 |
 |
 |
| |
"Hidden" Mutations Contribute to Failure of Trizivir/Tenofovir
|
| |
| |
46th ICAAC, September 27-30, 2006, San Francisco
Mark Mascolini
September 28, 2006
Clonal analysis of viral populations from people in whom Trizivir plus tenofovir failed detected both pretreatment mutations and failure-related mutations not spotted by standard population sequencing [1]. This resistance analysis by GlaxoSmithKline confirmed standard-sequencing data showing that thymidine analog mutations (TAMs) largely explain failure of this regimen. But clonal analysis did uncover the tenofovir-evoked K65R mutation in 2 of 14 people with virologic failure.
The original 48-week pilot study, COL40263, tested a once-daily regimen of AZT/3TC/abacavir (Trizivir) plus tenofovir in 123 previously untreated people. Defining virologic failure as a confirmed viral load above 400 copies at study week 24 or later, the Glaxo team counted 14 failures (11%) through week 48. People in whom the regimen failed tended to have a higher pretreatment viral load and lower pretreatment CD4 count (Table). Failure proved more common than virologic response in blacks and less common than response in whites and Hispanics.
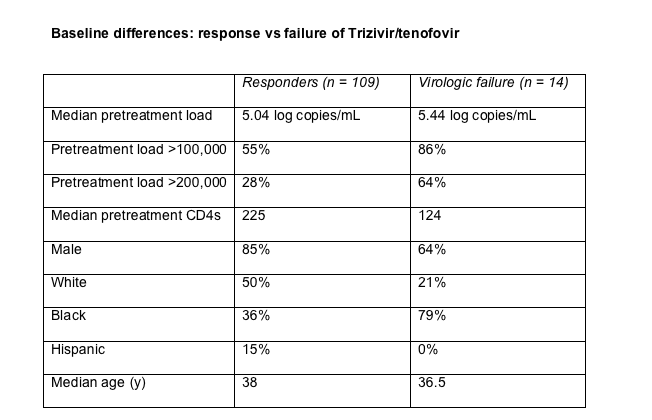
Standard population sequencing determined that 3 of 14 people in the failure group had nucleoside or nonnucleoside mutations before beginning treatment. Clonal analysis, in contrast, found pretreatment mutations in 9 of these 14. Seven of 14 people had pretreatment mutations that could make HIV resistant to one or more of the study drugs. TAMs or K65R emerged in all but 1 of these 7 people when their regimen faltered.
Four people with nonmutant ("wild-type") virus before treatment also had nonmutant virus upon failure, according to standard sequencing. But clonal analysis disclosed resistance-related mutations in 3 of these 4 upon failure: L210W (a TAM), M184I (a 3TC mutation), and Y188L (a nonnucleoside mutation not related to drugs in this regimen). In the 10 people in whom standard population sequencing detected resistance mutations upon failure, clonal analysis invariably found additional nucleoside-related mutations.
In 2 people with nonmutant virus or only the V118I substitution before treatment, both population sequencing and clonal analysis spotted the tenofovir-related K65R mutation when Trizivir/tenofovir stopped working. (The significance of the V118I change in reverse transcriptase without V44D is unknown.[2]) Treatment with standard-dose AZT typically prevents emergence of K65R, so the Glaxo team speculated that the once-daily AZT dose used in this trial may explain why K65R popped up. Another possibility, they suggested, is that using abacavir with tenofovir allowed K65R to evolve. In 1 person in whom K65R emerged, clonal analysis detected a K70E substitution before therapy. Some research suggests that K70E may favor later selection of K65R.
The Glaxo researchers observed that their clonal analysis bolsters the earlier conclusion that TAMs--and specifically T215F instead of T215Y--are the primary route to resistance among people taking Trizivir with tenofovir.
References
1. Rouse E, Preble L, Elion R, et al. Comparison of clonal genotyping and population genotyping of HIV reverse transcriptase at baseline and virologic failure in patients on once-daily Trizivir and tenofovir for <48 weeks. 46th ICAAC. September 27-30, 2006, San Francisco. Abstract H-1005.
2. Johnson VA, Brun-Vezinet F, Clotet B, et al. Update of the drug resistance mutations in HIV-1: 2005. Topics HIV Med 2005;13:51-57.
|
| |
|
 |
 |
|
|