 |
 |
 |
| |
Integrase MK-0518 Does Not Increase Lipids After 24 Weeks in Naives
|
| |
| |
Reported by Jules Levin
46th ICAAC
San Francisco, Sept 27-30, 2006
Probably the biggest news at this meeting is the results from this study: ART-naïve patients received the Merck integrase MK-0518 plus tenofovir/3TC compared to efavirenz plus tenofovir/3TC, and there were no changes in lipids for patients receiving the MK-0518 regimen.
"Differential Effects of MK-0518 and Efavirenz on Serum Lipids and Lipoproteins in Antiretroviral Therapy (ART)-naive Patients (24 Week Results)" Poster H-0256a
H. Teppler1, N. Azrolan1, J. Chen1, B.Y. Nguyen1 for the Protocol 004 Study Group2
1Merck & Co., Inc., Upper Gwynedd, PA
CONCLUSION:
MK-0518, when dosed at 100 mg to 600 mg b.i.d. in combination with TFV/3TC for 24 weeks in treatment-naive HIV infected patients:
was not associated with increases in serum cholesterol, LDL-Cholesterol, or triglycerides, which contrasts significantly with lipid changes associated with treatment with efavirenz.
ABSTRACT
Background. Some antiretroviral agents are associated with hyperlipidemia.
Objectives. To evaluate serum lipids during MK-0518 therapy.
Methods. Phase II study evaluating 4 doses of MK-0518, a novel HIV integrase inhibitor, vs efavirenz (EFV), both combined with tenofovir/lamivudine (TFV/3TC), in ART-naive HIV-infected patients (pts) with HIV-1 RNA ≥5000 copies/mL and CD4 cells ≥100 /uL. Changes from baseline in fasting serum cholesterol (SC), triglycerides (TG), LDL-C and HDL-C after 24 weeks treatment were evaluated.
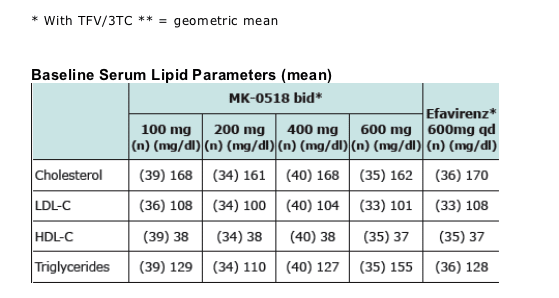
Results. Baseline lipids were similar in all treatment groups. Mean changes from baseline in serum lipids (mg/dL) at 24 weeks are:
The table immediately below shows there were no increases in cholesterol (SC) or triglycerides for patients taking MK-0518 after 24 weeks on therapy in the study. Baseline cholesterol was 161 to 170 for patients, and triglycerides were 110 to 155. In contrast patients taking efavirenz had average increase in cholesterol of +19 mg/dL and +47 mg/dl for triglycerides--
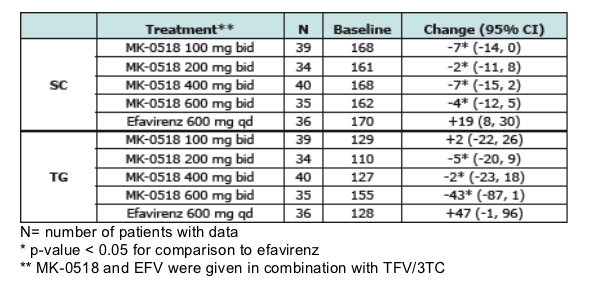
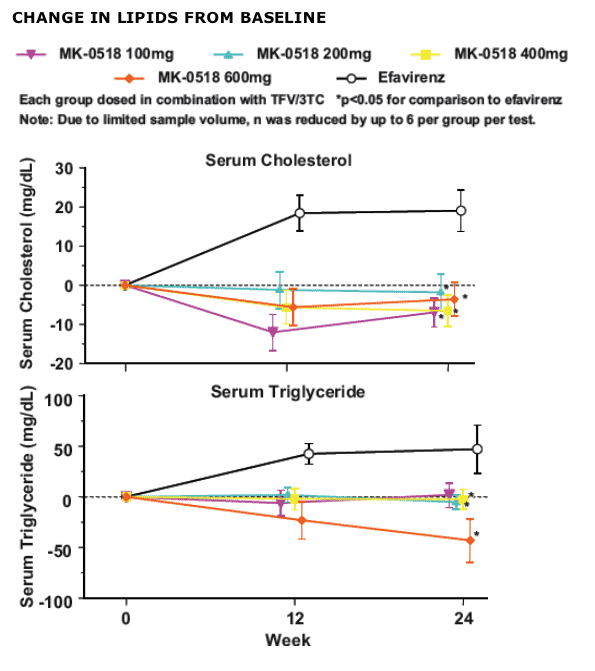
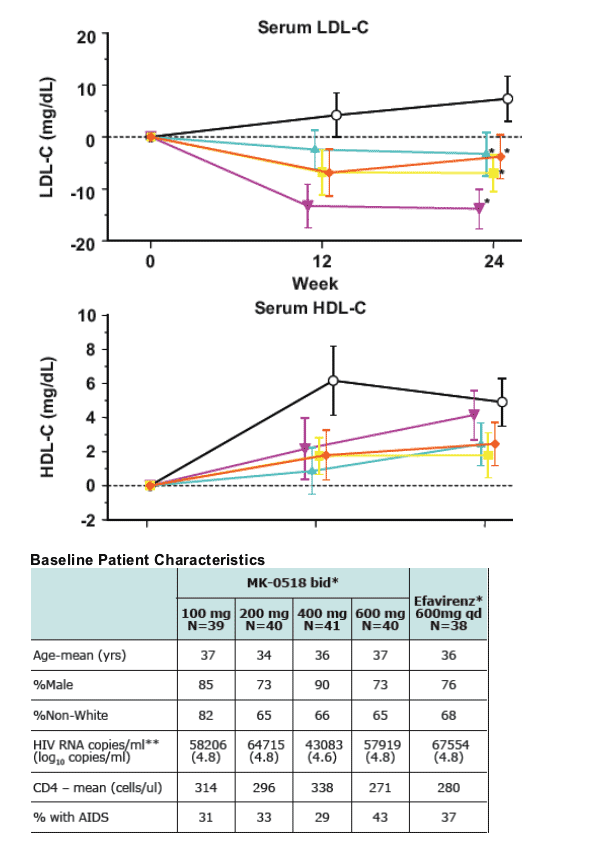
SC and TG values were relatively unchanged following MK-0518 therapy. In contrast, efavirenz treatment was associated with increases in both SC and TG. Effects on LDL-C and HDL-C will be presented.
Conclusions. MK-0518, in combination with TFV/3TC, had no adverse effects in serum lipids at 24-weeks.
BACKGROUND
Elevated serum cholesterol (SC), triglycerides (TG), low density lipoprotein cholesterol (LDL-C) and decreased hih density lipoprotein cholesterol (HDL-C) are often associated with antiretroviral therapy (ART) and may confer increased risk for cardiovascular disease and stroke (Int J STD AIDS 2005; 16 (Suppl. 1): 2-13).
MK-0518: a novel HIV integrase inhibitor
HIV integrase inhibition: a new mechanism of action
Potent antiviral activity in combination therapy
- In ART-naive patients at Week 24 (Markowitz et al, IAC 2006, Abst THLB0214)
- 85 - 95% with HIV RNA < 50 copies/mL
- In patients failing therapy with triple class resistant virus at Week 16
(Grinsztejn et al, CROI 2006, Abst 159LB)
- 56 - 72% with HIV RNA < 50 copies/mL*
Protocol 004 Study Design
Randomized, double-blind, dose - ranging study to evaluate the safety and efficacy of MK-0518 given at 100, 200, 400 or 600 mg b.i.d. versus efavirenz 600 mg q.d., both in combination with tenofovir (TFV) and lamivudine (3TC).
Key inclusion criteria:
- Susceptible to efavirenz (EFV), lamivudine (3TC) , and tenofovir (TFV)
(determined by genotype)
- No prior ART (<7 days OK)
- HIV RNA ≥ 5000 copies/mL
- CD4 ≥ 100 cells/mm3
Key Endpoints:
- HIV RNA, CD4 counts, at week 12, 24, 48
- Safety: labs and adverse experiences
Exploratory Endpoint:
Serum lipid levels at week 12, 24, 48
METHODS: All analyses were performed on fasting samples in a blinded manner by Covance, Inc. (Princeton, NJ) as part of a CDC lipid panel. Serum concentrations of total cholesterol (SC) and triglycerides (TG) and HDL-C were determined using automated enzymatic assays. HDL-C concentrations were determined after precipitation of the non-HDL fraction with dextran sulfate. LDL-C was calculated using the Friedewald formula.
12 and 24 week data available
Statistical methods
Changes from baseline in lipid levels were calculated by treatment group based upon observed data. Two-sided confidence intervals and p-values were computed using normal approximation.
|
| |
|
 |
 |
|
|