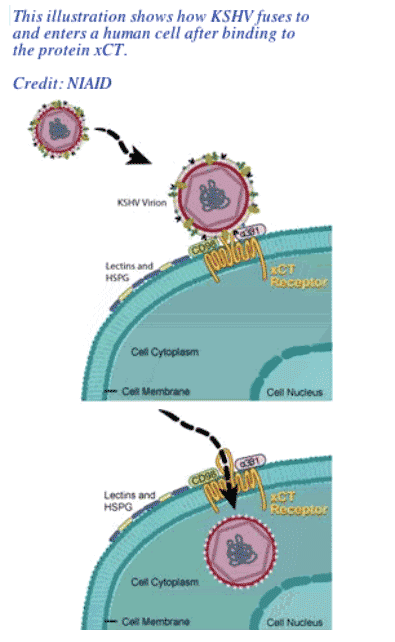
Researchers at the National Institute of Allergy and Infectious Disease (NIAID), a component of the National Institutes of Health (NIH), have identified a critical human cell surface molecule involved in infection by Kaposi's sarcoma herpesvirus (KSHV), the virus that causes Kaposi's sarcoma and certain forms of lymphoma. Kaposi's sarcoma is a major cancer associated with HIV/AIDS, and it typically manifests as multiple purple-hued skin lesions.
In the March 31, 2006 issue of Science, NIAID research fellow Johnan Kaleeba, Ph.D. and senior investigator Edward A. Berger; Ph.D., describe how the molecule xCT is a major gateway that KSHV uses to enter human cells. The molecule may also play a role in the development of Kaposi's sarcoma and other syndromes associated with the virus.
The natural function of xCT in the body is to transport molecules necessary for protecting against stress into cells. When cells are stressed, they express more xCT on their surfaces. Of note, this sort of stress can be caused by KSHV itself. This suggests that the virus may facilitate its own infectivity and dissemination in the body by inducing a physiological state that results in increased numbers of its own receptor.
"The advancement of knowledge achieved in this study highlights the outstanding intramural research that takes place here on the NIH campus," says Elias A. Zerhouni, M.D., NIH director.
"Understanding the mechanisms of cell entry of Kaposi's sarcoma herpesvirus is a landmark achievement in and of itself," says NIAID director Anthony S. Fauci, M.D. "But the connection between the virus and expression of its own receptor on a cell is even more provocative because it might change the way we think about KSHV-associated diseases and their treatment."
Although less common in the United States now than early in the AIDS pandemic, Kaposi's sarcoma is still the most common cancer associated with HIV infection. Prior to the AIDS pandemic, it was an obscure disease. First identified as a multi-pigmented skin disease by a Hungarian doctor named Moritz Kaposi in 1872, it was considered to be quite rare - a medical curiosity usually found in particular populations such as older Italian men, transplant patients and young men in certain parts of sub-Saharan Africa. But then at the dawn of the AIDS pandemic in the early 1980s, the small purplish Kaposi's sarcoma skin lesions began appearing on the bodies of young American men, many of whom went on to develop opportunistic infections.
Dr. Berger became interested in KSHV because of his interest in how viruses enter cells. A decade ago, his research team was the first to identify CXCR4 as one of the coreceptors that allows HIV to gain entry into cells of the immune system. This discovery quickly led to the identification by Dr. Berger's group and several other research teams of CCR5 as the other HIV coreceptor.
By applying the same technology used to identify CXCR4, Drs. Kaleeba and Berger ultimately identified the protein xCT as the receptor that can make cells permissive for KSHV fusion.
The NIAID discovery may lead to new avenues for treating KSHV, says Dr. Berger. Moreover, their finding should enable scientists to determine whether levels of xCT determine disease severity. It also will allow researchers to study whether the expression of xCT on cells varies among different groups of people and whether these variations are genetic or environmental. This research may ultimately explain why certain groups are more at risk for Kaposi's sarcoma.
"Our finding provides a new perspective on the disease," says Dr. Kaleeba, who is originally from Uganda where Kaposi's sarcoma accounts for at least 10 percent of known tumors. "Hopefully this will be the beginning of exciting new directions in this field, as it is likely to provide a useful framework for integration of the cell biology and epidemiology of this clinically important virus."
www.nih.gov
Kaposi's Sarcoma-Associated Herpesvirus Fusion-Entry Receptor: Cystine Transporter xCT
Johnan A. R. Kaleeba and Edward A. Berger*
Science 31 March 2006:
Vol. 311. no. 5769, pp. 1921 - 1924
DOI: 10.1126/science.1120878
Kaposi's sarcoma-associated herpesvirus (KSHV, human herpesvirus 8) is the causative agent of Kaposi's sarcoma and other lymphoproliferative syndromes often associated with HIV/AIDS. Functional complementary DNA selection for a receptor mediating KSHV cell fusion identified xCT, the 12-transmembrane light chain of the human cystine/glutamate exchange transporter system x -c. Expression of recombinant xCT rendered otherwise not susceptible target cells permissive for both KSHV cell fusion and virion entry. Antibodies against xCT blocked KSHV fusion and entry with naturally permissive target cells. KSHV target cell permissiveness correlated closely with endogenous expression of xCT messenger RNA and protein in diverse human and nonhuman cell types.
Laboratory of Viral Diseases, National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health, Bethesda, MD 20892, USA.
KSHV is etiologically linked to Kaposi's sarcoma and other lymphoproliferative syndromes that occur more frequently in immunocompromised individuals, including those with HIV/AIDS (1). Entry of herpesviruses into host cells generally occurs via attachment to the cell surface, followed by direct fusion between the virion and target cell plasma membranes; alternative routes involving receptor-mediated endocytosis have been reported in some cases (2). KSHV can infect a broad range of adherent cell types of different species and tissue lineages in vitro, which indicates broad distribution of the host cell components associated with virus entry; however, the outcome is almost always latency rather than productive infection (3). Cells expressing recombinant KSHV glycoproteins undergo cell fusion (4); this provides support for a direct fusion entry mechanism. Consistent with these infectivity and fusion studies, we have found that a wide range of cell types from diverse tissues and species are permissive for KSHV glycoprotein-mediated cell fusion and virion entry (5); we hypothesized that these processes require a specific KSHV fusion-entry receptor(s) on the target cell surface, distinct from molecules previously implicated in the attachment and entry process.
For KSHV receptor identification, we adapted a vaccinia-based cell fusion assay in which "effector" cells expressing surface viral glycoprotein(s) (and infected with a recombinant vaccinia virus encoding bacteriophage T7 RNA polymerase) were mixed with "target" cells expressing the relevant surface receptor(s) (and containing the Escherichia coli LacZ gene linked to the T7 promoter, introduced by plasmid transfection or recombinant vaccinia virus infection); B-galactosidase (B-gal) activity provides a quantitative measure of cell fusion (6). Our functional strategy for KSHV receptor identification (7) made no assumptions about the specific viral or cellular molecules involved but, instead, relied on the notion that a receptor-encoding cDNA within a library derived from a fusion-permissive cell line would confer fusion susceptibility to an otherwise nonpermissive target cell; use of a selectable reporter would enable enrichment of the fused cells containing the desired cDNA. As KSHV nonpermissive fusion targets, F515 or NIH 3T3 murine fibroblasts were transfected with plasmid pJK7-T7Beacon containing the T7 promoter linked to a tripartite reporter cassette encoding surface epitope tags from influenza virus hemagglutinin (HA) and Myc, plus cytoplasmic enhanced green fluorescence protein (EGFP) (fig. S1A). The targets were also infected with Mel-dT2 (fig. S1B), a library of recombinant vaccinia viruses (8) containing cDNAs from the Mel 1700 human melanoma cell line, a highly permissive KSHV fusion target (5). As effectors, the KSHV chronically infected BCBL-1 cell line was pre-activated with 12-O-tetradecanoyl-13-phorbol acetate (TPA) to induce KSHV glycoprotein expression, then infected with vaccinia MVA/T7 pol. Preliminary analyses confirmed the functionality of the reporter plasmid, as well as the presence of putative KSHV receptor-encoding cDNAs within the Mel-dT2 library (fig. S2A). The receptor cDNA identification procedure (7) involved successive selection rounds to enrich for fused cells; at each step, the associated vaccinia viruses were amplified and used for the next round. With three selection rounds (HA/Myc magnetic immunobeads, ring harvesting of EGFP-positive foci, HA/Myc immunobeads), the vaccinia viruses were increasingly enriched for fusion-promoting activity (fig. S2B). Individual vaccinia clones from the final round were plaque-purified, and several were tested in the KSHV cell fusion assay. Clones that scored positive all contained cDNA inserts with the identical nucleotide sequence, whereas those that scored negative contained other cDNA sequences.
The fusion-conferring cDNA sequence encoded human xCT [gene name SLC7A11; EMBL/GenBank accession numbers AB026891 [GenBank] (9) and AJ277882 [GenBank] (10)], the 501-amino acid light chain of transport system x -c that mediates uptake of extracellular L-cystine coupled to efflux of L-glutamate. The x -c system is a member of the family of heterodimeric amino acid transporters (HATS); each contains a distinct nonglycosylated light chain that determines substrate selectivity, linked by an extracellular disulfide bond to the 4F2hc heavy chain (a component of the 4F2 antigen, also designated CD98), a ubiquitous multifunctional type II glycoprotein (11). Like other HAT light chains, xCT is predicted to have 12-transmembrane domain (TMD) helices and cytoplasmic N and C termini (fig. S3).
The ability of recombinant xCT to confer KSHV permissiveness to nonsusceptible targets ("gain-of-function") was tested in the KSHV cell fusion assay (5, 7), which enabled analysis of KSHV glycoprotein function independent of other virus components. Transiently expressed xCT rendered NIH 3T3 targets highly permissive for fusion with effectors expressing transfected KSHV glycoproteins (Fig. 1A); in addition, stably expressed xCT converted BHK-21 and K-562 cells into efficient KSHV fusion targets and also significantly enhanced the moderate target cell activity of HeLa cells (Fig. 1B). The gain-of-function criteria were extended to the KSHV virion entry assay (5, 7) with the use of recombinant virus rKSHV.219 containing the EGFP gene linked to a constitutive cellular promoter (12). This method enabled analysis of viral glycoprotein function in the context of infectious virions, independent of KSHV gene expression. Whereas negligible virion entry occurred with parental BHK-21 and K-562 targets, the corresponding xCT stable transfectants supported significant entry, indicated by increased EGFP-positive cells (Fig. 1C). Complementary "loss-of-function" experiments were performed with target cells naturally permissive for KSHV fusion and entry (5) by examining the effects of rabbit antisera against synthetic peptides (7) derived from predicted antigenic regions of xCT, with topological arrangements based on the 12-TMD model (fig. S3). KSHV cell fusion with BS-C-1 (African Green Monkey kidney cell) targets (Fig. 2A) was inhibited in a dose-dependent fashion by antisera against two putative extracellular regions of xCT (peptides P66-77 and P218-232), but not by antisera against two putative intracellular regions (peptides P25-40 and P97-109); surprisingly, dose-dependent inhibition was also observed with antiserum against peptide P255-270 derived from a putative short intracellular region of xCT [discussed in the legend to (fig. S3)]. In all cases, the corresponding preimmune sera were without effect. KSHV virion entry into Vero (African Green Monkey kidney) cells (Fig. 2, B and C) was strongly inhibited by antisera against putative extracellular peptides P66-77 and P218-232, but not intracellular peptide P97-109; as observed for cell fusion, antisera against peptide P255-270 also blocked KSHV virion entry. Consistent results were obtained with other naturally permissive target cells: KSHV fusion with human PCI-13 (head-and-neck squamous cell carcinoma) and Mel-1700 (melanoma) targets was inhibited by antisera against P255-270 (Fig. 2D) and P218-232; KSHV virion entry into RD (muscle spindle rhabdosarcoma) cells was also blocked by antisera against P66-77, P218-232, and P255-270. Thus, xCT is the predominant mediator of KSHV fusion and entry permissiveness for all five cell lines tested from different species and tissue lineages.
xCT mRNA is found in many human tissues and cell lines (10). Reverse transcription polymerase chain reaction (RT-PCR) and flow cytometry analyses (7) of diverse human cell types indicated that KSHV fusion permissiveness correlated with xCT expression. Thus, xCT mRNA was detected in all fusion-positive cell lines, at especially high levels in the strongest targets (fusion activities >300: Mel-1700, U-87, and PCI-13); by contrast, xCT mRNA was barely detectable in the weak targets (fusion activities <5: CA-46, Ramos, BJAB, KG-1, HSB-2, Huh-7, and K562) (Fig. 3A). Similarly, xCT protein levels varied widely among the cells examined (Fig. 3B) and correlated closely with the corresponding KSHV fusion activities (Fig. 3C).
Transporter proteins containing multiple TMDs (including 12-TMDs) function as receptors for several - and -retroviruses (13); xCT represents an example of the use of this class of molecules by a herpesvirus. Although we demonstrated that xCT functions as a major KSHV fusion-entry receptor in a wide array of human and nonhuman cells from diverse tissue lineages, our findings do not exclude the possible involvement of an alternative molecule (or molecules) for some other targets, nor do they refute previous findings that KSHV enters some cell types by an endocytic pathway (14). Indeed, the potential for the same virus to use alternate receptors and entry mechanisms in a host cell-dependent fashion is a growing theme in the herpesvirus field (2). Our findings of insignificant KSHV target cell activity among human B cell lines, coupled with the low xCT mRNA levels in these cells, is consistent with, and may explain, a previous report demonstrating the failure of several B cell lines to support the early steps of KSHV infection (3). Furthermore, by using RT-PCR, we did not detect xCT mRNA in human CD19+ primary B cells isolated from fresh peripheral blood mononuclear cells. These results remain puzzling because KSHV can infect cultured primary B cells and is also associated with B cell neoplasms in infected people (1). Therefore, more in-depth studies are required to unravel the various factors that influence the relation between xCT regulation and KSHV entry into cells of the B lymphocyte lineage.
The receptor function of xCT suggests new perspectives on the role of other cell surface molecules previously implicated in KSHV entry, particularly heparan sulfate (15, 16) and integrin 3B1 (17). Sulfated proteoglycans and integrins play critical roles in enhancing attachment and internalization of diverse viruses (18-20); furthermore, accumulating evidence points to complex signaling pathways triggered by KSHV interaction with integrin 3B1 (17, 21). Coupled with the recent observation (22) that 4F2hc forms multimeric complexes containing HAT light chains (including xCT) plus B1 integrins (including 3B1), our findings suggest a possible interplay between integrin signaling events and xCT-mediated KSHV entry.
By mediating cystine uptake, a rate-limiting step for glutathione (GSH) biosynthesis, the x -c system plays a central role in maintaining intracellular GSH levels during oxidative stress. xCT is up-regulated in response to GSH depletion, e.g., on exposure to reactive oxygen species (ROS) (23, 24). Redox conditions also have substantial influences during KSHV infection, because hypoxia can induce lytic replication in chronically infected cell lines by activating hypoxia response elements in key lytic-phase genes (25). Moreover, it has been reported that KSHV induces ROS production in cultured endothelial cells and that ROS exposure results in enhanced virus entry (26). In light of these complex virus-host interactions, the identification of xCT as a KSHV receptor suggests novel pathogenic mechanisms whereby the virus might induce or exploit physiologic responses (i.e., ROS production and xCT up-regulation) that favor its own reactivation and dissemination. It is also noteworthy that intracellular GSH levels are progressively depleted during the course of HIV disease (27), at least in part because of HIV Tat-mediated down-regulation of GSH biosynthesis (28) and regeneration (29), plus enhancement of extracellular GSH hydrolysis (30). These concerted activities presumably contribute to the recently described Tat stimulation of xCT expression (30), as well as KSHV entry and infectivity (31). Thus, beyond the obvious consequences of immunosuppression, HIV coinfection might foster clinically aggressive HIV/AIDS-associated Kaposi's sarcoma by the additional mechanism of KSHV receptor up-regulation.
|
|
| |
| |
|
|
|