 |
 |
 |
| |
Low-level Viremia Persists for at Least 7 years in Patients on Suppressive Antiretroviral Therapy, using real-time RT-PCR assay with single-copy sensitivity
|
| |
| |
Reported by Jules Levin
XV Intl HIV Drug Resistance Workshop
June 13-17, 2006, Sitges, Spain
S. Palmer1, M. King2, A. Wiegand1, F. Maldarelli1, B Bernstein2, G. Hanna2, S. Brun2, D. Kempf2, J. Mellors3, and J. Coffin1
1HIV Drug Resistance Program, NCI, NIH, Frederick, MD; 2Abbott, Abbott Park, IL; 3University of Pittsburgh, Pittsburgh, PA
BACKGROUND
Persistent viremia can be detected in most HIV-1 infected patients on antiretroviral therapy despite suppression of plasma RNA to <50 copies/ml. Our previous studies have shown diverse antiretroviral regimens suppress
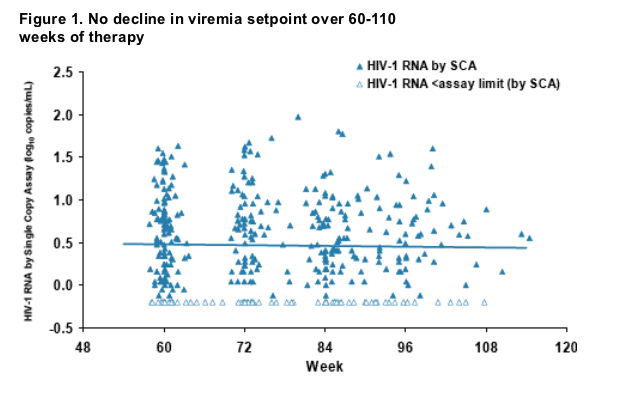
plasma viremia to a new setpoint that correlates with pretherapy viremia1. These
studies could not detect a significant decline in the viremia setpoint over 60-110 weeks of therapy (Figure 1). The current analysis assesses plasma HIV-1 RNA levels in subjects on suppressive therapy for 7 years, using a real-time RT-PCR assay with single copy sensitivity.
ASSAY
An internally controlled real-time RT-PCR assay with single-copy sensitivity (single-copy assay, SCA)2 was used to test all samples.
Based on sample volumes available in this study, the lower limit of assay sensitivity ranged from 0.4 to 1.0 copies/mL.
STUDY ENTRY CRITERIA
In Study 720, antiretroviral-naive subjects received lopinavir/ritonavir (400/100 mg twice daily) with stavudine and lamivudine twice daily for up to 7 years.
Subjects remaining on study for 7 years (360 weeks) who never demonstrated detectable viremia (>50 or >400 copies/mL) during weeks 96-360 were included (Figure 2).
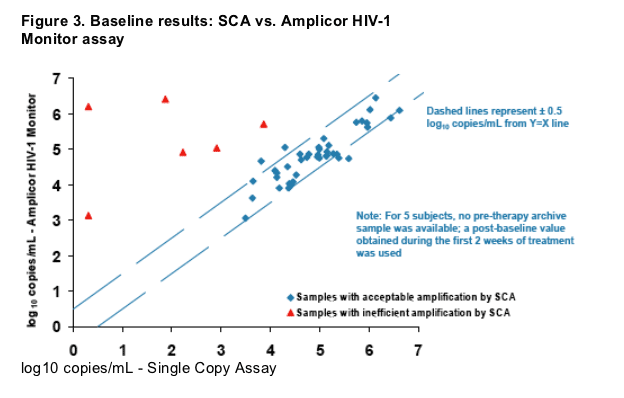
Subjects with comparable baseline assay results (SCA vs. Amplicor) were
included in the longitudinal analysis (Figure 3).
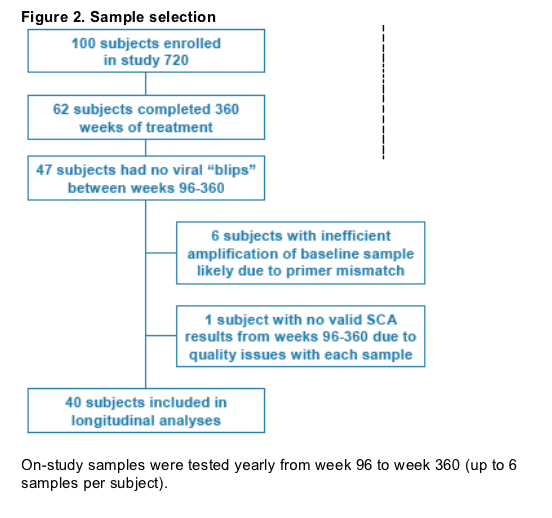
On-study samples were tested yearly from week 96 to week 360 (up to 6 samples per subject).
Linear mixed effects regression models were used to assess plasma HIV-1 RNA values over time.
Between year 6 and year 7, subjects were allowed to switch from stavudine to tenofovir DF3. Primary analyses described above excluded values after switch to
tenofovir; secondary analyses assessed changes in HIV-1 RNA values after the switch to tenofovir using a 1-sample t test.
RESULTS
Approximately 76% of samples obtained between weeks 96 and 360 had detectable low-level viremia ranging from 1-39 copies/mL (median 1.7 copies/mL).
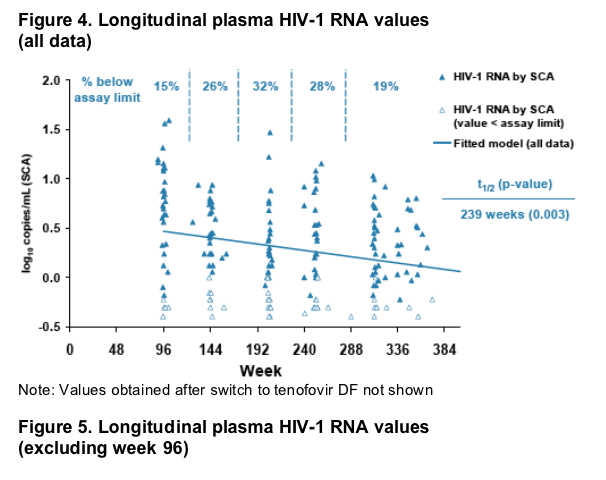
Based on all data from weeks 96-360, a statistically significant decrease
in plasma HIV-1 RNA level was observed (half-life=239 weeks, p=0.003, Figure 4).
However, when data from week 96 were excluded, no decrease in plasma
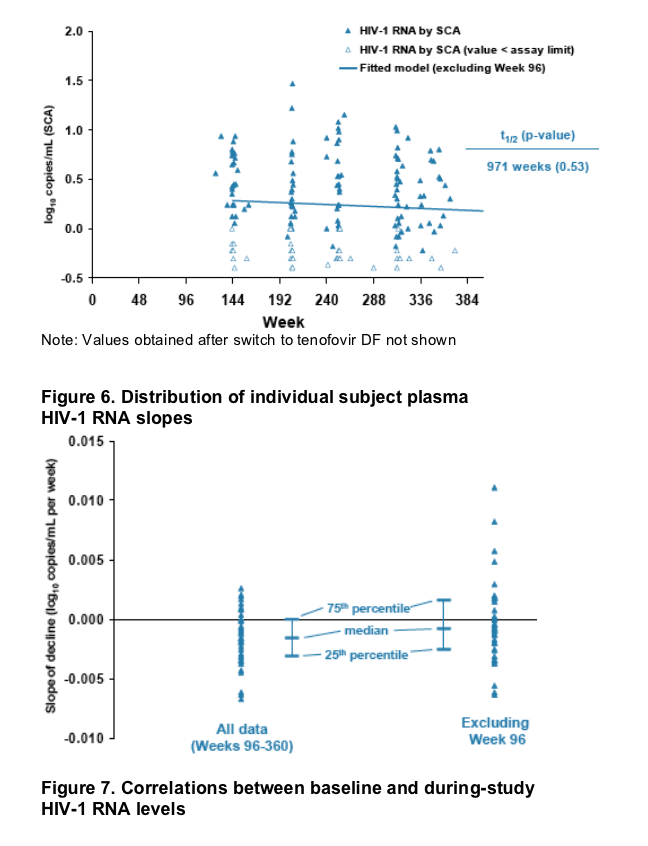
HIV-1 RNA over time was observed (half-life=971 weeks, p=0.53, Figure 5).
The distributions of individual subject slopes based on all data and on data from weeks 144-360 are displayed in Figure 6.
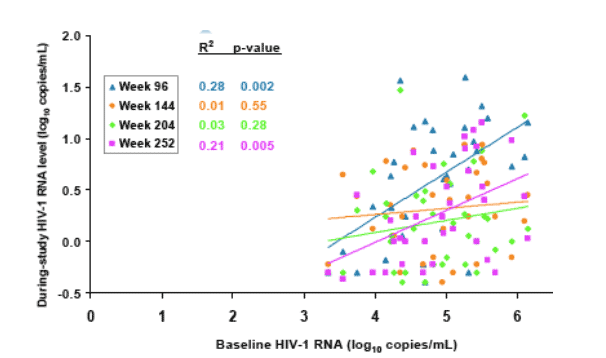
Pre-therapy plasma HIV-1 RNA levels were significantly associated with week 96 levels (p=0.002) and week 252 levels (p=0.005) but not with levels at other timepoints (Figure 7).
No evidence of a change in plasma HIV-1 RNA values was observed in 18
subjects who replaced stavudine with tenofovir DF.
Median value immediately prior to the switch was 1.37 copies/mL, compared to 1.33 copies/mL 12-24 weeks after the switch (p=0.82, 1-sample t test).
AUTHOR CONCLUSIONS
These results are consistent with our prior finding that persistent viremia on treatment may originate from virus produced by cells that are infected before initiation of therapy. The apparent biphasic decay in persistent viremia implies that relatively short-lived cells contribute to viremia through 96-144 weeks, and very long-lived cells contribute thereafter. Testing of additional samples between weeks 60-120 may help to elucidate distinctions between phases of decay of persistent viremia.
References:
1) Palmer S, et al. 12th Conference on Retroviruses and Opportunistic Infections, Boston, MA, February 2005. Abstract 163.
2) Palmer S, et al. J Clin Microbiol 2003;41(10):4531-36.
3) da Silva B, et al. 7th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV, Dublin, Ireland, November 2005. Abstract L957.
|
| |
|
 |
 |
|
|