 |
 |
 |
| |
Age, Depression, and CD4 Gains
|
| |
| |
46th ICAAC, September 27-30, 2006, San Francisco
Mark Mascolini
In developed countries the rolls of 50-and-older people with HIV infection continue to grow. By one estimate 15% of all HIV-infected people in the US have passed the half-century mark. The success of antiretroviral therapy largely explains this graying of HIV cohorts in Europe, Australia, and parts of the Americas. But as two Spanish studies at ICAAC found, plenty of people over 50 remain sexually active and so risk (and get) HIV infection. Do older people respond differently to antiretrovirals? How does depression factor into the equation? And what happens when CD4 gains peter out early--or even after a half-decade of antiretroviral therapy? Several intriguing ICAAC studies addressed these questions.
Mortality predictors according to age at first visit
A 4409-person Spanish study found that people older than 49 had a higher risk of death than people under 41 if they had AIDS when they first sought care for HIV [1]. On the other hand, a baseline CD4 count under 200 independently predicted death in the younger group but not in the older group.
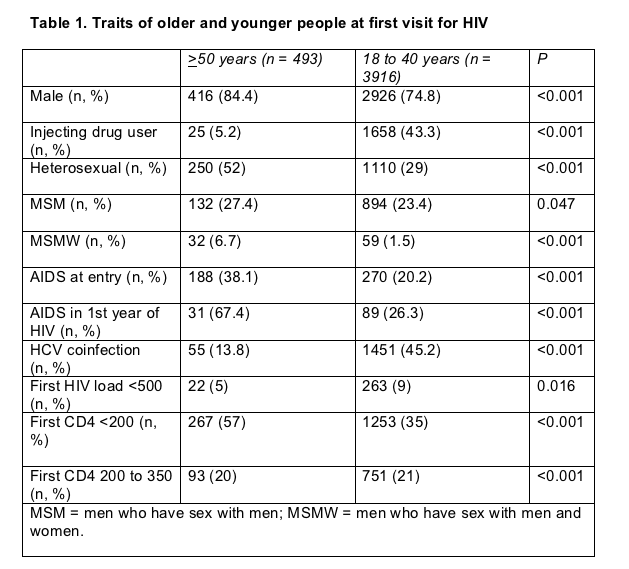
Gemma Navarro (Corporacio Parc Tauli, Barcelona) and colleagues from 10 Catalonian hospitals and one in the Balearic Islands logged clinical traits and tracked progression in 493 people first seen for HIV infection when 50 or older and in 3916 first seen when 18 to 40 years old. These people entered the cohort from January 1998 through December 2004 and follow-up averaged 33 months (range 0 to 65 months).
At their first visit for HIV infection, the older group had a higher proportion of men, a higher proportion who got infected during sex rather than injecting drugs, higher proportions with AIDS at the first visit or during the first year of infection, a higher proportion with a first CD4 count under 200, and a lower proportion coinfected with hepatitis C virus (HCV) (Table 1).
The older group began follow-up with a lower CD4 count than the younger group (about 225 versus 360 cells), but they gained significantly more CD4s over the course of the study (254 versus 186 cells). Yet as Abbott work described below also showed, CD4 gains leveled off after several years of therapy, and the older group never caught up with the younger group in absolute CD4 counts (about 450 versus 550 at last follow-up).
On the other hand, although the older group started follow-up with a higher viral load, their collective load leveled off at a lower reading than the younger group's (about 200 versus 300 copies/mL). This small viral load difference did not improve survival in the older people. At the end of the study all-cause mortality stood at 9% in the older group and 4% in the younger.
Multivariate analysis determined that AIDS at first visit raised the risk of death 4.7 times (95% confidence interval [CI] 2.4 to 9.4) in the older group (P = 0.001) and 1.8 times in the younger contingent (95% CI 1.1 to 3.0) (P = 0.017). A baseline CD4 count under 200 raised the death risk 3.7 times in the younger group (95% CI 1.9 to 7.0) and 3.6 times in the older people (95% CI 0.7 to 17), but the higher risk reached statistical significance only in the younger group (P = 0.001).
No response difference with 65-year cutoff
A single-center study at Madrid's Carlos III hospital used a higher age breakpoint than the just-discussed multicenter Spanish study, 65 years, to gauge response to antiretroviral therapy, HIV disease progression, hospital admission, and mortality in a retrospective analysis of 112 people 55 years old or older when they started treatment [2]. The 82 people under 65 did no better or worse than their 30 older counterparts by any of these measures.
The median age of the younger group was 57.8 years, compared with 69.4 years in the older group. Overall, 93 people (84%) picked up HIV infection during sex, a telling statistic that should raise awareness among clinicians seeing older patients. While 46 people (47%) had a CD4 count under 200 upon diagnosis of their infection, 52 (48%) had an opportunistic infection.
After a median follow-up of 41 months, Fatima Branas and colleagues recorded a 9% incidence of virologic failure, a new CDC class B event rate of 2%, a new class C event rate of 4%, and mortality of 2%--with no differences between the people under 65 and those 65 or older. Nor did the groups differ in proportions reaching an undetectable viral load or enduring virologic failure, or in the number of hospital admissions. Through 48 weeks of follow-up, the older people did gain CD4 cells more slowly than the younger people (r2 = 0.5698 versus 0.8389 for CD4 slope), but this difference lacked statistical significance.
Older age, lower starting CD4s predict blunted CD4 recovery
Another single-center Spanish study, this one at Madrid's Gregorio Maranon Hospital, found that a lower pretreatment CD4 count and older age when therapy began predicted lower absolute CD4 counts after 6, 12, and 18 months of therapy [3]. Older age also lowered the likelihood of getting the viral load under 50 copies in this analysis of 187 treatment-naive people who began a PI or an NNRTI regimen with a CD4 count under 200 from 1996 through 2004.
The study group included 155 men (83%) with a median age of 39.5 years when they started antiretrovirals. A higher proportion, 46%, got infected with HIV during sex (25% heterosexual, 25% homosexual), while 43% picked up HIV when injecting drugs. The big injecting population may largely explain the 42% prevalence of HCV coinfection. Most cohort members (62%) had CDC category C HIV infection. Pretreatment CD4 count stood at 56 and pretreatment viral load at 5.28 log.
Among people who completed 6 months of treatment, Juan Berenguer and coworkers reported that 75% reached a viral load below 50 copies, compared with 66% who completed at least 12 months and 58% who finished at least 18 months. Multivariate statistical analysis adjusted for pretreatment CD4 count, viral load, CDC category C, HCV coinfection, and PI therapy determined that every extra 10 years of age lowered the chance of getting under 50 copies 16% (adjusted hazard ratio [AHR] 0.84, 95% CI 0.72 to 0.95). It took HCV-coinfected people 69% longer to get under 50 copies (AHR 1.69, 95% CI 1.23 to 2.32).
While 96% of the study group notched at least one CD4 count above 100, 78% had at least one above 200, 62% at least one above 300, 44% at least one above 400, and 57% at least one above 500. Pretreatment CD4 count proved the strongest predictor of reaching a count above 350 or above 200. Each additional 50 CD4s before treatment boosted the chance of getting above 350 by 30% (AHR 1.30, 95% CI 1.09 to 1.55) and the chance of getting above 200 by 39% (AHR 1.39, 95% CI 1.19 to 1.61).
But older age also proved a crucial factor in CD4 recovery. Every additional 10 years of age when treatment began made it 4.6 times less like to have a 6-month count above 500, 3.2 times less likely to have a 12-month count above 500, and 3.4 times less likely to have an 18-month count that high. And every extra decade of age made it 1.9 times less likely to reach a count above 350 during the 18 months after starting therapy.
The effect of baseline CD4 count on absolute count during follow-up points in the same direction as findings from a longer Abbott study described below under "Lower pretreatment CD4s".
Depression and adherence in older people
Depression in HIV-infected people over 50 predicted worse adherence to antiretroviral therapy and poor control of HIV replication, according to results of a 121-person study in the United States [4]. Oluwatoyin Adeyemi and colleagues at Chicago's Stroger Hospital proposed that the simple depression scale they used--the Center for Epidemiology Study of Depression (CESD) score--can help HIV clinicians identify and manage depression. The CESD is a validated 20-item self-report scale [5].
The study involved 121 HIV-infected people 50 years old or older enrolled in the CORE50 cohort from May 2005 through February 2006. Median age stood at 54 years (range 50 to 66 years), 79% were men, 83% African American, 9% Hispanic, and 6% white. Most cohort members, 53%, had become infected while injecting drugs, and 57% had HCV infection. Median duration of HIV infection measured 12 years (range 1 to 23), 84% were taken antiretrovirals, and 67% had a viral load below 50 copies.
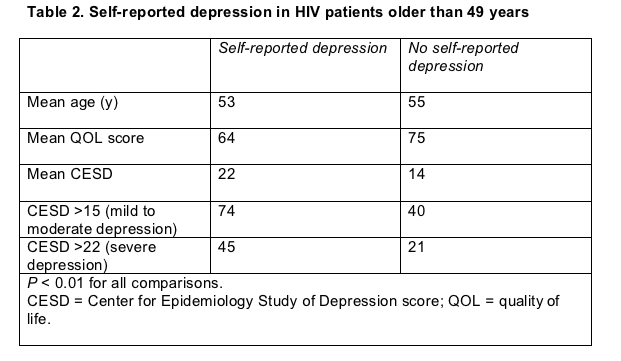
While 58% of the cohort had a CESD score above 15 (signaling mild to moderate depression), 35% had a CESD above 22 (signaling severe depression). The group's collective CESD averaged 18. Just over half of the cohort had self-reported depression, and that group had significantly higher CESDs but lower age and quality of life (Table 2).
People with a CESD above 22 proved significantly more likely to smoke (83% versus 51%, P = 0.05) and to have missed at least one antiretroviral dose in the last week (62% versus 31%, P = 0.01). CORE50 members who missed a dose in the past week had a significantly higher CESD (25.6 versus 17, P = 0.00). In a finding reflecting these adherence scores, people with a viral load under 50 copies had a significantly lower CESD (16.3 versus 20.8, P = 0.03). Tellingly, CESD score--but not self-reported depression--was an independent predictor of a sub-50-copy viral load. Only 57% of people with self-reported depression were taking antidepressants.
Adeyemi and coworkers proposed that HIV-infected people taking antidepressants "should be screened regularly by a standardized tool like CESD to assess the efficacy of therapy because we found high CESD scores even in patients on antidepressants."
Lower pretreatment CD4s limit CD4 recovery
How long do CD4 counts climb after successful antiretroviral therapy starts, and how do pretreatment counts affect that upswing? Martin King and Abbott colleagues tackled those questions in the longest analysis of this type so far, a 7-year follow-up of treatment-naive people starting lopinavir/ritonavir plus d4T and 3TC in a clinical trial [6]. CD4 gains appear to flatten out after about 300 weeks of treatment, this 100-person study showed. Plateaus settled in the normal range among people who began treatment with more than 200 CD4s, but T-cell gains stalled at 500 to 600 among people who started with lower counts.
While 64 people in this study group began their PIs with more than 200 CD4s, 17 started with counts under 50 and 19 with counts between 50 and 199. Seven years later the sub-50-CD4 starting group gained an average 532 cells, the 50-to-199 group gained an average 476, and the 200-plus group gained an average 495. Although people with lower pretreatment counts continued to add CD4 cells longer, they never caught up with people who began lopinavir/ritonavir with more than 200 CD4s. Counts hit a plateau around 500 to 600 cells in the sub-200 starting group and around 900 in the 200-plus starting group.
King and colleagues devised a mathematical model to describe CD4 climbs over time and plugged in data from these 100 people. This model determined that people who began lopinavir/ritonavir with d4T/3TC when they had fewer than 50 CD4s reached a plateau at treatment week 371 with 588 CD4s, people who began with 50 to 199 CD4s hit their plateau at treatment week 336 with 633 CD4s, and people who began with 200 or more CD4s reached their plateau at week 312 with 957 cells (Table 3). Analyses based on CD4 change from baseline and CD4 changes only in people who completed the 7-year study found similar plateau times.

Model 1 involves absolute CD4 counts in 100 people, including last-visit data from some people who did not complete 7 years of follow-up. Model 2 relies on changes in CD4 count instead of absolute CD4 counts. Model 3 involves only people who finished the study instead of also incorporating last-visit data from those who did not.
The Abbott results reflect findings from a Dutch ATHENA Cohort analysis [7]. Looking at people who began successful combination therapy and had more than 5 years of follow-up, the ATHENA team found that those who started with fewer than 50 CD4s gained an average 499 cells, those who started with 50 to 199 CD4s gained an average 429, and those who started with 200 or more CD4s gained an average 412.
The ATHENA findings appear to confirm a link between CD4 jumps and starting CD4 count regardless of which potent regimen a person takes. But no one can say whether the plateaus predicted by Abbott's model apply to nonlopinavir regimens for several reasons. First, and most obviously, Abbott's modeling results involve only people taking lopinavir/ritonavir with two NRTIs. But there are other reasons. An earlier EuroSIDA study found that people taking a lopinavir regimen added more CD4s every year than those taking other ritonavir-boosted PIs [8]. And AIDS Clinical Trials Group study A5142 found significantly higher 96-week CD4 counts in previously untreated people randomized to lopinavir/ritonavir plus two nucleosides than in people starting efavirenz and two nucleosides [9].
King and colleagues did not address an even bigger question--why CD4 gains peter out at a lower level among people who start antiretrovirals with fewer T cells.
What happens when CD4s stay under 200?
People in the just-reviewed Abbott study [6] who started their first antiretrovirals with a CD4 count under 200 stopped at a lower CD4 plateau than those who started at higher counts. But most of these late starters topped out with a generally safe 500 to 600 CD4s. What happens, though, when people get a great virologic response from therapy but never push their CD4s past 200?
That's the question asked by Mona Loutfy (University of Toronto) and colleagues in Vancouver [10]. To find out they compared clinical trends in 97 people whose CD4 gains stagnated under 200 and in 202 who reached a higher count after 1 year of treatment. The cohort consisted of adult British Columbians who began triple therapy between August 1996 and September 2003 and registered a viral load below 50 at least twice in a row in the first year of treatment. Follow-up continued until September 2004 or until the viral load topped 50 copies or the person had a new AIDS diagnosis or died.
The sub-200 CD4 group had a median pretreatment count of 40 (interquartile range [IQR] 10 to 80), while the 200-plus group began treatment with a median count of 120 (IQR 50 to 160), a highly significant difference (P < 0.001). But pretreatment viral loads did not differ significantly between the two groups; both had starting medians above 100,000 copies. The groups were balanced in age, gender, proportion with AIDS, and injecting drug use. But a lower proportion in the sub-200 group than in the control group began treatment with a nonnucleoside (40% versus 53%, P = 0.032).
After 1 year of therapy overall clinical event rates did not differ greatly between people with a CD4 count still below 200 (10.3%) and those whose CD4s climbed higher (8.4%). Adherence proved the overwhelming factor when Loutfy reckoned clinical event risk after 1 year of therapy in a univariate Cox regression analysis. Better than 95% adherence figured by pharmacy refills lowered the risk of progression 83% (hazard ratio 0.17, 95% CI 0.04 to 0.62, P = 0.008). This analysis also saw a trend toward a four times higher progression risk in people whose CD4s languished below 200 (hazard ratio 3.94, 95% CI 0.85 to 18.2, P = 0.08). And the clinical setbacks recorded in this cohort were bad ones--including 6 deaths, 6 mycobacterial infections, 5 Kaposi sarcomas, 3 cases of dementia or encephalopathy, and 2 cases each of lymphoma, Pneumocystis pneumonia, and multifocal leukoencephalopathy. Longer follow-up with a larger cohort could tip the progression difference between these two groups into statistical significance.
Mark Mascolini writes about HIV infection (markmascolini@earthlink.net).
References
1. Navarro G, Nogueras MM, Segura F, et al. HIV infection in older patients: clinical and epidemiological characteristics and response to combined antiretroviral therapy. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1397.
2. Branas F, Berenguer J, Sanchez Conde M, et al. Effectiveness of HAART in the elderly. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1394.
3. Resino R, Berenguer J, Bellon J, et al. Influence of age in HIV-infection outcomes among antiretroviral-naive patients with severe immunodeficiency during highly active antiretroviral therapy. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1399.
4. Adeyemi OM, Thomas-Gossain N, Lee M, et al. Depression in older HIV+ patients: data from the CORE50 cohort. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1395.
5. Radloff LS. The CES-D scale: a self-report depression scale for research in general populations. Appl Psychol Meas 1997;1:385-340.
6. King M, Da Silva BA, McMillan F, et al. When does the CD4+ cell count plateau? Evidence from subjects treated with a lopinavir/ritonavir-based regimen for up to 7 years. 46th ICAAC. September 27-30, 2006, San Francisco. Abstract H-1401.
7. Gras L, Van Sighem A, Frase C, et al. Predictors for changes in CD4 cell count 7 years after starting HAART. 13th Conference on Retroviruses and Opportunistic Infections. February 5-8, 2006. Denver. Abstract 530.
8. Mocroft A, Phillips A, Ledergerber B, et al. Relationship between antiretrovirals used as part of a cART regimen and CD4 cell count increases in patients with suppressed viremia. AIDS 2006;20:1141-1150.
9. Riddler SA, Haubrich R, DiRienzo G, et al. A prospective, randomized, phase III trial of NRTI-, PI-, and NNRTI-sparing regimens for initial treatment of HIV infection--ACTG 5142. XVI International AIDS Conference. August 13-18, 2006. Toronto. Abstract THLB0204.
10. Loutfy MR, Yip B, Moore D, et al. Increased clinical events in HIV-infected patients who achieve full virologic suppression but fail to attain a CD4 count >200 cells/mm3 after one year of combination antiretroviral therapy. September 27-30, 2006, San Francisco. Abstract H-1403.
|
| |
|
 |
 |
|
|