 |
 |
 |
| |
Pharmacokinetic interaction between the HIV protease inhibitors TMC114 and saquinavir, in the presence of low-dose ritonavir
|
| |
| |
Reported by Jules Levin
IDSA, Toronto, Oct 12-15, 2006
Sekar V,1 Lefebvre E,1 Mariën K,1 De Pauw M,2 Vangeneugden T,2 Hoetelmans R2
1Tibotec Inc., Yardley, PA, USA; 2Tibotec BVBA, Mechelen, Belgium
AUTHOR CONCLUSIONS
The pharmacokinetics of TMC114 were affected by the co-administration of SQV and RTV; TMC114 Cmin, Cmax and AUC12h were decreased by 42%, 17% and 26%, respectively, when SQV was added to TMC114/r.
SQV PK parameters were unaffected by the addition of TMC114/r.
Co-administration of TMC114/SQV/r was associated with a higher incidence of AEs and discontinuations due to AEs than administration of TMC114/r or SQV/r alone.
SQV should not be combined with TMC114/r as it results in significant reduction in TMC114 exposure.
ABSTRACT
Objectives: The primary objective of this study was to investigate the potential for a pharmacokinetic (PK) interaction between TMC114 (darunavir), in combination with low-dose ritonavir (RTV; TMC114/r), and saquinavir (Invirase® hard gelatin capsules; SQV). The secondary objective was to assess the short-term safety and tolerability of co-administered TMC114/r and SQV.
Methods: The study population consisted of two panels of 16 HIV-negative healthy volunteers each. Panel 1 received TMC114/r 400/100mg bid (Treatment A) in one session and TMC114/SQV/r 400/1,000/100mg bid (Treatment C) in the other session. Panel 2 received SQV/r 1,000/100mg bid (Treatment B) in one session and TMC114/SQV/r 400/1,000/100mg bid (Treatment C) in the other session. All treatments were administered for 13 days bid with an additional single dose on Day 14. PK assessments were performed on Day 14 and PK parameters for TMC114, RTV and SQV were compared between treatments. Safety and tolerability were also assessed.
Results:
Based on the least square means (LSM) ratio and 90% confidence intervals (CI), when TMC114/r and SQV were co-administered, TMC114 area under the curve (AUC12h), maximum plasma concentration (Cmax) and minimum plasma concentration (Cmin) decreased by 26% (LSM 74%; CI 63-86%), 17% (LSM 83%; CI 75-92%) and 42% (LSM 58%; CI 47-72%), respectively, compared to when TMC114/r was administered alone.
Upon co-administration, SQV AUC12h, Cmax and Cmin decreased by 6% (LSM 94%; CI 76-117%), 6% (LSM 94%; CI 78-113%) and 18% (LSM 82%; CI 52-130%), respectively, compared to when SQV/r was administered alone.
Systemic exposure to RTV was increased by 34% when SQV was added to treatment with TMC114/r. The co-administration of TMC114/SQV/r was generally safe and well tolerated.
Conclusions: The combination of SQV and TMC114/r is currently not recommended as exposure to TMC114 is decreased when combined with SQV, which is expected to be clinically relevant.
SQV is administered as 1,000mg bid in combination with 100mg bid RTV (SQV/r), as this is a frequently used regimen for SQV in HIV-infected patients.
Potential for a drug-drug interaction exists when TMC114, SQV and RTV are co administered, as these drugs are all essentially completely metabolized by CYP3A4. Furthermore, RTV is a potent inhibitor of CYP3A4 metabolism, able to increase SQV and TMC114 concentrations at low dosages.
This study assessed the effect of steady-state concentrations of SQV on the steady-state pharmacokinetics of TMC114, and vice versa, co-administered with low-dose RTV. Short-term safety and tolerability of co-administered TMC114, SQV and RTV were also assessed.
Study design
This was a Phase I, open-label, randomized, two-way cross-over trial (TMC114-C138) designed to investigate the PK interaction between TMC114 (formulated as the tablet TF001) and SQV (formulated as the hard gel capsule Invirase®) both co-administered with low-dose RTV (formulated as a capsule Norvir®).
The study population consisted of 32 HIV-negative, healthy volunteers, aged 18-55 years, divided into two panels of 16 participants.
Volunteers were randomized to one of two panels:
- Panel 1 (n=16) received TMC114/r 400/100mg bid (Treatment A) and TMC114/SQV/r 400/1,000/100mg bid (Treatment C) in two sessions
- Panel 2 (n=16) received SQV/r 400/100mg bid (Treatment B) and Treatment C in two sessions.
All treatments were administered orally under fed conditions for 13 days, with an additional single morning dose on Day 14. Treatment sessions were separated by a wash-out period of at least 14 days.
Volunteers remained in the testing facility for at least 24 hours after receiving the first dose of treatment. SQV/r and/or TMC114/r were administered for 14 days to allow plasma concentrations to reach steady-state.
PK and safety assessments
PK profiles of TMC114, SQV and RTV were determined on Day 14 up to 12 hours after intake of study medication.
Predose plasma concentrations (C0h) of TMC114, SQV and RTV were determined on Days 1, 4, 7, 10, 12, 13 and 14.
Blood samples were taken at screening, on Days 1, 4, 7, 10, 12, 13 and predose, 1, 2, 3, 4, 5, 6, 9 and 12 hours after dosing on Day 14. An additional sample was taken at the time of discontinuation for volunteers who withdrew prematurely.
Plasma concentrations of TMC114, SQV and RTV were determined by validated liquid chromatography mass spectrometry/mass spectrometry methods.
The primary PK parameters were Cmin, Cmax and AUC12h on the logarithmic scale. Other parameters determined were time to Cmax (tmax), average steady-state plasma concentration (Css,av) and fluctuation index (FI).
Statistical analyses were performed to compare
- TMC114: Treatment A and C (Panel 1)
- SQV: Treatment B and C (Panel 2)
- RTV: Treatment A, B and C (Panels 1 and 2).
The LSMs of the primary parameters for each treatment group were estimated with a linear mixed effects model, controlling for period, sequence and treatment as fixed effects and individual as a random effect. A 90% CI was constructed around the difference between the LSMs. Tmax was subjected to the non-parametric Koch test.
Safety and tolerability evaluations were recorded continuously. Adverse events (AEs) were checked at every visit from screening onwards. Severity and drug relationship of AEs towards TMC114, SQV and RTV were recorded.
Withdrawal criteria included withdrawal of consent, withdrawal as deemed necessary by investigators, any grade 3 or 4 AE or toxicity, 3 grade 2 rash in combination with any increase in aspartate aminotransferase (AST) or alanine
aminotransferase (ALT), 3 grade 2 elevation in AST/ALT, clinical hepatitis, renal complications related to trial medication and persistent 3 grade 2 nausea.
RESULTS
Participant disposition
Of the 32 volunteers randomized to treatment, 26 completed the trial while six discontinued due to AEs. The majority of volunteers were male (78%) and the median age was 40.5 years (range 20-53 years).
PK profiles of TMC114, SQV and RTV for Day 14 were available for 14 volunteers of Treatment A (TMC114/r), 14 of Treatment B (SQV/r), 15 of Treatment C, Panel 1 (TMC114/SQV/r) and 12 of Treatment C, Panel 2 (TMC114/SQV/r).
PK results for TMC114
Steady-state concentrations of TMC114 were achieved prior to PK sampling on Day 14.
Plasma concentrations of TMC114 were lower when TMC114/r was co administered with SQV (Treatment C) than when administered alone (Treatment A; Figure 1).
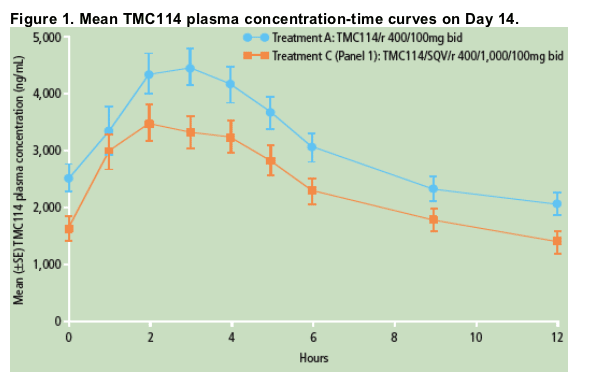
A summary of key PK parameters of TMC114 for Treatments A and C is shown in Table 1
- TMC114 median Cmax were reached 3.0 hours (range 1.0-5.0) post-dose for TMC114/r and 2.0 hours (range 1.0-9.0) post-dose for TMC114/SQV/r
- mean Cmin, Cmax and AUC12h of TMC114 were lower with SQV/r than with administration of TMC114/r alone; based on the LSM ratio and 90% CI, TMC114 Cmin, Cmax and AUC12h decreased by 42% (LSM 58%; CI 47-72%), 17% (LSM 83%; CI 75-92%) and 26% (LSM 74%; CI 63-86%), respectively, when SQV was added to TMC114/r. TMC114 tmax was not affected.
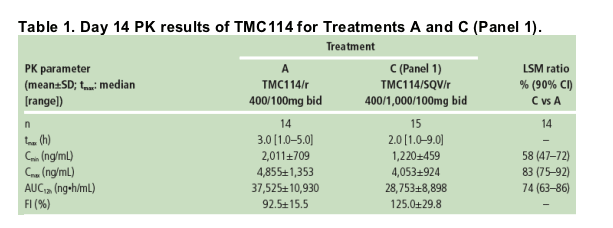
PK results for SQV
Steady-state concentrations of SQV were achieved prior to full PK sampling on Day 14.
Plasma concentrations of SQV were slightly lower when SQV/r was co-administered with TMC114 (Treatment C) than when administered alone (Treatment B; Figure 2).
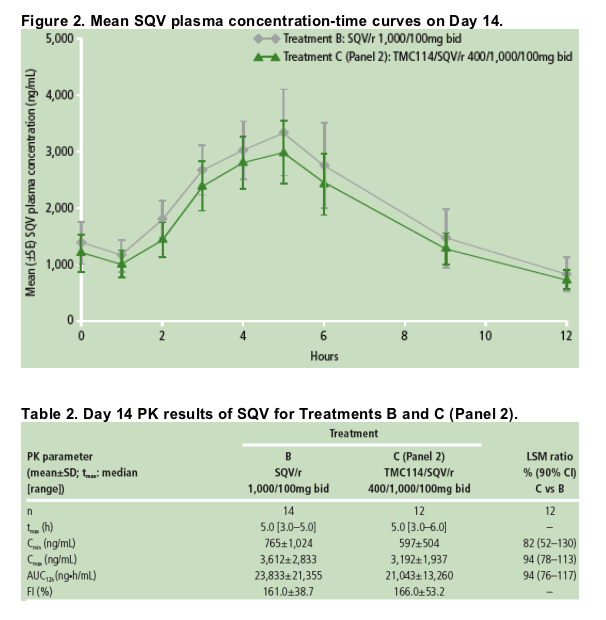
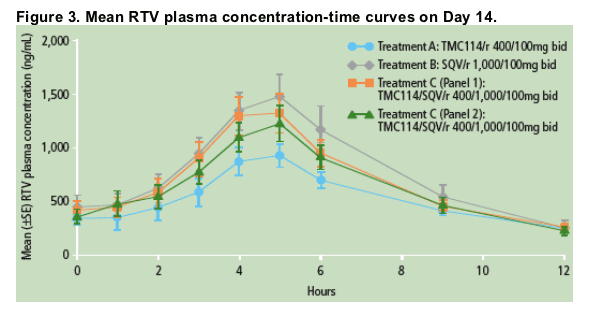
PK results for RTV
Steady-state concentrations of RTV were achieved prior to full PK sampling on Day 14.
Mean plasma concentration-time profiles of RTV are shown in Figure 3. RTV plasma concentrations were highest in the presence of SQV (Treatment B) and lowest in the presence of TMC114 (Treatment A). There was no difference in RTV plasma concentrations between Panels 1 and 2 receiving combined treatment with TMC114/SQV/r (Treatment C).
Safety and tolerability
Overall, 14 volunteers (88%) in Panel 1 and 16 volunteers (100%) in Panel 2 reported at least one AE during the study.
Nine volunteers (56% of Panel 1) reported AEs during Treatment A, 12 (86% of Panel 2) during Treatment B, and nine (60%) and 13 (87%) volunteers during Treatment C (Panels 1 and 2, respectively).
No serious AEs were reported. The majority of AEs (117 in total) were grade 1-2 in severity. Seven volunteers experienced a grade 3 AE, the majority of which occurred in volunteers receiving Treatment C (TMC114/SQV/r). In Panel 1, two volunteers had elevated pancreatic enzyme levels (one during Treatment A and one in the wash-out period after Treatment C) and two had hypercholesterolemia (one during Treatment A and one during Treatment C). In Panel 2, one volunteer experienced hyperbilirubinemia and one had hypercholesterolemia, both during Treatment C, and one volunteer experienced prolonged prothrombin time during Treatment B
- the volunteer who experienced grade 3 hyperbilirubinemia during combined TMC114/SQV/r treatment was diagnosed with Gilbert's disease and grade 2 elevated total bilirubin at screening.
More volunteers discontinued due to AEs with TMC114/SQV/r (n=4; one case each of grade 3 elevated pancreatic enzyme levels, grade 3 hyperbilirubinemia, grade 3 hypercholesterolemia and grade 2 influenza-like illness) than with TMC114/r (one case of grade 3 elevated pancreatic enzyme levels) or SQV/r (one case of grade 2 hypertension). All discontinuations were considered possibly related to treatment.
The most commonly reported AEs were grade 1-2 gastrointestinal (Panel 1: 63%, Panel 2: 69%) and grade 1-2 nervous system disorders (Panel 1: 50%, Panel 2: 69%)
- diarrhea (seven volunteers, 44%) and headache (eight volunteers, 50%) were reported most frequently in Panel 1, while diarrhea (five volunteers, 31%), dizziness (four volunteers, 25%) and headache (four volunteers, 25%) were
reported most frequently in Panel 2. No apparent differences between the treatments were observed.
No consistent or meaningful changes over time in median laboratory parameters were noted. No grade 4 abnormalities were recorded. Several volunteers in Panel 1 developed a treatment-emergent grade 3 elevation in cholesterol (three
volunteers during Treatment A and two during Treatment C) or lipase (one volunteer during Treatment A). For Panel 2, treatment-emergent grade 3 elevations were noted for cholesterol (one volunteer during Treatment B), prolonged prothrombin time (two volunteers during Treatment B) and total bilirubin (one volunteer during Treatment C).
The most common graded laboratory abnormalities for both panels were increases in total bilirubin and cholesterol, and increases in partial thromboplastin and plasma prothrombin time. The incidence of increases in bilirubin and plasma
prothrombin time was highest in Panel 2 during Treatment C compared with all other treatments. The incidence of the remaining graded laboratory abnormalities was comparable across the different treatments.
No clinically relevant changes in vital signs or electrocardiogram parameters were reported.
|
| |
|
 |
 |
|
|