 |
 |
 |
| |
Clinical course of increased LFTs and hepatic events associated with ritonavir (RTV)-boosted tipranavir
(TPV/r) based therapy in the RESIST trials
|
| |
| |
Reported by Jules Levin
ICAAC, Sept 27-30, 2006, San Francisco
M Sulkowski1, J Rockstroh2, V Soriano3, JO Stern4, J Mikl4
1The Johns Hopkins Univ. Sch. of Med, Baltimore, MD, USA; 2Univ. of Bonn, Bonn, Germany; 3Hosp. Carlos III, Madrid, Spain; 4Boehringer Ingelheim, Ridgefield, CT, USA
Abstract
Background: The RESIST trials showed superior efficacy of TPV/r vs comparator RTV boosted protease inhibitors (CPI/r) in treatment experienced patients (pts).
Methods:
1486 pts enrolled and at cut-off for these analyses 771 pts completed 48 wks of treatment (TPV/r = 541 and CPI/r = 230).
Results:
The population had advanced HIV disease and 10.3% (TPV/r) and 15.3% (CPI/r) of pts were co-infected with HCV or HBV. At 48 weeks, the K-M probability of
emergent Grade 3 or 4 (G3/4) ALT was more common with TPV/r (9.9%) than CPI/r (2.8%).
Cox analyses identified:
-- TPV/r treatment (HR=2.77, 95% CI 1.65-4.67),
-- Hepatitis co-infection (HR=2.01, 95% CI 1.17-3.46) and
-- CD4+ cell count > 200 (HR=1.64, 95% CI 1.07-2.53)
as significant (p<0.05) risk factors for G3/4 LFTs.
-- Elevated baseline ALT and/or AST was also a risk factor for both treatment arms (RR=2.03, 95% CI 0.88-4.70, p=0.10).
Of the 76/749 (10.1%) TPV/r pts who developed G3/4 ALT and/or AST, followup
data were available for 74 pts.
Among pts with G3/4 ALT/AST, 60/74 (81.1%) remained on study treatment.
Only 14/749 (1.9%) of TPV/r pts permanently discontinued treatment due to G3/4 ALT/AST.
The majority (85%) of patients with G3/4 ALT/AST had LFTs ∅ to ≦Grade 2 by end of follow-up.
In the TPV/r arm, 7/749 (0.9%) pts developed a hepatic SAE associated with G3/4 ALT/AST.
4/7 pts with hepatic SAEs permanently discontinued TPV/r treatment.
6/7 hepatic SAEs resolved and 1 was ongoing at last f/u.
One hepatic SAE pt not included above had advanced AIDS and HBV co-infection with isolated G3/4 ≠ bilirubin and died of renal and hepatic failure.
Conclusions: While LFT elevations were observed in the RESIST studies, only 1.9% of pts who received TPV/r discontinued therapy due to emergent G3/4 LFTs. Among these pts, hepatic SAEs were rare. Hepatic monitoring should occur frequently throughout the course of therapy with TPV/r. The majority of pts (81.1%) who developed G3/4 LFTs were able to continue therapy.
Note: The analyses have been updated to reflect the completion of 48 weeks treatment instead of 24 weeks.
Introduction
Tipranavir (TPV, Aptivus®) is a new generation protease inhibitor (PI) with potent activity against multiple PI-resistant HIV-1. TPV/r is effective and well tolerated in PI-experienced patients [1-5].
RESIST 1 and 2 are randomized, ongoing, open label, comparative Phase III studies of TPV/r (500/200 mg BID) [2-5]. RESIST 1 is taking place in N America and Australia; RESIST 2 is being conducted in Europe and Latin America. The RESIST studies enrolled triple antiretroviral (ARV) class experienced patients, who were followed for at least 48 weeks. Patients were randomized to receive an optimized background regimen (OBR) plus TPV/r or a standard of care, ritonavir boosted, comparator PI (CPI/r).
Cases of elevated serum liver transaminases levels and hepatic serious adverse events associated with TPV/r treatment during the RESIST studies were evaluated.
Study design and patients
RESIST 1 and 2 enrolled male and female patients with HIV infection who fulfilled the following entry criteria:
- ≥18 years old
- ≥3 consecutive months' experience with 3 classes of ARVs (NRTIs, NNRTIs, PIs)
- ≥2 PI-based regimens for ≥3 months; one of which was the current treatment regimen
- Any CD4+ cell count
- Viral load (VL) ≥1000 copies/mL
- HCV and HBV co-infected patients were eligible
- Baseline serum ALT and AST levels < Grade 2*
(*Grade 2 ALT or AST defined as >2.5 - 5 x the upper limit of normal)
- Viral isolate containing ≥1 primary protease mutation at 30, 33, 46, 48, 50, 82, 84, 90
- Viral isolate carrying ≦2 mutations at codons 33, 82, 84, 90
The investigators selected the CPI/r (lopinavir/r, indinavir/r, saquinavir/r or amprenavir/r) and the OBR prior to randomization. The use of enfuvirtide (ENF) was allowed; patients were stratified by ENF usage, as well as by the CPI/r.
Patients received 500/200 mg TPV/r or standard doses of the CPI/r plus approved doses of the components of the OBR.
After 8 weeks, patients who failed virologically in the CPI/r arm were able to receive TPV/r prior to licensing via a long term safety study, provided that there was documented evidence that they had been adherent to their study medication.
Results
1486 patients received TPV/r or CPI/r plus OBR. Baseline characteristics were similar in both studies and both treatment arms (Table 1).
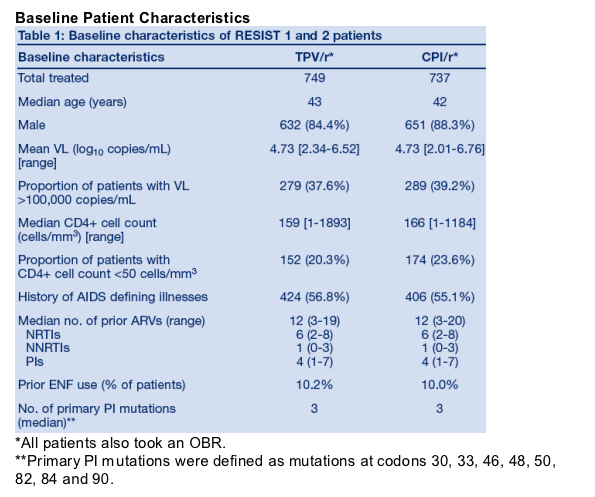
Extent of exposure to study medication differed considerably between the two
treatment arms because more patients in the CPI/r arm discontinued study medication due to virologic failure than those in the TPV/r arm. Median exposure was 384 days in the TPV/r arm (738 patient exposure years [PEYs]) and 173 days in the CPI/r arm (459 PEYs) at the time of the Week 48 analysis.
Transaminase elevations
Incidence of Grade 3 or greater transaminase elevations
Table 2 presents the number of RESIST patients with Grade 3/4 ALT or AST levels, adjusted for exposure to study medication. In the TPV/r arm, 76 patients developed a Grade 3/4 ALT and/or AST. Of these 76 pts, 71 had Grade 3/4 ALT and 45 had grade 3/4 AST. Only 5 patients had Grade 3/4 AST without associated Grade 3/4 ALT. The rates of Grade 3/4 ALT or AST were approximately two to three fold higher in the TPV/r arm than in the CPI/r arm.
The Kaplan-Meier probability of developing a Grade 3/4 ALT at 48 weeks was 9.9% and 2.8% for TPV/r and CPI/r arms, respectively (Figure 1); and 6.1% and 2.1% for Grade 3/4 AST in the TPV/r and CPI/r arms, respectively.
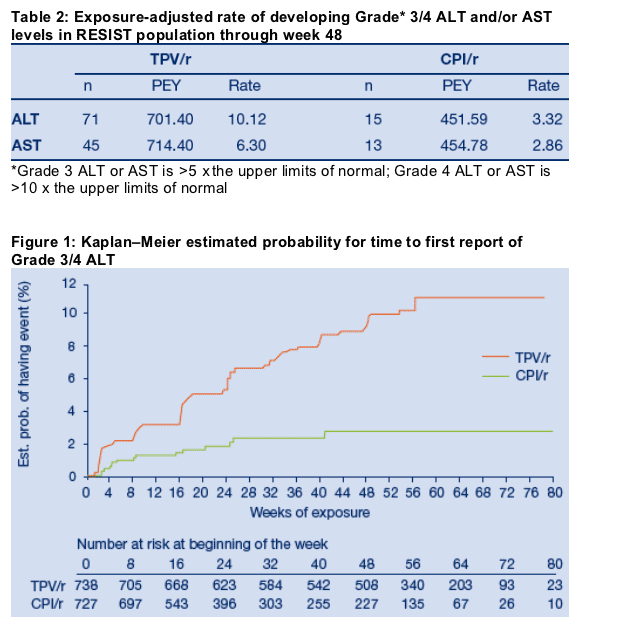
Risk Factors
Table 3 lists potential risk factors for the occurrence of Grade 3/4 ALT and/or AST (ALT/AST) elevations during week 48 of the RESIST studies. Cox multivariate regression analyses that included treatment, baseline grade of ALT/AST, viral hepatitis co-infection, baseline CD4+ cell count, race and gender, showed that TPV/r treatment (HR=2.77, 95% CI 1.65-4.67), hepatitis co-infection (HR=2.01, 95% CI 1.17-3.46) and baseline CD4+ >200 cells/mm3 (HR=1.64, 95% CI 1.07-2.53) were the most significant independent risk factors for development of Grade 3/4 ALT/AST (Table 3). Elevated baseline ALT/AST were also a risk factor for both treatment arms (RR=2.03, 95% CI 0.88-4.70) but did not reach significance (p=0.10).
Gender (HR=1.16, 95% CI 0.63-2.15, p=0.636) and race (HR=1.32, 95% CI 0.66-2.65, p=0.431) were not risk factors.
Median TPV/r trough levels between 6.5-120 m M (the range attained by >95% of all TPV/r RESIST patients) were not associated with development of Grade 3-4 ALT/AST.
Actions and outcomes associated with Grade 3/4 ALT
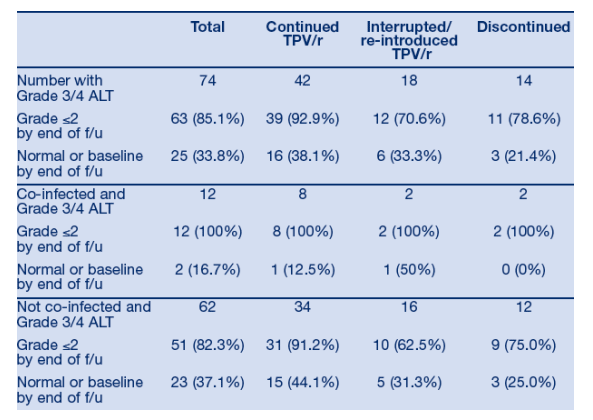
During the first 48 weeks of RESIST, 76/749 (10.1%) TPV/r patients developed Grade 3/4 ALT and/or AST elevation. For these patients, investigators had the option to continue TPV/r, interrupt and re-introduce TPV/r, or discontinue TPV/r permanently. Follow-up data were available for 74/76 patients at the time of database lock. Table 4 summarizes the actions taken and the outcomes after the first occurrence of a Grade 3/4 ALT/AST elevation. Only 14/749 (1.9%) of TPV/r patients permanently discontinued treatment due to G3/4 ALT/AST, while 60/74 (81.1%) patients with G3/4 ALT/AST were able to continue or interrupt/re-introduce TPV/r therapy (Figure 2). The majority of patients (92.9%) with G3/4 ALT/AST who continued TPV/r and the majority of patients (78.6%) with G3/4 ALT/AST who discontinued TPV/r had improvement to Grade 2 by end of follow-up. G3/4 ALT/AST elevations resolved to Grade 2 in 51/60 patients (85%) whom investigators selected to continue or temporarily interrupt therapy. Hepatitis co-infection status did not appear to influence the investigators' actions in TPV/r patients who developed G3/4 ALT/AST; and all co-infected patients who developed Grade 3/4 ALT had improvement to Grade 2 by end of follow-up (Table 4).
Hepatic SAEs and outcomes
In the TPV/r arm, 7/749 (0.9%) patients developed a hepatic serious adverse event (SAE) associated with G3/4 ALT/AST; treatment was permanently discontinued in 4 of these patients. Overall, 6/7 hepatic SAEs resolved. One patient with a hepatic SAE has had recurring G3/4 ALT/AST as of last follow up. One additional patient with a hepatic SAE not included above had an isolated G3/4 increase in bilirubin levels without G3/4 ALT/AST and died with advanced AIDS and HBV co-infection associated with renal and hepatic failure.
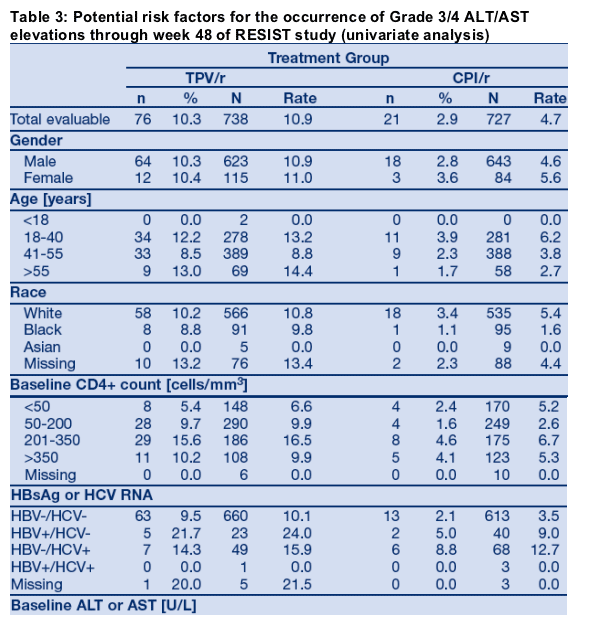
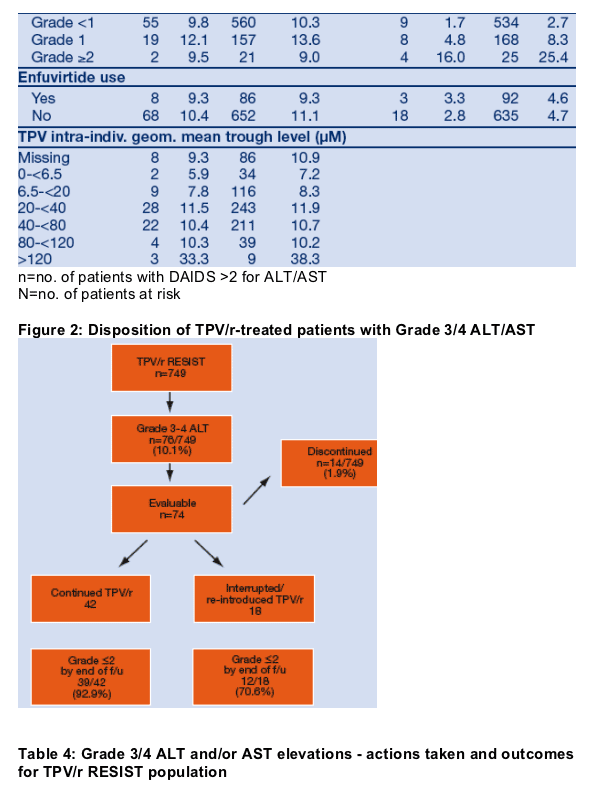
Summary
While liver events were observed with TPV/r exposure, the majority (89.9%) of patients treated with TPV/r did not develop Grade 3/4 ALT/AST elevations.
Hepatic serious adverse events associated with Grade 3/4 ALT/AST were infrequent (0.9%) through the first 48 weeks of TPV/r treatment.
The frequency of Grade 3/4 ALT/AST, adjusted for exposure to study medication, was compared for the TPV/r and CPI/r arms. The Kaplan-Meier probability of Grade 3/4 ALT at week 48 was 9.9% and 2.8% for TPV/r and CPI/r arms, respectively; and 6.1% and 2.1% for Grade 3/4 AST in the TPV/r and CPI/r arms, respectively.
The most significant risk factors for Grade 3 or higher ALT/AST elevation in the RESIST population were hepatitis co-infection and TPV/r use.
The majority of patients with Grade 3/4 ALT/AST were able to continue or to temporarily interrupt TPV/r therapy. Continuation or interruption/re-introduction strategies allowed 60/74 patients (81.1%) to continue TPV/r therapy. G3/4 ALT/AST elevations resolved to Grade ≦2 in 51/60 patients (85%) whom investigators selected to continue or temporarily interrupt therapy.
References
1. Markowitz M, et al. Safety and efficacy of tipranavir, a non-peptidic protease inhibitor, in multiple PI-failure patients (BI 1182.2). In 1st IAS Conference on HIV Pathogenesis and Treatment. 2001. Buenos Aires. Abstract 3.
2. Cahn P and the RESIST 2 Study Team. 24-week data from RESIST-2: Phase 3 study of the efficacy and safety of background therapy plus tipranavir/ritonavir (TPV/r) or optimized ritonavir-boosted standard-of-care (SOC) comparator PI (CPI) in a large randomized multicenter trial in treatment-experienced HIV+ patients. in 7th International Congress on Drug Therapy in HIV Infection. 2004. Glasgow, UK. Abs. PL14.3.
3. Hicks C and the RESIST 1 Study Team. RESIST-1: a phase 3, randomized, controlled, open label, multicenter trial comparing tipranavir/ritonavir (TPV/r) to an optimized comparator protease inhibitor/r (CPI/r) regimen in antiretroviral (ARV) experienced patients: 24 week data. In 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). 2004. Washington. Abs. H-1137a.
4. Cahn P, Hicks C, and the RESIST Study Teams. RESIST-1 (R-1) and RESIST-2 (R-2) 48 week meta-analyses demonstrate superiority of protease inhibitor (PI) tipranavir+ritonavir (TPV/r) over an optimised comparator PI (CPI/r) in antiretroviral (ARV) experienced patients. In 10th European AIDS Conference (EACS). 2005. Dublin, Ireland. Abs. PS3/8.
5. Hicks CB, et al. Durable efficacy of tipranavir-ritonavir in combination with an optimised background regimen of antiretroviral drugs for treatment-experienced HIV-1-infected patients at 48 weeks in the Randomized Evaluation of Strategic Intervention in multi-drug reSistant patients with Tipranavir (RESIST) studies: an analysis of combined data from two randomised open-label trials. Lancet, 2006. 368(9534): p. 466-75.
Acknowledgements
Thanks to all of the patients who took part in RESIST 1 and 2, as well as the investigators at each of the study sites. Boehringer Ingelheim International GmbH sponsored the RESIST studies.
|
| |
|
 |
 |
|
|