 |
 |
 |
| |
ANALYSES OF SUSCEPTIBILITY AND CROSS-RESISTANCE BETWEEN TMC114 AND OTHER PROTEASE INHIBITORS AMONG >56,000 ROUTINE SAMPLES, USING LINEAR REGRESSION MODEL-BASED FOLD CHANGE PREDICTIONS
|
| |
| |
Download the poster this article is based on in PDF format
Reported by Jules Levin
XV Intl HIV Drug Resistance Workshop
June 13-17, 2006, Sitges, Spain
M Staes,1 E Van Craenenbroeck,1 Hans Vermeiren,1 Jorge Villacian,1 Lee Bacheler,2 Marie-Pierre de BŽthune,3 Sandra De Meyer,3 Gaston Picchio4
1Virco BVBA, Mechelen, Belgium; 2VircoLab, Inc., Durham, NC, USA; 3Tibotec BVBA, Mechelen, Belgium; 4VircoLab, Inc., Bridgewater, NJ, USA
AUTHOR CONCLUSIONS
These results confirm and extend, using a large number of recent routine samples, the suggestion that TMC114 has a high genetic barrier to the development of resistance, and that HIV-1 isolates with high (MinR; FC>CCO2) and intermediate (RR; CCO1< FC≦CCO2) levels of resistance to individual or multiple PIs remain fully susceptible (MaxR; FC≦CCO1) to TMC114.
The use of fold-change information in combination with two clinical cut-offs provides an accurate and refined classification of susceptibility to TMC114. In addition, the strong association observed between TMC114 predicted phenotypic susceptibility and the current TMC114-specifi c mutational count (11I, 32I, 33F, 47V, 50V, 54L or 54M, 73S, 76V, 84V, 89V) suggests that the latter could also provide information to determine susceptibility to this protease inhibitor in clinical practice.
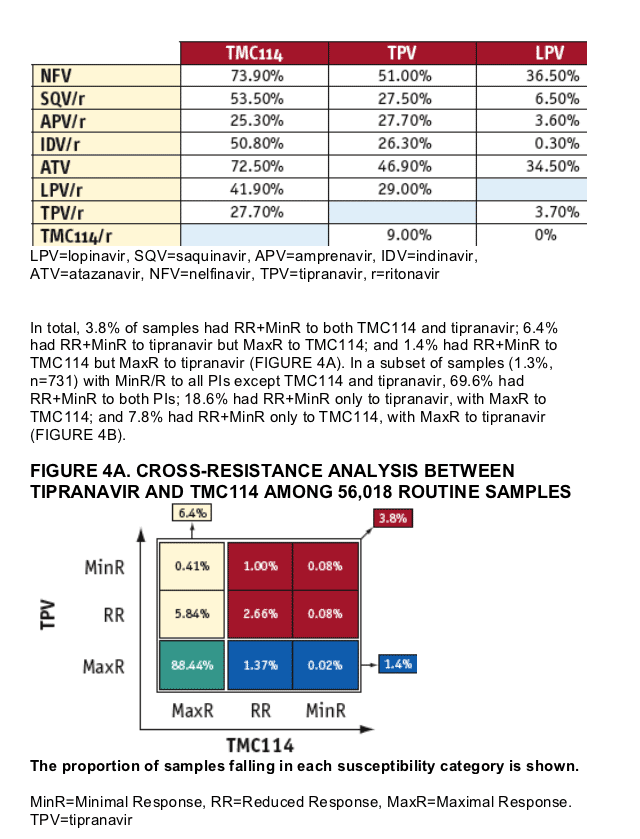
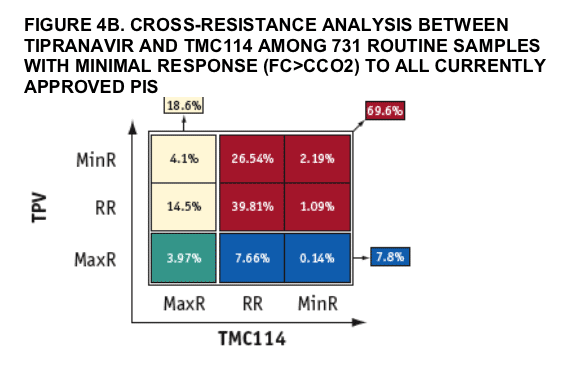
Some significant level of cross-resistance between TMC114 and tipranavir was observed among the samples analyzed. However, samples retaining full susceptibility (MaxR; FC≦CCO1) to either drug while showing resistance (MinR; FC>CCO2 or RR; CCO1< FC≦CCO2) to the other one were also identified.
The conclusions derived from this study around TMC114 and tipranavir cross-resistance should be taken cautiously since the samples analyzed came from patients with little exposure to tipranavir and no exposure to TMC114.
BACKGROUND
Previous studies have shown that TMC114 (darunavir) exhibits potent in-vitro and in-vivo activity against protease inhibitor (PI)-resistant HIV-1 isolates.
Determining the baseline susceptibility that an HIV-1 isolate exhibits to TMC114 could be useful to determine the likelihood of treatment success when using this drug in combination regimens.
This study evaluates, in a large set of recent routine clinical samples with different levels of PI resistance, two approaches to determine HIV-1 susceptibility to TMC114; a) predicted phenotype (VirtualPhenotype-LM), b) TMC114-specific genotypic score (DE MEYER, AT AL. POSTER #73).
METHODS
Fold changes in EC50 (FC) for TMC114 and other PIs were predicted using multiple linear regression models (VirtualPhenotype-LM) (FOR DETAILS SEE WINTERS, ET AL. POSTER #160) on 56,018 sequences submitted to Virco for routine resistance analysis in 2004Đ2005.
FC values were interpreted using vircoTYPE HIV-1 v4.0.00-specific clinical cut-offs (CCOs) for seven PIs; a biological cut-off (BCO) was used for atazanavir (Table 1). For TMC114, a CCO1=3.4 and CCO2=96.9 were used (FOR DETAILS SEE WINTERS, ET AL. POSTER #160). Two categories (susceptible [S], FC≦BCO and resistant [R], FC>BCO), or three categories (maximal response (MaxR), FC≦CCO1; reduced response (RR), CCO1< FC≦CCO2; minimal response (MinR), FC>CCO2) of PI susceptibility were defined by applying one BCO or two CCOs, respectively.
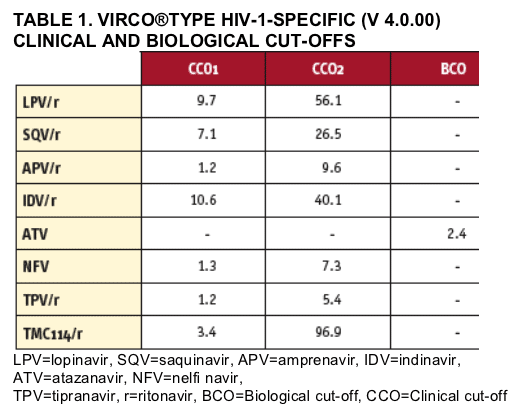
Genotypic susceptibility was determined based on the presence of 0, 1, 2, 3, 4, or ≥5 TMC114-specifi c mutations. Isolates containing ≥3 TMC114Đspecific mutations were classified as having decreased susceptibility to TMC114 (FOR DETAILS SEE DE MEYER, AT AL. POSTER #73). The TMC114-specific genotypic count is composed of the following mutations: 11I, 32I, 33F, 47V, 50V, 54L or 54M, 73S, 76V, 84V, 89V. All mutations included in the TMC114-specifi c genotypic count were identified by Virco's linear model as significant in predicting TMC114 FC (FOR DETAILS SEE WINTERS, ET AL. POSTER #160).
RESULTS
Of 56,018 samples, 34% showed decreased susceptibility (RR+MinR) to at least one PI (nelfi navir). When compared with other PIs, TMC114 exhibited the lowest (0.2%) and second lowest (5.0%) proportion of samples with MinR and RR, respectively, followed by tipranavir in the MinR category (1.5%) (TABLE 2). The lowest (4.9%) proportion of samples in the RR category was observed for saquinavir. We further compared, on the same set of samples, TMC114 susceptibility based on phenotypic and genotypic analyses. Results are summarized on FIGURE 1A. An increase in the TMC114-specifi c mutational count was associated with a progressive decrease in phenotypic susceptibility to
TMC114. This is also illustrated in FIGURE 1B that shows the distribution of TMC114 FC values with respect to the TMC114-specific mutational count. A wide variation in FC values was observed within each TMC114-specific mutational count category. Out of 56,018 routine samples, 98.4% had a TMC114-specific mutational count ≦2.
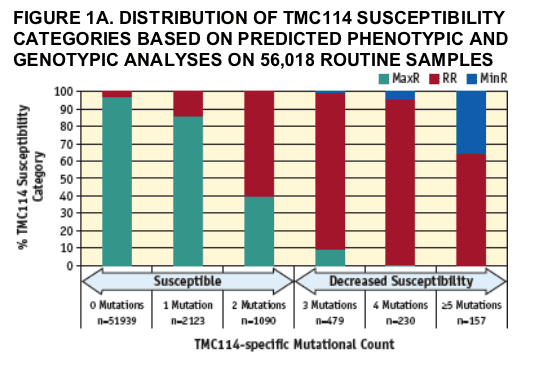
The arrows denote the two susceptibility categories defined by the TMC114-specific mutational count (DE MEYER, AT AL. POSTER #73); Susceptible ≦2 TMC114-specifi c mutations, Decreased Susceptibility ≥3 TMC114-specific mutations. MinR=Minimal Response, RR=Reduced Response, MaxR=Maximal Response.
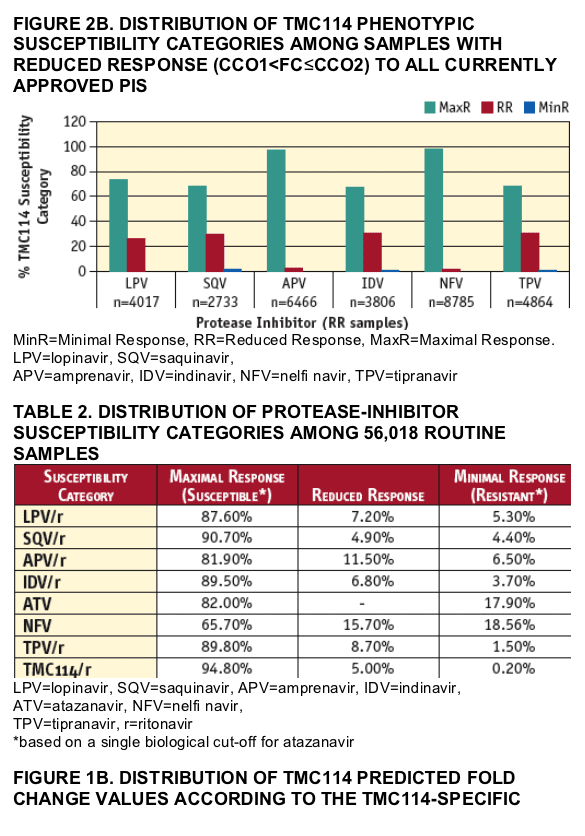
TMC114 susceptibility was also studied in a subset of samples with MinR or RR to all currently approved PIs. Results are summarized in FIGURES 2A and 2B. Susceptibility to TMC114 (FIGURE 2A) scored as MaxR in 50.8%, 53.5%, 25.3%, 41.9%, 27.7%, 73.9%, and 72.5% of samples with MinR or R to indinavir (n=2,064), saquinavir (n=2,471), amprenavir (n=3,645), lopinavir (n=2,947), tipranavir (n=836), nelfi navir (n=10,401) and atazanavir (n=10,076), respectively. In addition, susceptibility to TMC114 (FIGURE 2B) scored as MaxR in 67.5%, 68.0%, 97.0%, 73.3%, 68.4%, 97.7% of samples with RR to indinavir (n=3,806), saquinavir (n=2,733), amprenavir (n=6,466), lopinavir (n=4,017), tipranavir (n=4,864), nelfinavir (n=8,785), respectively.
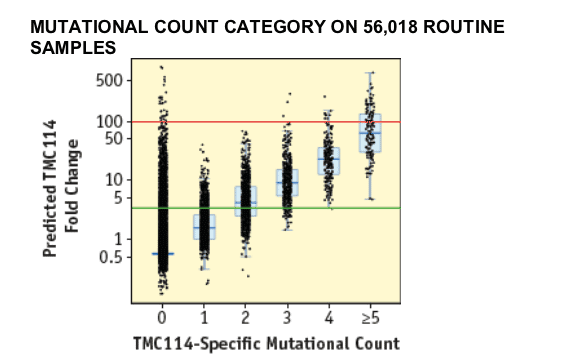
The black dots represent the actual predicted FC values per category of TMC114-specific mutational count carrying 0 to ł5 mutations from 11I, 32I, 33F, 47V, 50V, 54L or 54M, 73S, 76V, 84V, 89V. The grey boxes represent the inter-quartile range (IQR) from 25th to 75th percentile FC value while the horizontal grey line in the box represents the median FC value. The whiskers extend to the most extreme data point within 1.5x the IQR from the box. The green horizontal line represents CCO1 (3.4) and the red horizontal line CCO2 (96.9) for TMC114, respectively.
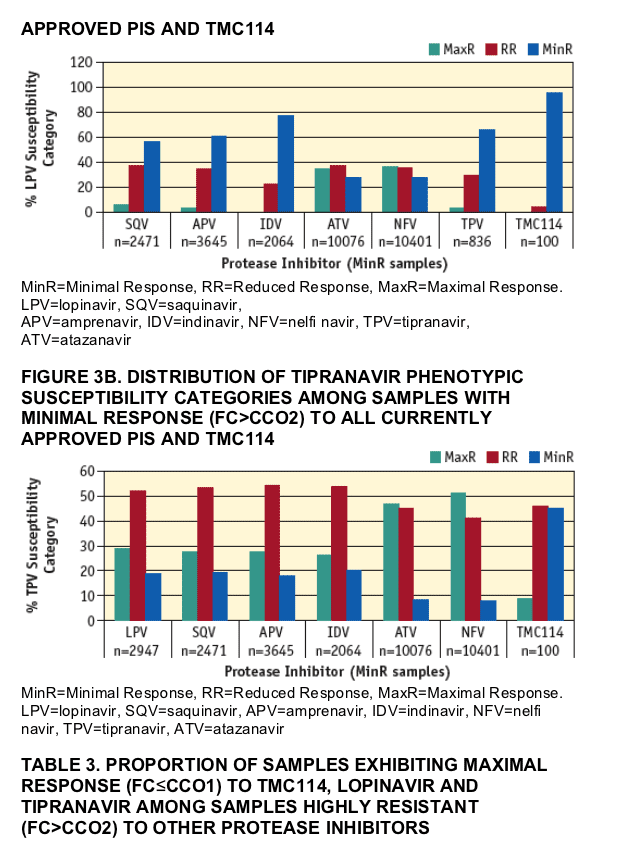
Lopinavir and tipranavir susceptibility was studied in the same subset of samples with MinR and R to all currently approved PIs. Results are summarized in FIGURES 3A and 3B. Susceptibility to lopinavir (FIGURE 3A) scored as MaxR in 0.3%, 6.5%, 3.6%, 3.7%, 36.5%, 0% and 34.5% of samples with MinR or R to indinavir (n=2,064), saquinavir (n=2,471), amprenavir (n=3,645), tipranavir (n=836), nelfi navir (n=10,401), TMC114 (n=100) and atazanavir (n=10,076), respectively. Susceptibility to tipranavir (FIGURE 3B) scored as MaxR in 26.3%, 27.5%, 27.7%, 29.0%, 51.0%, 9.0% and 46.9% of samples with MinR or R to indinavir (n=2,064), saquinavir (n=2,471), amprenavir (n=3,645), lopinavir (2,947), nelfi navir (n=10,401), TMC114 (n=100) and atazanavir (n=10,076), respectively. The proportion of samples exhibiting MaxR to TMC114, lopinavir and tipranavir among this subset of highly PI-resistant samples is summarized in TABLE 3.
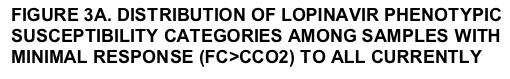
|
| |
|
 |
 |
|
|