 |
 |
 |
| |
Development of virco®TYPE Resistance Analysis,
including Clinical Cut-offs, for TMC114
|
| |
| |
Reported by Jules Levin
poster 160
XV Intl HIV Drug Resistance Workshop
Sitges, Spain, June 2006
B Winters,1 H Vermeiren,1 E Van Craenenbroeck,1 P Lecocq,1 T Vangeneugden,2 M-P de Bethune,2 L Bacheler3
1Virco BVBA, Mechelen, Belgium; 2Tibotec BVBA, Mechelen, Belgium; 3VircoLab, Inc., Durham, NC, USA
Background
TMC114 (darunavir ) , a potent inhibitor of a wide range of protease
inhibitor-resistant strains of HIV-1, has demonstrated significant clinical
activity in treatment-experienced patients.
Evaluation of the clinical impact of viral resistance in such populations is a complex and evolving process.
Methods
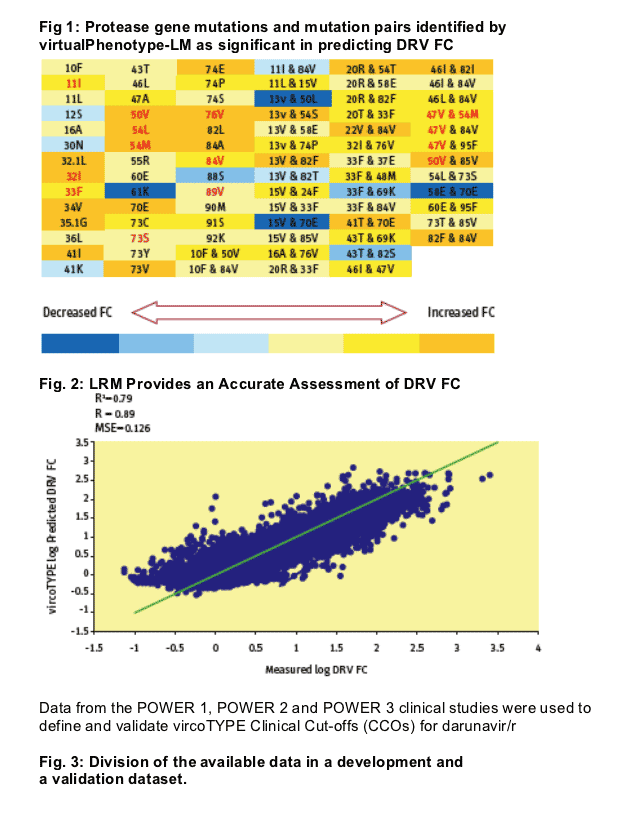
Based on >10,000 clinical isolates with both drug susceptibility phenotypes (Antivirogram®) and viral genotypes, a multiple linear regression model was developed to predict DRV fold change in IC50 (FC) from the viral genotype (vircoTYPE 4.0.00).
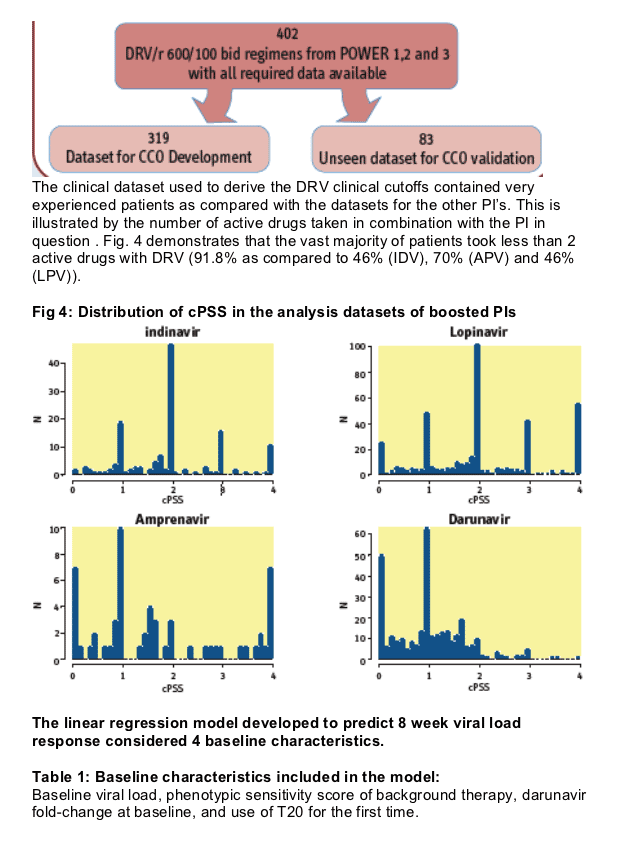
Using data from POWER 1, 2 and 3, a separate linear regression model
was developed to predict 8-week change in viral load on regimens
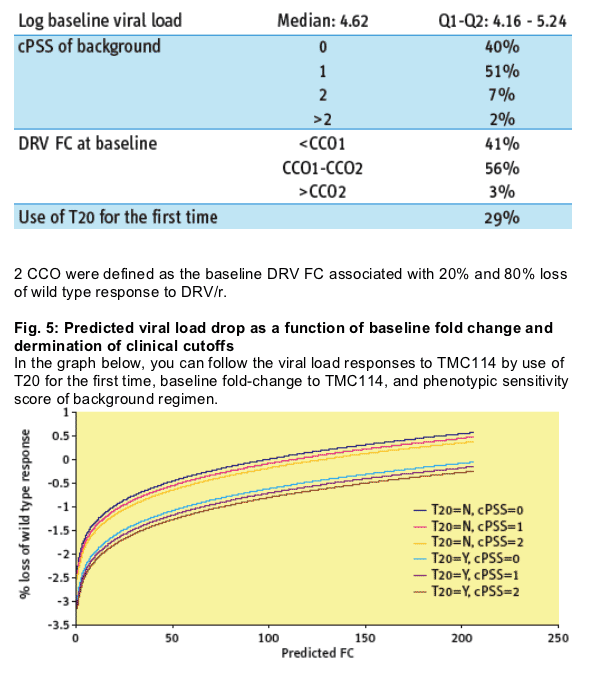
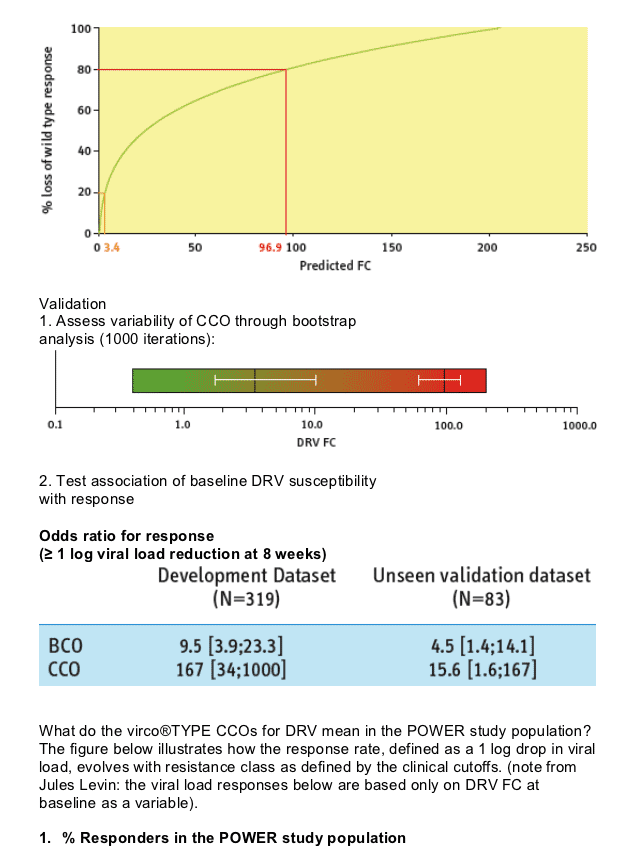
containing ritonavir-boosted DRV (DRV/r). 319 and 83 DRV/r containing regimens were used to define and validate two clinical cut-offs (CCO) corresponding to predicted DRV FC values associated with a 20% or 80% loss of the DRV/r response predicted for subjects infected with wild type strains.
RESULTS
A Linear Regression Model (vircoTYPE HIV-1 v 4.0.00) was developed to predict DRV drug susceptibility from the viral genotype based on >10000 viral genotypes with DRV phenotypes in Virco's databases.
Author Conclusions
virco®TYPE resistance analysis integrates complex interactions among multiple protease gene mutations to provide a quantitative prediction of TMC114 drug susceptibility, which can be interpreted utilizing CCO.
Since these CCO were defined utilizing the POWER studies, their relevance for subjects with different baseline characteristics is not known.
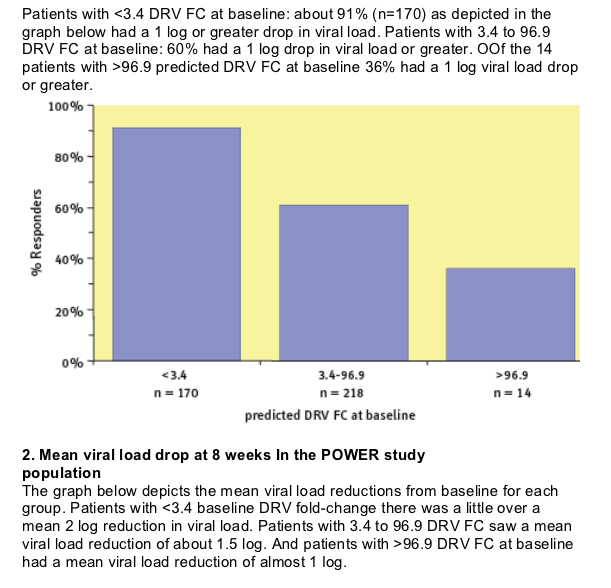
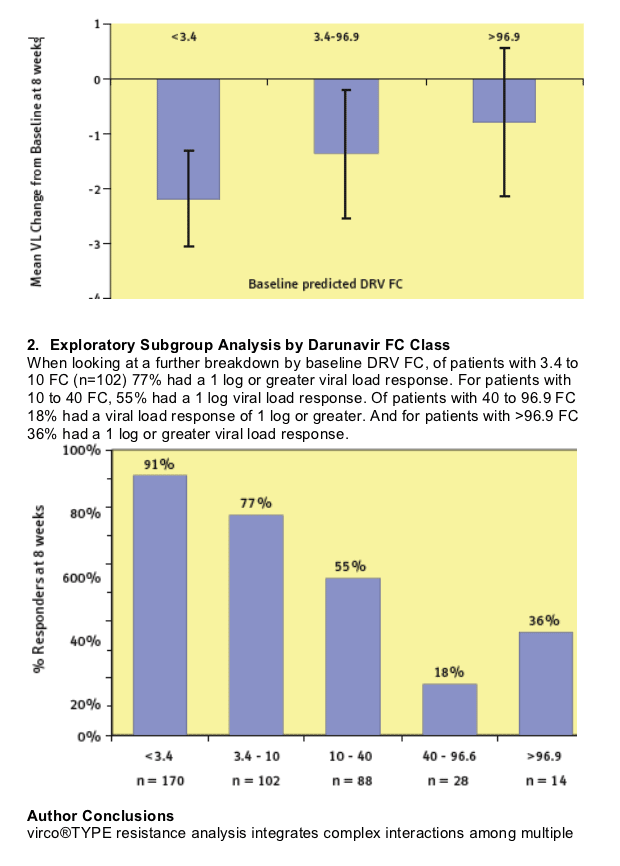
In the POWER studies, 8-week virologic response declined as baseline
predicted DRV FC increased.
However, darunavir/r maintained considerable activity even for patients with a predicted fold change higher than the lower clinical cutoff.
|
| |
|
 |
 |
|
|