| |
Is HIV out of control in the UK? An example of analysing patterns of HIV spreading using incidence-to-prevalence ratios
|
| |
| |
AIDS: Volume 20(14) 11 September 2006 p 1898-1901
[Research Letters]
White, Peter J; Ward, Helen; Garnett, Geoff P
Department of Infectious Disease Epidemiology, Imperial College Faculty of Medicine, London, UK.
Abstract
The incidence of diagnosed HIV in the UK is increasing. We calculated the incidence-to-prevalence ratio, IPR(t), which must exceed an 'epidemic threshold', the reciprocal of the mean infectious period, for sustained transmission to occur. For heterosexuals the IPR(t) is too low for a self-sustained UK epidemic, and incidence is driven by increasing prevalence, mostly from imported cases. For men who have sex with men the IPR(t) is around the epidemic threshold, suggesting that spread is not yet under control.
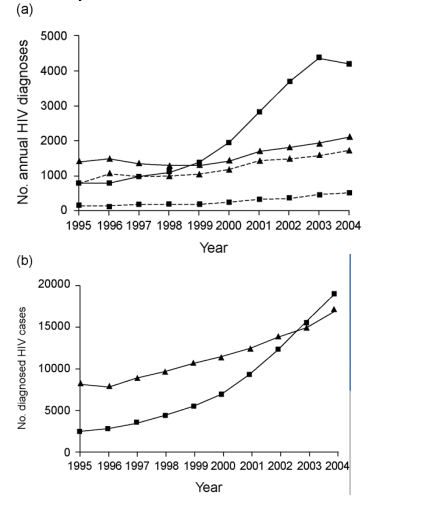
Annual new diagnoses of HIV have been increasing in the UK. New infections are mostly sexually acquired, from distinct epidemics in men who have sex with men (MSM; mostly UK acquired), and in heterosexual individuals (mostly overseas acquired) (Fig. 1a) [1,2]. Heterosexual transmission has predominated in UK-diagnosed cases since 1999, having increased rapidly since 1996 [3].
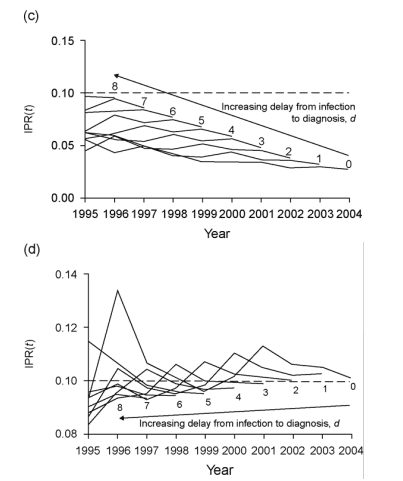
Fig. 1. Numbers of newly diagnosed and prevalent diagnosed HIV infections and incidence-to-prevalence ratios in England, Wales and Northern Ireland, 1995-2004. (a) Annual numbers of newly diagnosed HIV infections in England, Wales and Northern Ireland (EWNI) 1995-2004, acquired through heterosexual sex (squares) and sex between men (triangles). The solid lines indicates all infections, the dashed lines those acquired in the UK (place of acquisition records do not distinguish between EWNI and UK. Source: Health Protection Agency Centre for Infections. Data for 2005 were not complete at the time of the analysis. (b) Numbers of prevalent diagnosed sexually acquired HIV infections in EWNI 1995-2004, acquired through heterosexual sex (squares) and sex between men (triangles). Source: Health Protection Agency Centre for Infections. Data for 2005 were not available at the time of the analysis. (c) and (d) Annual estimates of incidence-to-prevalence ratio, IPR(t), for HIV in (c) heterosexual individuals and (d) men who have sex with men (MSM) in EWNI 1995-2004. Each line is labelled with the assumed duration in years between infection and diagnosis, d, which was varied from 0 to 8 years; the longer this duration, the further into the past is our most recent estimate. The dashed line indicates the 'epidemic threshold', TE, which IPR(t) must exceed for a self-sustained epidemic to occur. In heterosexual individuals, as d increased, IPR(t) increased, as a result of the incidence having risen relatively more rapidly than prevalence in recent years; this did not affect our key findings. In all cases IPR(t) was too low for an epidemic to occur in EWNI. The general trend was for IPR(t) to remain steady over time. For MSM, in contrast to heterosexual individuals, as d increased, IPR(t) decreased, as a result of the incidence in MSM having increased relatively more slowly than prevalence; also the effect size was smaller because the incidence and prevalence in MSM have been much more stable over time, note the different vertical scale. IPR(t) was close to TE, and at times exceeded it, except for when a long delay (d ≥ 6 years) between infection and diagnosis was assumed. The general trend was for IPR(t) to have increased slightly in earlier years, before having remained steady or even having declined in recent years.
This increase in diagnosed incidence is of course a cause for concern, but does not necessarily indicate a critical failure of HIV control policies. A growing infectious disease epidemic requires sustained onward transmission, i.e. that the mean number of new infections originating per already-infected individual, the reproductive number, R(t), exceeds 1 so that infection is amplified [4]. As R(t) applies to the 'infectious lifetime' of the average infected individual, to make an assessment at a timepoint we use the incidence-to-prevalence ratio, IPR(t), to determine whether sustained transmission has been occurring in heterosexuals or MSM in the UK.
As R(t) = IPR(t) X D, where D is the mean duration of infectiousness, the requirement for a growing epidemic, R(t) > 1, is equivalent to IPR(t) > TE, where TE is the 'epidemic threshold', equal to the reciprocal of the mean duration of infectiousness (1/D) [5]. For D = 10 years (although modern treatment has increased survival times to perhaps 20 years or more for some patients, many diagnosed infections, particularly in in-migrants, are late stage, with much shorter survival times) the 'epidemic threshold' required to be exceeded for an epidemic to occur is TE = 0.1 p.a. A comparison of incidence and prevalence was used to examine the history of the HIV epidemic in the USA [6] and Scandinavia [7].
We obtained data on annual new diagnoses in England, Wales and Northern Ireland (EWNI) of sexually acquired HIV in heterosexual individuals and MSM, including place of acquisition (cases in which this was unknown were assigned proportionately to EWNI/overseas; Fig. 1a) [8,9], and numbers of prevalent diagnosed HIV cases in EWNI (collected from 1995; Fig. 1b) [9]. Scotland (which, together with EWNI, comprises the UK) was excluded, because of a lack of data on diagnosed prevalent infections. Analysis was restricted to diagnosed cases because data on the incidence of undiagnosed infections were not available.
Analyses were performed separately for heterosexual individuals and MSM. As the delay between infection and diagnosis, d, is poorly characterized, we varied its assumed duration from 0 to 8 years. Where d > 0, newly diagnosed EWNI-acquired infections were (undiagnosed) prevalent cases in EWNI from infection to diagnosis, so we added these formerly undiagnosed infections to the diagnosed prevalent infections; thus an EWNI-acquired infection diagnosed in year y counts as an undiagnosed prevalent case at the end of years (y - d) to (y - 1). For overseas-acquired infections, we did not know the duration between arrival in EWNI and diagnosis, so we did not count their having been present in EWNI before diagnosis. We defined IPR(t) for year y as the number of new ('incident') diagnoses in year (y + d) divided by the number of people with (prevalent) diagnosed and subsequently diagnosed infections at the end of year (y - 1).
Results for heterosexual individuals are shown in Fig. 1c: IPR(t) was below the epidemic threshold (TE), indicating that a growing epidemic has not been occurring, and has remained steady or even declined slightly over time, regardless of the 'diagnosis delay' (d).
This reassuring finding is no reason for complacency. An increasing number of prevalent infections, through in-migration, transmission in the UK, and the increased survival of patients, means an increasing acquisition risk per susceptible individual. This has driven the increased incidence of EWNI-acquired infection, with IPR(t) indicating that transmission rates per prevalent infection have remained steady. Even if IPR(t) remains below the epidemic threshold, increasing prevalence will drive increasing incidence unless risk behaviour is reduced. Risk behaviour is increasing [10], along with the incidence of sexually transmitted infections (STI) [11]. As STI enhance both HIV susceptibility and HIV infectivity [12], these increases in STI incidence and risk behaviour can interact synergistically, promoting HIV transmission.
Figure 1d shows the results for MSM: IPR(t) was close to TE, sometimes exceeding it, except when a long 'diagnosis delay' (d ≥ 6 years) was assumed. The general trend was for IPR(t) to have increased slightly in earlier years, before remaining steady or even declining in more recent years.
Consistent with this, new diagnoses have increased steadily rather than explosively, explained by the increasing prevalence driving increasing incidence even when the transmission rate per prevalent infection, which IPR(t) measures, declines. Also, expanded testing may have increased detection. Control measures appear to have had partial success, with many MSM heeding health promotion messages. Nevertheless, IPR(t) is substantially higher than for heterosexuals. The spread of HIV in MSM cannot yet be said to be under control. Although the predominant route of acquisition in EWNI-diagnosed HIV cases is heterosexual sex, in EWNI-acquired cases it remains sex between men (Fig. 1a). Furthermore, as with heterosexual individuals, increasing rates of other STI, some showing considerable overlap with the HIV epidemic, are a cause for concern [2].
Data limitations required assumptions that overestimated IPR(t), biasing our analysis towards the 'detection' of an epidemic that is not occurring. The prevalence of those at risk of transmitting HIV sexually was underestimated by: (i) not counting overseas-acquired infections as prevalent cases before diagnosis in EWNI; and (ii) assuming that only those infected through heterosexual sex are at risk of transmitting it heterosexually, and making the corresponding assumption for MSM, as a result of having insufficient data to estimate the amount of transmission between MSM and heterosexual individuals, and the contribution to sexual transmission of infections acquired through another means. Having to assume that all UK-acquired infections were EWNI-acquired (records do not distinguish [8]) slightly overestimated IPR(t).
In addition to studying HIV transmission patterns in both heterosexual individuals and MSM in more detail to target interventions appropriately, enhanced surveillance is essential. Unlinked anonymous testing indicates that approximately one-third of prevalent HIV infections in the UK are currently undiagnosed [2]. The diagnosis delay needs to be minimized, for several reasons. First, to improve the individual's prognosis [13,14]. Second, to reduce transmission, through safer-sex counselling [15,16], and by reducing viral load through treatment [17,18]. Third, we need high-quality timely data to monitor the spread of HIV and intervene appropriately.
As increases in HIV prevalence and risk behaviour both increase the per-susceptible risk of infection, safer sex messages need to be reinforced. Nevertheless, the incidence of UK-acquired infection will probably continue to increase, and it is important that control policies are correctly evaluated, using IPR(t).
References
1. Dougan S, Gilbart VL, Sinka K, Evans BG. HIV infections acquired through heterosexual intercourse in the United Kingdom: findings from national surveillance. BMJ 2005; 330:1303-1304.
[Fulltext Link] [Medline Link] [CrossRef] [Context Link]
2. Health Protection Agency. The UK collaborative group for HIV and STI surveillance. Mapping the issues: HIV and other sexually transmitted infections in the United Kingdom: 2005. London: Health Protection Agency, Centre for Infections. November 2005. Available from: http://www.hpa.org.uk/publications/2005/hiv_sti_2005/default.htm . Accessed: 14 June 2006.
[Context Link]
3. Health Protection Agency Centre for Infections website. Available at: http://www.hpa.org.uk/infections/topics_az/hiv_and_sti/hiv/hiv.htm . Accessed: 14 June 2006.
[Context Link]
4. Anderson RM, May RM. Infectious diseases of humans: dynamics and control. Oxford: Oxford University Press; 1991.
[Context Link]
5. White PJ, Garnett GP. The changing risks of STIs within the UK population: a modelling analysis. In: International Society for STD Research 2003 Congress. Ottawa, Canada, 27-30 July 2003. Satellite symposium on Phase-specific Strategies for the Prevention, Control and Elimination of Sexually Transmitted Diseases II.
[Context Link]
6. Holtgrave DR. Estimation of annual HIV transmission rates in the United States, 1978-2000. J Acquir Immune Defic Syndr 2004; 35:89-92.
[Fulltext Link] [Medline Link] [CrossRef] [Context Link]
7. Amundsen EJ, Stigum H, Rottingen JA, Aalen OO. Definition and estimation of an actual reproduction number describing past infectious disease transmission: application to HIV epidemics among homosexual men in Denmark, Norway and Sweden. Epidemiol Infect 2004; 132:1139-1149.
[Fulltext Link] [Medline Link] [CrossRef] [Context Link]
8. Health Protection Agency Centre for Infections, Health Protection Scotland and Institute for Child Health. AIDS/HIV quarterly surveillance tables cumulative UK data to end March 2006; No. 70:06/1. London: Health Protection Agency Centre for Infections, April 2006. Available from: http://www.hpa.org.uk/infections/topics_az/hiv_and_sti/hiv/epidemiology/files/2006_Q1_Mar_HIV_Quarterlies.pdf . Accessed: 14 June 2006.
[Context Link]
9. Health Protection Agency, Centre for Infections. Data on annual new diagnoses of HIV infection and numbers of prevalent diagnosed infections for England, Wales and Northern Ireland. Diagnoses obtained by request, see: http://www.hpa.org.uk/infections/topics_az/hiv_and_sti/hiv/hiv.htm . Accessed: 14 June 2006.
[Context Link]
10. Johnson AM, Mercer CH, Erens B, Copas AJ, McManus S, Wellings K, et al. Sexual behaviour in Britain: partnerships, practices, and HIV risk behaviours. Lancet 2001; 358:1835-1842.
[Medline Link] [CrossRef] [Context Link]
11. Brown AE, Sadler KE, Tomkins SE, McGarrigle CA, LaMontagne DS, Goldberg D, et al. Recent trends in HIV and other STIs in the United Kingdom: data to the end of 2002. Sex Transm Infect 2004; 80:159-166.
[Fulltext Link] [Medline Link] [CrossRef] [Context Link]
12. Galvin SR, Cohen MS. The role of sexually transmitted diseases in HIV transmission. Nat Rev Microbiol 2004; 2:33-42.
[Context Link]
13. Egger M, May M, Chene G, Phillips AN, Ledergerber B, Dabis F, et al. Prognosis of HIV-1-infected patients starting highly active antiretroviral therapy: a collaborative analysis of prospective studies. Lancet 2002; 360:119-129.
[Medline Link] [CrossRef] [Context Link]
14. Sterling TR, Chaisson RE, Keruly J, Moore RD. Improved outcomes with earlier initiation of highly active antiretroviral therapy among human immunodeficiency virus-infected patients who achieve durable virologic suppression: Longer follow-up of an observational cohort study. J Infect Dis 2003; 188:1659-1665.
[Medline Link] [CrossRef] [Context Link]
15. Weinhardt LS, Carey MP, Johnson BT, Bickham NL. Effects of HIV counseling and testing on sexual risk behavior: a meta-analytic review of published research, 1985-1997. Am J Public Health 1999; 89:1397-1405.
[Medline Link] [Context Link]
16. Leaity S, Sherr L, Wells H, Evans A, Miller R, Johnson M, et al. Repeat HIV testing: high-risk behaviour or risk reduction strategy? AIDS 2000; 14:547-552.
[Fulltext Link] [Medline Link] [CrossRef] [Context Link]
17. Quinn TC, Wawer MJ, Sewankambo N, Serwadda D, Li C, Wabwire-Mangen F, et al. Viral load and heterosexual transmission of human immunodeficiency virus type 1. N Engl J Med 2000; 342:921-929.
[Medline Link] [CrossRef] [Context Link]
18. Fang C-T, Hsu H-M, Twu S-J, Chen M-Y, Chang Y-Y, Hwang J-S, et al. Decreased HIV transmission after a policy of providing free access to highly active antiretroviral therapy in Taiwan. J Infect Dis 2004; 190:879-885.
[Medline Link] [CrossRef] [Context Link]
|
|
| |
| |
|
|
|