 |
 |
 |
| |
Complications of Three Simplifying Strategies
|
| |
| |
46th ICAAC, September 27-30, 2006, San Francisco
Mark Mascolini
Attempts to build a better mousetrap, virologically speaking, have driven many antiretroviral strategy trials over the past decade. Results show, sadly, that prematurely sprung traps can catch the baiters' fingers (and the baiters' patients) as often as they trap the wily retrovirus. Most trials of simple triple-nucleoside combos and structured treatment interruptions, for example, have stung study participants more readily than they've stunned HIV. Depending on how one interprets the results, three trials reviewed at ICAAC may perpetuate this tradition.
Spare the nukes and spoil the virus?
The allure of first-line nucleoside (NRTI)-sparing regimens rests mainly on the wish to avoid short- and long-term side effects of these agents. But that aim must be weighed against the combined toxicities of the antiretrovirals used (protease inhibitors [PIs] and nonnucleosides [NNRTIs] for now)--as well as against the potency of a PI/NNRTI coupling (or boosted PI monotherapy) and the consequent risk of resistance.
A small US multicenter trial comparing two NRTI-sparing combinations--atazanavir/ritonavir/efavirenz at 300/100/600 mg and 400/100/600 mg once daily--found equivalent 48-week response rates just above 60% in both arms [1]. Triglycerides climbed dramatically in the first 8 weeks with both regimens, then leveled off.
One hesitates to read too much into the virologic results because only 61 people got treated in this pilot trial, which included no nucleoside-containing control arm. Fifty-two study participants (85%) were men, and 30 of them (49%) were white. None had antiretroviral experience, and all had a viral load above 1000 copies and a CD4 count above 50. Median pretreatment load stood at 4.97 log copies/mL (just under 100,000 copies) and mean CD4 count at 305.
Gary Thal of Bristol-Myers Squibb, maker of atazanavir and efavirenz (and two less popular NRTIs) explained that the trial tested a 400/100-mg atazanavir/ritonavir dose because efavirenz lowers atazanavir exposure with the standard 300/100-mg dose to levels seen with unboosted atazanavir. But Thal and colleagues did not collect drug-level data during the trial, so he couldn't say whether the higher atazanavir dose yielded higher exposure than the lower dose. The higher dose did not appear to improve response.
Five of 31 people (16%) quit the 300/100-mg arm and 7 of 30 (21%) dropped out of the 400/100-mg arm before week 48, but only 1 of them (in the higher-dose arm) because of side effects. At week 48 both intent-to-treat and on-treatment analyses showed equivalent virologic responses with the two doses (Table 1). The sub-50-copy intent-to-treat response rates--63% with 300/100 mg and 61% with 400/100 mg--do not measure up to responses in some recent trials of standard regimens including NRTIs. Cross-study comparisons, though widely despised, are irresistibly intriguing and perhaps mildly informative. For example, 48-week intent-to-treat sub-50 rates were 80% with efavirenz/tenofovir/emtricitabine and 70% with efavirenz/AZT/3TC in an open-label study that enrolled 517 previously untreated people [2]. On the other hand a 48-week intent-to-treat sub-50 rate of 67% with two ritonavir-boosted regimens--fosamprenavir or lopinavir plus 3TC/abacavir [3]--barely exceeded rates with atazanavir/ritonavir/efavirenz. This study, KLEAN, included 878 treatment-naive people.

But 48-week response was not the primary endpoint of this trial; the main aim was to gauge changes in fasting triglycerides between week 0 and week 8. Triglycerides vaulted impressively over those 2 months in both dosage arms (Table 2). Thal said these triglyceride jumps exceed those logged earlier with unboosted 400-mg atazanavir, 300/100 mg of atazanavir/ritonavir, or efavirenz. After week 8 these gains leveled off, but some people ended up atop the normal range. After 48 weeks of follow-up 3 people (5%) in the combined arms had triglyceride tallies above 400 mg/dL.
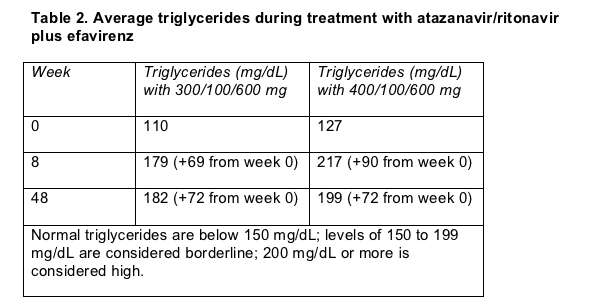
Ill-boding low-density lipoprotein (LDL) cholesterol climbed 11% in the 300/100-mg arm by week 8 and 13% in the 400/100-mg arm, but LDL did not rise farther through 48 weeks. "Good" high-density lipoprotein (HDL) cholesterol did continue to climb after week 8. In the 300/100-mg group the week-8 gain averaged 29% and the week-48 gain 54%. HDL cholesterol surged 26% by week 8 in the 400-mg group and 45% by week 48.
The higher PI dose caused no more side effects or out-of-line lab readings than the lower dose, except for grade 3 or 4 bilirubin, which affected 40% in the 400/100-mg group and 13% in the 300/100-mg group. Grade 3 or 4 alanine and aspartate aminotransferase gains affected 10% and 7% taking the lower atazanavir dose and 7% and 3% taking the higher dose. Overall rates of treatment-related grade 2 to 4 problems proved equivalent in the two treatment arms--in 8 people (26%) taking 300/100 mg and in 9 (30%) taking 400/100 mg.
Nucleoside-sparing first-line regimens have not earned the sanction of major guideline writers and probably will not until someone finds a non-NRTI mix superior to standard NRTI-backbone combos in potency, safety, or tolerance. The atazanavir trial, lacking an NRTI-bolstered control arm, says nothing about whether atazanavir/ritonavir/efavirenz does better than--or as well as--atazanavir/ritonavir or efavirenz plus two nukes. Lopinavir/ritonavir/efavirenz (533/133 twice daily, 600 once daily) did not match efavirenz plus two nucleosides as a maintenance regimen through 110 weeks in AIDS Clinical Trials Group (ACTG) study A5116, largely because of heightened toxicity in the NRTI-sparing arm [4]. But ACTG statisticians divined a trend toward a higher virologic failure rate in the lopinavir/efavirenz group than in the efavirenz/nucleoside group (P = 0.088 intent-to-treat, P = 0.064 excluding participants who modified treatment).
Another worry with up-front PI/NNRTI duos is a potentially higher risk of NNRTI resistance upon virologic failure, a threat uncovered in ACTG A5142 [5]. This trial randomized 753 treatment-naive people to efavirenz plus two nucleosides, to lopinavir/ritonavir plus two nucleosides, or to efavirenz plus lopinavir/ritonavir with no nucleosides. Among the 33 virologic failure samples genotyped in the efavirenz/nucleoside arm, 16 (48%) had nonnucleoside mutations. Among the 39 failures genotyped in the efavirenz/lopinavir arm, 27 (69%) had nonnucleoside mutations.
These analyses are preliminary but nonetheless striking. Why two nucleosides would protect efavirenz from resistance better than such a powerful drug as lopinavir/ritonavir remains unclear. If continuing work in this trial, and perhaps others, bears out this trend, avoiding the generally mild toxicities of today's first-line nucleoside duos hardly seems worth this resistance risk.
In the much smaller atazanavir trial, Thal had genotypic results on only 3 people whose regimen flopped. None had major PI mutations, 2 had the nonnucleoside K103N mutation, and one had a substitution at reverse transcriptase position 179.
Efavirenz and tenofovir misfire as maintenance mates
Once-daily efavirenz, tenofovir, and 3TC provide a potent, convenient, and relatively safe combination. Is efavirenz plus tenofovir as potent, more convenient, and even safer? Not according to 48-week results of the COOL trial run by Pierre-Marie Girard (St. Antoine Hospital, Paris) and colleagues across France [6]. They recruited 143 people with at least 3 months of antiretroviral experience, a viral load under 50 copies for at least 6 months, no regimen failures in their records, no threatening lab or clinical abnormalities, and good kidney function (creatinine clearance above 60 mL/min). Girard and confreres randomized these people to efavirenz plus tenofovir/3TC once daily or efavirenz plus only tenofovir once daily.
The study group had a median age of 40 years, a median treatment duration of 3.7 years, and median CD4 counts of 497.5 in the triple-drug arm and 410 in the double-drug arm. About one third had CDC stage C disease, and 70% were taking AZT/3TC in their current regimen.
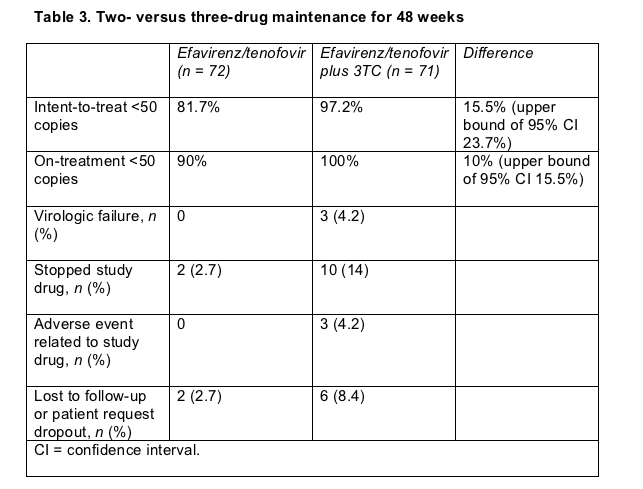
After 48 weeks of follow-up both an intent-to-treat analysis and an as-treated analysis rated efavirenz/tenofovir virologically inferior to the three-drug combo (Table 3). Girard counted 3 virologic failures in the efavirenz/tenofovir group (4.2%) and none in the triple-therapy arm. Nonnucleoside resistance mutations arose during these failures. One person wound up with Y188L, another with G190A, and the third with G190E. In a finding that contradicts a key premise of the study--better tolerance and safety with two drugs versus three--dropouts for side effects and other reasons proved higher in the two-drug arm (Table 3). These results clearly demonstrate that reasonable assumptions do not always hold up in clinical trials.
People taking three drugs added 35 CD4 cells during the study, compared with 14 CD4s in the two-drug group, but that difference lacked statistical significance (P = 0.94). Median hemoglobin rose 0.80 g/dL in the triple-therapy arm and 0.45 g/dL in the double-therapy arm, a difference that also fell short of significance (P = 0.14). Creatinine clearance fell 3.3 mL/min in the three-drug arm and rose 1.7 mL/min in the two-drug arm, again a nonsignificant difference (P = 0.17).
In the group as a whole, triglycerides fell 0.25 mmol/L (P < 0.001) and total cholesterol fell 0.3 mmol/L (P < 0.001). Dangerous low-density lipoprotein cholesterol dropped 0.2 mmol/L in the triple-therapy group (P = 0.015 versus baseline) but rose (nonsignificantly) by 0.15 mmol/L in the two-drug group (P = 0.5). The treatment groups did not differ in lipid changes or fat distribution. Subcutaneous abdominal fat rose significantly in the whole study group (9 cm2, P = 0.017), while the visceral-to-subcutaneous adipose tissue ratio fell by 0.05 (P = 0.06).
Three first-line NRTIs--one more time
People with long memories and good HIV meeting attendance records may recall that Princey Kumar (Georgetown University, Washington, DC) rose as one of several troubled questioners when Trip Gulick first presented ACTG A5095 results showing Trizivir (AZT/3TC/abacavir) inferior to efavirenz-containing regimens at the 2003 Paris IAS meeting [7,8]. Despite the higher risk of failure with Trizivir, she wondered, does the fixed-dose threesome still merit first-line ranking for some people because of its potentially greater tolerability, because it requires lower copayments (in countries with antique health insurance like the US), and because it preserves both PIs and NNRTIs for backup regimens?
Unlike other questioners, Kumar did something about her concern that A5095 might peremptorily ban Trizivir from the first-line ranks: She organized another randomized trial to test Trizivir as initial therapy [9]. But unlike the ACTG A5095 team, she pitted Trizivir against a generally unfavored first-line drug, unboosted atazanavir (400 mg daily), instead of a universally acknowledged up-front component like efavirenz or lopinavir/ritonavir. The latest US Department of Health and Human Services guidelines list unboosted atazanavir as an "alternative" rather than a "preferred" first-line ingredient [10]. Trizivir now ranks as an "acceptable" first-line regimen that is "inferior to preferred or alternative components." Those ratings seem unlikely to change after Kumar's ACTION trial.
Kumar and coworkers at other US sites signed up 279 people with a viral load below 200,000 copies and at least 100 CD4 cells, randomizing them to Trizivir or unboosted atazanavir plus AZT/3TC. Median pretreatment CD4 counts stood at 274 in the Trizivir group and 262 in the atazanavir group, while median loads measured 4.48 and 4.64 log copies/mL (about 30,000 and 44,000 copies). More than 80% in each treatment group had fewer than 100,000 HIV RNA copies when treatment began.
The primary endpoint was the percentage of people with a viral load below 50 copies at week 48 in an intent-to-treat analysis that counted missing data or a drug change as failure. The protocol did allow switches for abacavir hypersensitivity reactions and for two side effects tied to atazanavir--jaundice and scleral icterus.
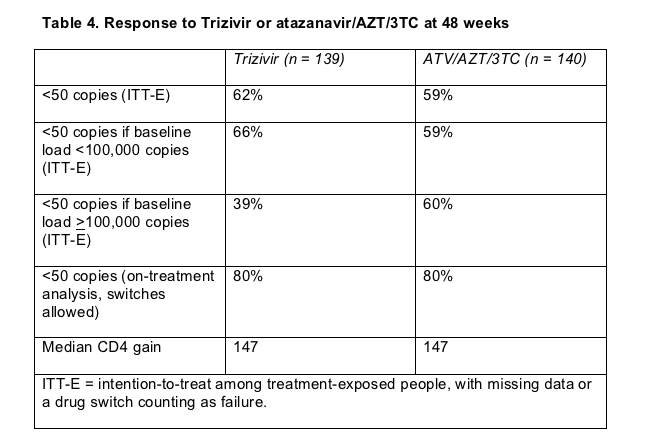
Among people assigned to Trizivir, 103 (74%) finished 48 weeks of study, compared with 98 (70%) in the atazanavir arm. Virologic failure accounted for most dropouts. Sub-50 copy rates at week 48 measured 62% in the Trizivir arm and 59% in the atazanavir arm, a result indicating equivalence of the two regimens (Table 4) but probably not equivalence with accepted first-line options (see the cross-study comparison under "Spare the nukes" above) .
People who started Trizivir with a load above 100,000 copies did particularly poorly (Table 4). That result confirms a 48-week finding in a trial comparing AZT, 3TC, and abacavir with unboosted indinavir plus AZT/3TC in previously untreated people [11]. In that trial comparing the three NRTIs with a now-outmoded PI combo, overall virologic outcomes proved equivalent between the two arms at week 48. When Gulick compared Trizivir with efavirenz regimens in ACTG A5095, however, Trizivir did worse regardless of pretreatment viral load [8].
Kumar and colleagues defined failure five ways--as failure to have at least a 1-log (10-fold) drop in viral load by week 12, failure to have a load under 400 copies by week 24, a confirmed load below 50 copies followed by a confirmed load above 400 copies by week 24, a confirmed load above 400 copies at week 24 or later, or a single load above 400 copies at week 48. By those criteria the ACTION researchers counted 18 virologic failures (13%) in the Trizivir group (8 [44%] with a baseline load above 100,000 copies) and 17 (12%) in the atazanavir group (5 [29%] with a baseline load above 100,000 copies).
Among people taking Trizivir, 10 had treatment-related mutations by week 48--nine M184Vs and one thymidine analog mutation. Eleven people taking unboosted atazanavir ended up with resistance mutations, but none were major PI mutations.
Grade 3 or 4 hyperbilirubinemia affected 21 people (16%) taking atazanavir and none taking Trizivir. Triglycerides rose 5% in the atazanavir group and 11% in the Trizivir group. "Bad" low-density lipoprotein cholesterol fell 6% with atazanavir and 5% with Trizivir, while "good" high-density lipoprotein cholesterol rose in both groups.
Kumar proposed that Trizivir remains a reasonable first-line option for people with a viral load under 100,000 copies, but not everyone attending this ICAAC slide session agreed. Paul Volberding of the San Francisco VA and Christine Katlama from Pitie-Salpetriere Hospital in Paris both voiced concern about touting Trizivir for first-line therapy. Session cochair Constance Benson, from the University of California, San Diego, noted that durability has been fragile with triple-nucleoside regimens. Kumar did not have data beyond 48 weeks to allay that concern.
Mark Mascolini writes about HIV infection (markmascolini@earthlink.net).
References
1. Ward D, Bush L, Thiry A, et al. Atazanavir/ritonavir and efavirenz NRTI-sparing regimens in treatment-naive adults: BMS-121 study. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1057.
2. Gallant JE, DeJesus E, Arribas JR, et al. Tenofovir DF, emtricitabine, and efavirenz vs. zidovudine, lamivudine, and efavirenz for HIV. N Engl J Med 2006;354:251-260.
3. Eron J Jr, Yeni P, Gathe J Jr, et al. The KLEAN study of fosamprenavir-ritonavir versus lopinavir-ritonavir, each in combination with abacavir-lamivudine, for initial treatment of HIV infection over 48 weeks: a randomised non-inferiority trial. Lancet 2006;368:476-482.
4. Fischl M, Bassett R, Collier A, et al. Randomized, controlled trial of lopinavir/ritonavir + efavirenz vs efavirenz + 2 nucleoside reverse transcriptase inhibitors following a first suppressive 3- or 4-drug regimen in advanced HIV disease. 12th Conference on Retroviruses and Opportunistic Infections. February 22-25, 2005. Boston. Abstract 162.
5. Riddler SA, Haubrich R, DiRienzo G, et al. A prospective, randomized, phase III trial of NRTI-, PI-, and NNRTI-sparing regimens for initial treatment of HIV infection--ACTG 5142. XVI International AIDS Conference. August 13-18, 2006. Toronto. Abstract THLB0204.
6. Girard PM, Cabie A, Michelet C, et al. Tenofovir DF + efavirenz vs tenofovir DF + efavirenz + lamivudine maintenance regimen in virologically controlled patients: COOL trial. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1383.
7. Gulick RM, Ribaudo HJ, Shikuma CM, et al. ACTG 5095: a comparative study of 3 protease inhibitor-sparing antiretroviral regimens for the initial treatment of HIV infection. 2nd IAS Conference on HIV Pathogenesis and Treatment. July 13-16, 2003, Paris. Abstract 41.
8. Gulick RM, Ribaudo HJ, Shikuma CM, et al. Triple-nucleoside regimens versus efavirenz-containing regimens for the initial treatment of HIV-1 infection. N Engl J Med 2004;350:1850-1861.
9. Kumar PN, Patel P, Salvato P, et al. ACTION study: efficacy and safety of abacavir/lamivudine/zidovudine BID versus lamivudine/zidovudine BID + atazanavir QD in ART-naive HIV-1 infected subjects. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract H-1058.
10. US Department of Health and Human Services Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. October 10, 2006 (http://www.aidsinfo.nih.gov/Guidelines/GuidelineDetail.aspx?MenuItem=Guidelines&Search=Off&GuidelineID=7&ClassID=1).
11. Staszewski S, Keiser P, Montaner J, et al. Abacavir-lamivudine-zidovudine vs indinavir-lamivudine-zidovudine in antiretroviral-naive HIV-infected adults: a randomized equivalence trial. JAMA 2001;285:1155-1163.
|
| |
|
 |
 |
|
|