| |
Micronutrient Supplementation Increases CD4 Count in HIV-Infected Individuals on Highly Active Antiretroviral Therapy
|
| |
| |
Micronutrient Supplementation Increases CD4 Count in HIV-Infected Individuals on Highly Active Antiretroviral Therapy: A Prospective, Double-Blinded, Placebo-Controlled Trial
JAIDS Journal of Acquired Immune Deficiency Syndromes: Volume 42(5) 15 August 2006 pp 523-528
[Rapid Communication]
Kaiser, Jon D. MD*; Campa, Adriana M. PhD ; Ondercin, Joseph P. PA-C ; Leoung, Gifford S. MD ; Pless, Richard F. PhD ; Baum, Marianna K. PhDdagger;
From the *Department of Medicine, University of California at San Francisco Medical School, SF, CA; Florida International University, Miami, FL; The Jonathan Lax Treatment Center, Philadelphia, PA; HIVCare, Saint Francis Memorial Hospital, SF, CA; and Ovation Research Group, Highland Park, IL.
Received for publication January 26, 2006; accepted May 25, 2006.
Supported in full by grant from the Bristol-Myers Squibb Company.
Abstract
Objective: To examine the immunologic, metabolic, and clinical effects of broad spectrum micronutrient supplementation in HIV-infected patients taking highly active antiretroviral therapy (HAART).
Design: A prospective, randomized, double-blinded, placebo-controlled trial.
Methods: Forty HIV-infected patients taking a stavudine and/or didanosine-based HAART regimen were prospectively randomized to receive micronutrients or placebo twice daily for 12 weeks. Data were collected at 4-week intervals including immunologic, metabolic, and clinical measurements. The study examined the effect of micronutrient supplementation on immunologic parameters as the primary end point. The secondary end points were metabolic and clinical effects and distal symmetrical polyneuropathy.
Results: The mean absolute CD4 count increased by an average of 65 cells in the micronutrient group versus a 6-cell decline in the placebo group at 12 weeks (P = 0.029). The absolute CD4 count increased by an average of 24% in the micronutrient group versus a 0% change in the placebo group (P = 0.01). The mean HIV-1 RNA decreased in the micronutrient supplementation group, although not significantly. Neuropathy scores improved in the micronutrient group by 42% compared with a 33% improvement in the placebo arm. This difference did not reach statistical significance. Fasting serum glucose, insulin, and lipids were not adversely affected in the patients taking the micronutrients.
Conclusions: Micronutrient supplementation can significantly improve CD4 cell count reconstitution in HIV-infected patients taking HAART. The micronutrient supplement tested was well tolerated and may hold promise as an adjuvant therapy in the treatment of HIV. Further investigation is warranted.
Introduction
In the era of highly active antiretroviral therapy (HAART), viral suppression is not always accompanied by complete immune reconstitution.1 After viral control is achieved, immune recovery is frequently slow and incomplete with an initial increase in memory CD4 cells followed by an increase in naive CD4 cells.2 The relationship between viral suppression and immune recovery is dynamic and complex and involves multiple factors,3 of which nutritional status is believed to play a pivotal role.
The relationship between immune function and nutritional supplementation is a well-described phenomenon.4-7 Numerous studies have reported a high prevalence of nutrient deficiencies early in the course of HIV infection.8-10 These deficiencies have been shown to be associated with more frequent opportunistic infections, faster disease progression, and a greater incidence of HIV-related mortality.11-15 Possible mechanisms include increased intracellular oxidative stress, enhanced viral replication, and a reduction in the number of circulating CD4 lymphocytes associated with individual or accumulated nutrient deficiencies.14-18 These mechanisms, alone or in part, may contribute to the increased morbidity, more rapid disease progression, and the higher mortality seen in HIV-infected patients with nutrient deficiencies.18-21
Several prospective, randomized clinical trials now suggest that HIV-infected patients who take micronutrient supplements have improved clinical outcomes. Fawzi et al22 showed that daily doses of a micronutrient supplement taken by HIV-infected Tanzanian women, in multiples of the recommended dietary allowances (including B-complex, vitamin C, and vitamin E), produced a significant increase in CD4 and CD8 cell counts when compared with those given placebo. In this prospective, double-blinded trial, patients taking the micronutrient supplement were also significantly less likely to die or progress to WHO stage 4 HIV disease classification.23 Jiampton and colleagues have also demonstrated that by administering a micronutrient supplement to HIV-infected men and women living in Thailand, overall HIV-associated mortality decreased by 50% compared with the placebo arm. This effect was most pronounced in patients with lower CD4 cell counts (<200 cells/μL).24
The occurrence of mitochondrial dysfunction due to drug toxicity is believed to promote increased levels of oxidative stress in a large number of cell lines. Peripheral neurons and adipocytes seem to be significantly affected.25-28 The dideoxynucleosides stavudine and didanosine have been shown to produce significant depletion of mitochondrial DNA in both of these tissues.29,30
The cohort we chose to study consisted of HIV-infected patients exhibiting neurologic toxicity from either stavudine and/or didanosine so that we might examine the effects of micronutrient supplementation on HIV disease progression and on one clinical manifestation of mitochondrial toxicity: distal symmetrical polyneuropathy (DSP).
METHODS
Study Design
This was a prospective, randomized, double-blinded, placebo-controlled clinical trial designed to determine the effect of micronutrient supplementation on HIV-1 disease progression and on DSP in HIV-infected patients taking HAART. Four study centers serving HIV-infected patients in the USA were selected. The study design and protocol were approved by an independent institutional review of the Copernicus Group Institutional Review Board (Cary, NC). Written informed consent was obtained from all study participants before enrollment.
Study Subjects
Enrollment took place between January 2002 and May 2003. Forty HIV-infected patients taking a stable HAART regimen for at least 3 months and who developed symptoms of DSP after beginning either stavudine and/or didanosine were randomized at each study site using a random block design to receive either the micronutrient supplement or an identically appearing placebo taken twice daily for 12 weeks. Patients who were pregnant, receiving treatment of an active opportunistic infection or malignancy, had vitamin B12 deficiency at screening, or were already taking more than 1 micronutrient supplement pill per day were excluded from the study. All clinical and follow-up staff were unaware of the patient treatment assignments.
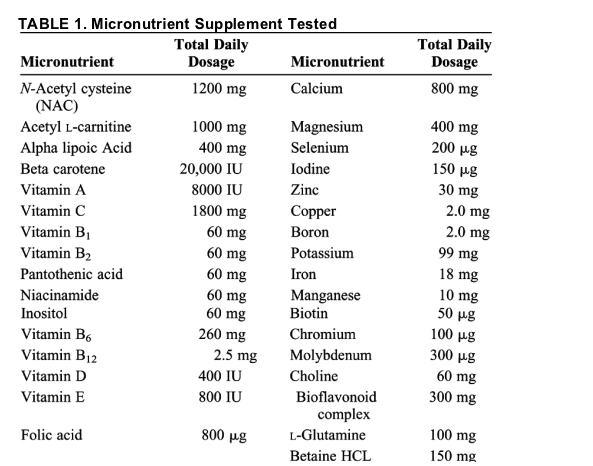
The micronutrient supplement tested included 33 ingredients (Table 1) and was consumed twice daily with food. Patients were allowed to take their antiretroviral medication at the same time as the micronutrient supplement. The micronutrient and placebo capsules were produced to our specifications by Thorne Research, Dover, ID.
Clinical and Laboratory Evaluations
Study patients visited their respective research centers for screening, which included the collection of demographic information, medication history, and laboratory tests. Eligible participants returned to the clinic for baseline and follow-up visits every 4 weeks for a total of 12 weeks. Each clinic visit consisted of the following evaluations: a laboratory-monitoring panel, a clinical assessment, and a neurological examination.
Laboratory testing was performed at baseline and every 4 weeks for a total of 12 weeks. It included a CBC, CD4 lymphocyte panel, HIV-1 RNA, fasting glucose, fasting lipids, fasting insulin, liver function tests (aspartate aminotransferase, alanine aminotransferase (ALT), total bilirubin), serum creatinine, and fasting venous lactate. The CD4 lymphocyte count was measured by conventional flow cytometry. Plasma HIV-1 RNA was measured using an ultraquantitative polymerase chain reaction assay with a lower limit of quantification to 20 copies/mL (serial diluted standards calibrated against an AIDS Clinical Trials Group reference). Fasting venous lactate levels were collected in the resting state, without the use of a tourniquet or fist clenching, and the blood sample was immediately immersed into a chilled water bath before centrifugation and freezing. All laboratory samples were shipped on dry ice to a central laboratory for processing (Immunodiagnostic Laboratories, San Leandro, CA).
The clinical assessment incorporated 2 additional self-administered patient questionnaires. The linear analogue self-assessment tool assessed the patient's energy level, ability to perform daily activities, and overall quality of life on a linear scale.
The neurological examination at baseline and follow-up was performed by a clinician using the neurological examination assessment tool. This tool consisted of the objective measurement of sensory function of the lower extremities by sharp pin and tuning fork methods and motor function of the toes and ankles by movement against resistance. Abnormal findings were rated on an increasing numerical scale (0-4) based on severity. An examination absent of abnormal findings received a zero score. A neuropathy inventory linear analog scale was also completed by the patient at each clinic visit to assess the extent of pain, numbness, and paresthesias in the feet. This tool used a visual analogue scale; each question was analyzed individually, and the mean of the 3 questions was also calculated and analyzed.
Statistical Analyses
All analyses were performed on an intent-to-treat basis. Repeated measures models were used to examine the impact of the treatment over time. This analysis was used with variables for which multiple measurements were available for each study participant to estimate the variance and the covariance for each observation, providing a mechanism to adjust for potential covariates. Baseline continuous data were compared using the Student t test and categorical data were compared using the x2 test. Fisher exact test was used in place of the x2 test where 1 or more cells had fewer than 5 members. All 2-sided tests were performed at the a = 0.05 level with no adjustment made for multiple comparisons.
The primary end point of the study was to examine the effect of micronutrient supplementation on immunologic parameters (CD4 cell count). Secondary end points were virologic and metabolic parameters (fasting glucose, fasting lipids, fasting insulin, liver function tests, serum creatinine, and venous lactate), DSP symptoms, and general health status.
RESULTS
Forty patients were randomized, 18 into the micronutrient and 22 into the placebo groups. The groups were well balanced among the 2 treatment arms except for 3 parameters; none of these baseline differences, however, was statistically significant: (1) the micronutrient group had a lower CD4 count at baseline when compared with the placebo group (CD4: 357 ± 154 cells/μL vs. 467 ± 262 cells/μL, P = 0.13), (2) the participants in the micronutrient treatment group reported a greater number of months of neuropathy symptoms preceding enrollment than those in the control group (means: 21.4 months vs. 12.2 months, P = 0.14; medians: 14.2 months vs. 2.5 months), and (3) the micronutrient treatment group contained 3 patients with diabetes mellitus compared with zero in the placebo group (P = 0.09). All study participants were taking at least 3 antiretroviral drugs, and there were no significant differences in the composition or duration of HAART between the 2 arms. A complete summary of the demographic and baseline characteristics for both groups is presented in Table 2.
Immunologic, Virologic, and Metabolic Parameters
The mean absolute CD4 count increased significantly by an average of 65 cells in the micronutrient group versus a 6-cell decline in the placebo group at 12 weeks (P = 0.029) (Table 3). The mean change in CD4 cell count from baseline to 12 weeks also increased significantly by 24% in the micronutrient group compared with no change in the placebo group (P = 0.01) (Table 3). Our analysis also shows that the effect of the micronutrient supplement was not influenced by the initial CD4 cell count.
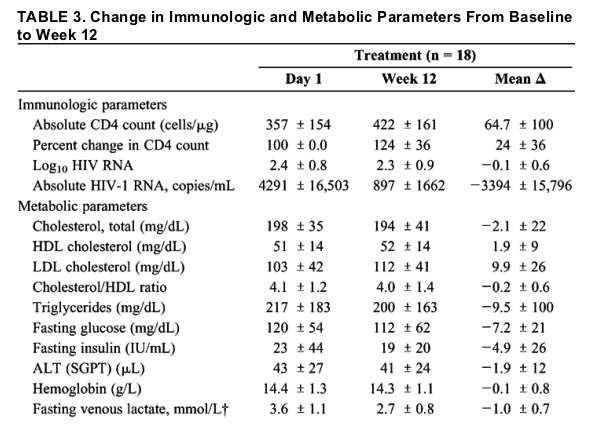
The mean absolute HIV-1 RNA decreased from 4291 to 897 copies in the treatment group compared with an increase from 2648 to 5935 copies in the placebo group. Although the changes in absolute mean HIV-1 RNA seem large, the HIV RNA Log10 decreased by 0.1 logs in the treatment group and increased by 0.1 logs in the placebo group. The difference in these responses was not statistically significant (P = 0.26).
We also measured a number of metabolic parameters often adversely influenced by HIV infection, HAART therapy, or both. To explore whether the micronutrient supplement affected these metabolic parameters, we measured fasting glucose, insulin, lipids, and venous lactates, as well as markers for kidney and liver function (serum creatinine, ALT, total bilirubin, alkaline phosphatase). There were no significant differences in these parameters when compared with placebo after 12 weeks of micronutrient supplementation.
Clinical Parameters and Adverse Events
Neuropathy scores improved in both groups, with the micronutrient group improving their overall neuropathy symptoms by 42% compared with a 33% improvement in the placebo arm. This difference between the groups did not reach statistical significance over 12 weeks.
The micronutrient supplement we tested was well tolerated. There were no reports of nausea, diarrhea, dyspepsia, or other gastrointestinal symptoms despite consuming this formula in combination with a HAART regimen.
Only 2 adverse events were reported during the study, both in the micronutrient arm and both were judged unrelated to the study treatment. One patient was hospitalized and treated with a diagnosis of bacterial pneumonia. The study supplement was suspended for 5 days and resumed upon the patient's discharge from the hospital. This patient completed the study protocol without further incident. A second patient was diagnosed with a bladder stone by cystoscopy. He did not require discontinuation of the study treatment and recovered uneventfully.
DISCUSSION
An increase in CD4 cell count after micronutrient supplementation in HIV-infected persons has previously been reported by Fawzi and colleagues,23 but this cohort was not receiving HAART. To our knowledge, this investigation is the first trial of micronutrient supplementation to show a significant immunologic benefit in patients taking a stable HAART regimen. Moreover, the robust recuperation of the CD4 cell count (a mean increase of 65 cells/μL) in the micronutrient group occurred after 12 weeks of supplementation, a relatively short period of time compared with the slower reconstitution reported by other cohorts after initiating HAART without micronutrient supplementation; Connick and colleagues2 reported a median increase of 170 cells in 48 weeks, whereas Koletar et al3 reported a much slower rate of increase of 5.9 cells/μL every 8 weeks.
The mechanism of precisely how micronutrient supplementation may increase the CD4 count is presently unknown. Although it is not possible to determine from this study which of the micronutrients or their combination is responsible for the observed immune reconstitution, several of the B vitamins and vitamins C and E have previously been shown to enhance cellular immunity.31,32 Supplementation of HIV-infected African women with these nutrients significantly improved CD4+, CD8+, and CD3+ cell counts, and this beneficial effect was sustained for a median of 5 years.23 Moreover, the supplement used in our study included 3 additional antioxidants (acetyl l-carnitine, N-acetyl-cysteine, and alpha lipoic acid). Each of these nutrients has individually been shown to produce positive effects in HIV-infected individuals.33-42
In addition, antioxidant supplementation has previously been associated with a reduction in viral load23,43 and has been observed to produce a significant decrease in CD4 cell apoptosis.34 This effect was reported in 2 studies after supplementation with the antioxidant l-carnitine in both symptomatic33 and asymptomatic HIV-infected patients.34 The mechanism of action of apoptosis in HIV-infected CD4 cells is believed to result from the effect of HIV itself and from concomitant antioxidant imbalances in host cells.44-46 A correction of these imbalances, leading to a subsequent decrease in the apoptosis rate of CD4 lymphocytes, may be partially responsible for the increases in CD4 cell counts observed in our study.
Because N-acetyl-cysteine was also included in the micronutrient supplement in this study, an additional mechanism of action could be through the replacement of sulfur in the form of N-acetyl-cysteine. Previous studies have shown that sulfur is lost on a massive scale in HIV-infected patients as a consequence of peripheral tissue cysteine catabolism, and this sulfur loss is not ameliorated by HAART.36 Supplementation of HIV-infected patients with N-acetyl-cysteine for 7 months in placebo-controlled randomized clinical trials resulted in a significant increase in immunological function35-37 and in a release of the chemokines MIP-1alpha, MIP-1beta, and RANTES, which are implicated in blocking the interaction of HIV with the CCR5 receptor38 and in lowering apoptosis.39,40 Thus, as part of this micronutrient supplement, the N-acetyl-cysteine provided may have contributed toward the observed immunological reconstitution.
Although there was no statistically significant change in the DSP symptoms in this trial, the mean 42% improvement in DSP symptoms in patients taking the micronutrients compared with a 33% improvement in the placebo arm is clinically significant. This finding is in accord with a recently published trial by Hart and colleagues,41 who showed improvement in DSP symptoms when HIV-infected patients were supplemented with oral acetyl-l-carnitine for 24 months. The improvement in DSP symptoms was accompanied by significant nerve fiber regeneration determined by immunohistochemical staining techniques.41 The 12-week duration of our study may not have been sufficient to observe a statistically significant change in neuropathy symptoms.
The micronutrient supplement we tested was well tolerated. McComsey et al47 reported in an uncontrolled clinical trial that antioxidant supplementation with vitamins C and E and N-acetyl-cysteine provided to HIV-infected patients taking HAART was associated with an adverse effect on fasting glucose levels and insulin resistance at 24 weeks. Our study found no such effect. Fasting glucose, insulin, and lipid parameters (as well as aspartate aminotransferase, ALT, and serum creatinine) were not adversely affected after administering this micronutrient formula for 12 weeks.
CONCLUSION
This study demonstrates that a micronutrient supplement administered to HIV-infected patients taking stable HAART significantly enhances CD4 lymphocyte reconstitution. Our findings support the potential for a broad-spectrum micronutrient supplement to be used as adjuvant therapy in combination with HAART to provide patients with a more robust CD4 cell rebound after initiating antiretroviral treatment. Additional research including longer-term studies addressing the mechanism of action of micronutrient supplementation is warranted.
|
|
| |
| |
|
|
|