 |
 |
 |
| |
The PRESCO study: role of extended therapy and/or optimal doses of ribavirin
in the treatment of chronic hepatitis C in HIV-infected patients
|
| |
| |
Reported by Jules Levin
ICAAC, Sept 2006, San Francisco
M. Nunez,1 C. Miralles,2 M.A. Berdun,3 K. Aguirrebengoa,4 A. Ocampo,2 E. Losada,5 J. García-Samaniego,6 V. Soriano,1 on behalf of the PRESCO Group
1Department of Infectious Diseases, Hospital Carlos III, Madrid; 2Hospital Xeral-Cíes, Vigo; 3Hospital San Jorge, Huesca; 4Hospital de Cruces, Bilbao; 5Hospital Clínico Universitario, Santiago; 6Hepatology Unit, Hospital Carlos III, Madrid, Spain
This work was supported in part by grants from Fondo de Investigaciones Sanitarias (#02/3040), Fundacion Investigacion y Educacion en SIDA (IES), the European VIRGIL Network, Red de Investigacion en SIDA (RIS), Agencia Lain Entralgo, Instituto de Salud Carlos III and Roche.
Brief Summary: this was an open-label study in almost 389 HCV/HIV coinfected patients who received Pegasys plus 1000-1200 mg ribavirin daily to evaluate weight-based ribavirin dosing of 1000 or 1200 mg based on weight, and to exaluate extended duration of therapy. There was no comparison arm of 800 mg daily ribavirin. The study results found an SVR of 50% overall, 36% in genotype 1 and 72% in genotype 2/3. The APRICOT Study in coinfection used 800mg RBV flat dosing & overall SVR was 40%, 29% in genotype 1 and 62% in genotype 2/3. The PRESCO results find extended duration of therapy, in particular for 72 weeks rather than 48 weeks in genotype 1 was superior (53% vs 31%). Patients with genotype 2/3 had an SVR of 82% with 48 weeks therapy and 66.7% with 24 weeks therapy. However, there was a 44% discontinuation rate among genotype 1 patients who were in the prolonged treatment arm. Still, the relapse rate was 35.9% for genotype _ receiving 48 weeks therapy and 27.3% receiving 72 weeks therapy. Bear in mind, other studies have found benefit of extended therapy duration in patients who mount a partial viral load response. A study in patients who are 'difficult-to-treat' and have an ETR would I think find benefit to extended duration of therapy and reduce the relapse rate.
STUDY OBJECTIVE
The objectives of this study were therefore to evaluate
- the efficacy of weight-based standard-dose ribavirin (1000-1200 mg/day) in
combination with peginterferon alfa-2a (40KD)
- the role of extended periods of therapy for chronic hepatitis C in HIV-infected
patients.
AUTHOR CONCLUSIONS
In this study, an overall SVR rate of 49.6% was achieved. This SVR rate was
higher than previous studies with pegylated interferon-based therapy in HIV-HCV co-infected patients.[3,6]
The higher SVR rate in this study is likely to be the result of the use of ribavirin 1000-1200 mg/day with peginterferon alfa-2a (40KD) (PEGASYS), compared
with previous reports that treated HIV-HCV co-infected patients with a fixed low (800 mg/day) dose of ribavirin (COPEGUS).[3] No major toxicity concerns were
seen with the higher dosing schedule used in this study.
The benefit associated with extension of treatment should be interpreted with caution because of the high drop-out rates in patients allocated to the longer treatment schedules.
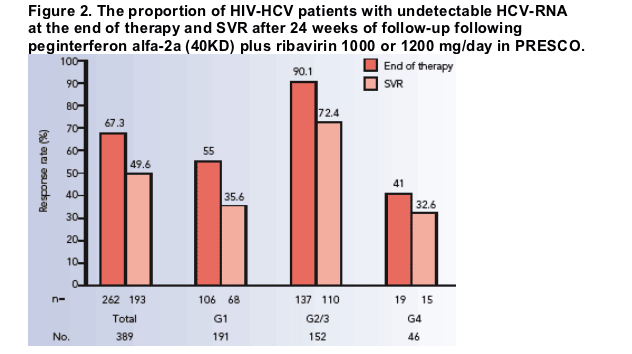
RESULTS
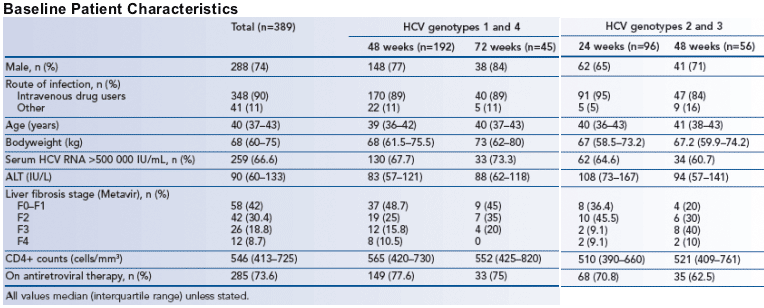
Baseline characteristics
A total of 389 patients were included in the trial and received at least one dose of study medication.
- of these, 191 were infected by HCV genotype 1, 152 with HCV genotype 2/3, and 46 with HCV genotype 4.
Baseline characteristics for these patients are shown in Table 1.
Efficacy
Overall
In total, 193 patients achieved an SVR (50%) following treatment with peginterferon alfa-2a (40KD) plus ribavirin 1000 or 1200 mg/day.
- similar SVR rates were seen in genotype 1 and 4 patients (36% and 33%, respectively; Figure 2)
-- the highest SVR rate was recorded in HCV genotype 2/3 patients (72%; Figure 2).
In an intent-to-treat (ITT) analysis, end-of-treatment response was seen in 262
(67%) patients.
- end-of-treatment responses by genotype are shown in Figure 2.
Comparison between short and extended treatment durations
Overall, patients allocated to the extended treatment arms showed better treatment responses (irrespective of HCV genotype) compared with those allocated to the shorter treatment arms (Figure 3).
- however, it should be noted that a substantial proportion of HCV genotype 1
and 4 patients allocated to the extended treatment arm did not complete the
planned length of therapy.
Although more patients relapsed in the shorter treatment group, the difference
did not reach statistical significance.
Logistic regression analysis
In multivariate analysis, infection with HCV genotype 2 or 3 (odds ratio [OR] 4.41
[95% CI 2.82-6.88; p<0.0001]) and HCV-RNA <500 000 IU/mL (OR 2.06 [95%CI
1.31-3.26; p=0.002]) were identified as independent predictors of SVR.
Prolonged treatment was not associated with higher SVR.
Safety
The most commonly seen adverse events were depression and/or other psychiatric illnesses along with weight loss, followed by hematologic events and asthenia.
Treatment was interrupted because of severe adverse events in 33 cases (8.5%), most commonly hematologic disorders and depression.
Treatment was interrupted within the first 24 weeks because of virologic failure in
66 patients (17%).
In addition, 108 patients (28%) either stopped treatment prematurely because of
side effects, voluntarily, or were lost-to-follow-up.
- twenty-three patients (5.9%) voluntarily interrupted treatment or were poorly
compliant during the first 24 weeks of therapy for HCV genotype 2 and 3
patients, or the first 48 weeks of therapy for HCV genotype 1 and 4 patients.
Treatment interruptions occurred mainly in the prolonged treatment arms.
- forty-five of 101 patients (44.5%) allocated to the extended treatment arms
refused to continue the medication beyond the minimum period of therapy
- this was particularly frequent in patients infected with HCV genotype 1 and 4.
Overall, the dose of either peginterferon alfa-2a (40KD) or ribavirin was modified
in 116 (29.8%) patients.
- the dose of peginterferon alfa-2a (40KD) was decreased at some point during
treatment in 60 patients (15.4%), and the dose of ribavirin in 84 (21.6%)
- changes in the dosage of peginterferon alfa-2a (40KD) and ribavirin were not
associated with lower SVR.
Introduction
Chronic hepatitis C virus (HCV) infection affects around 30% of human
immunodeficiency virus (HIV)-positive individuals in developed countries.[1]
Complications associated with chronic HCV infection represent a major cause of
morbidity and mortality in HIV-HCV co-infected patients,[2] and given the faster
progression to end-stage liver disease in this population, these patients have a
considerable need for treatment.[2]
Treatment of chronic hepatitis C with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) has been associated with lower sustained virologic response (SVR) rates (approximately 40%) in this population[3] compared with HCV mono-infected individuals, in whom SVR rates of up to 66% have been achieved.[4]
The poorer therapeutic response in patients with HIV-HCV co-infection compared
with HCV mono-infected individuals has yet to be elucidated, but could be due
to a higher incidence of drug discontinuation (due to side effects) or a lower
intrinsic activity of HCV medication in the HIV setting.[5] One factor that may favor the latter hypothesis is that most studies in HIV-HCV co-infection have used a low flat-dose (800 mg/day) of ribavirin[3,6] and/or short durations of therapy.
STUDY METHODS
Patients
Patients eligible for inclusion into PRESCO were co-infected with HIV and HCV viruses, were over 18 years of age, had elevated serum alanine aminotransferase (ALT), compensated liver disease and CD4+ lymphocyte counts above 300 cells/_L.
Patients were naive to interferon-based therapy.
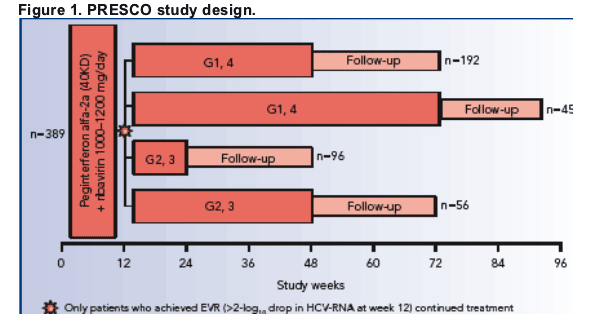
Study design
PRESCO was a prospective, multicenter, open-label, comparative study conducted in Spain.
- the study design of PRESCO is shown in Figure 1.
All patients received (Pegasys) peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin 1000-1200 mg/day.
Ribavirin was administered at either 1000 mg/day for patients weighing <75 kg
and 1200 mg/day for patients weighing 375 kg.
Patients received either a short or extended treatment duration (Figure 1).
- patients infected with HCV genotypes 2 and 3 were treated for 24 or 48 weeks
- patients infected with HCV genotypes 1 and 4 were treated for 48 or 72 weeks
- following treatment there was a follow-up period of 24 weeks for all patients
- at week 12, only patients with >2-log10 drop in HCV RNA were allowed to
continue on therapy.
All demographic data and laboratory assessments were recorded at baseline.
Serum HCV RNA was assessed at weeks 4, 12 and then every 12 weeks.
- other laboratory assessments included serum HIV RNA, CD4+ cell counts, ALT
levels, and blood counts
- the use of granulocyte colony-stimulating factors and erythropoietin was
permitted to manage hematologic adverse events.
Efficacy and safety
The primary efficacy end-point was SVR defined as undetectable serum HCV RNA (<50 IU/mL, COBAS AMPLICORä HCV Test, v2.0), 24 weeks after end of treatment.
Early virologic response (EVR; >2-log10 drop in HCV RNA at week 12) and rapid
virological response (RVR; undetectable HCV RNA at week 4) were also assessed.
Safety assessments were carried out at weeks 4, 12 and every 12 weeks thereafter by personal interview, physical examination and laboratory tests.
Stepwise reductions in the dose of peginterferon alfa-2a (40KD) to 135 or
90 _g/week were allowed, as were reductions in the dose of ribavirin by 200 mg/day to a minimum of 600 mg/day. Ribavirin could be temporarily interrupted for a minimum of 1 week and maximum of 2 weeks in cases of severe anemia.
Logistic regression analyses
Univariate logistic regression and stepwise forward multivariate regression
analyses were performed to examine the association of several variables with the
response to anti-HCV therapy.
REFERENCES
1. Rockstroh J, Mocroft A, Soriano V, et al. Influence of hepatitis C virus infection on HIV-1 disease progression and response to highly active antiretroviral therapy. J Infect Dis 2005; 192: 992-1002
2. Martin-Carbonero L, Soriano V, Valencia E, et al. Increasing impact of chronic viral hepatitis on hospital admissions and mortality among HIV-infected patients. AIDS Res Hum Retroviruses 2001; 17: 1467-71
3. Torriani F, Rodriguez-Torres M, Rockstroh J, et al. Peginterferon alfa-2a plus ribavirin forchronic hepatitis C virus infection in HIV-infected patients. N Engl J Med 2004; 351: 438-50
4. Zeuzem, S, Pawlotsky JM, Lukasiewicz E, et al., International, multicenter, randomized, controlled study comparing dynamically individualized versus standard treatment in patients with chronic hepatitis C. J Hepatol 2005; 43: 250-7
5. Torriani F, Ribeiro R, Gilbert T, et al. HCV and HIV dynamics during HCV treatment in HCV/HIV coinfection. J Infect Dis 2003; 188:1498-507
6. Carrat F, Bani-Sadr F, Pol S, et al. Pegylated interferon alfa-2b vs standard interferon alfa-2b, plus ribavirin, for chronic hepatitis C in HIV-infected patients. JAMA 2004; 292: 2839-48
|
| |
|
 |
 |
|
|