 |
 |
 |
| |
Limb Fat Loss: nelfinavir, efavirenz, nukes
|
| |
| |
Reported by Jules Levin
8th Intl Workshop on Adverse Drug Reactions and Lipodystrophy
San Francisco, Sept 24-26, 2006
Here are some brief reports on what was interesting today at the Lipodysytrophy Workshop. More in-depth reporting will follow. There was a talk on inflammation in adipose tissue that I thought was interesting. K Clements said in metabolic diseases a moderate increase of inflammatory factors has been well described. Accompanying this so-called 'low-grade systemic inflammation', it was recently discovered that inflammatory cells (macrophage, lymphocytes) can infiltrate the white adipose tissue. These adipose tissue cells produce a wide panel of inflammatory and immune molecules, some of them being secreted. Adipose-produced molecules play distinct and sometimes overlapping roles in a variety of functions, including energy homeostasis, glucose and lipid metabolism, inflammation, and vascular function. Peripheral fat wasting and visceral fat distribution in HIV are associated with increased inflammatory factors in adipose tissue and with macrophage infiltration.
"DEXA outcomes in ART-naive subjects receiving nelfinavir or efavirenz or both plus dual nucleosides: long-term as treated results from A5005s"
"...authors concluded: over 144 weeks, AZT/3TC continued to be superior to ddi/d4T with regards to limb fat loss. The combination of AZT/3TC/efavirenz had greater trunk fat increases, but showed no overall pattern suggesting limb fat loss over time and was significantly superior to the pooled AZT/3TC/nelfinavir (with and without efavirenz) group".
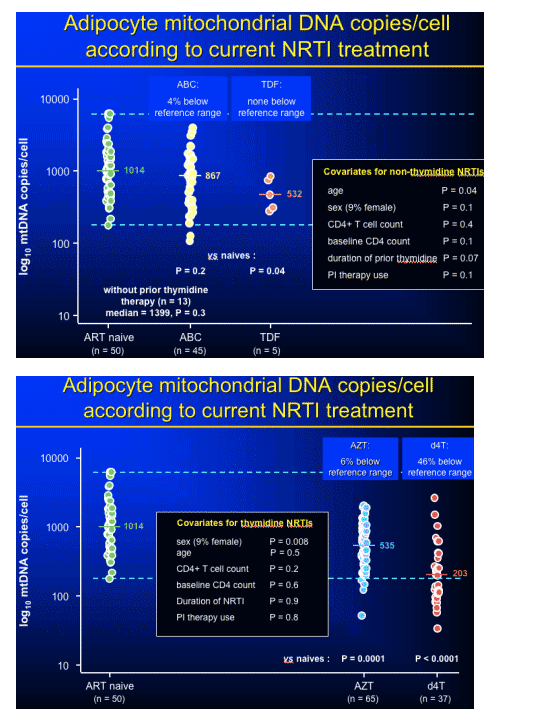
Comments from Jules Levin: Patients taking AZT/3TC showed no limb fat loss at week 144 compared to baseline, but there were a lot of patient dropouts (discontinued therapy). So, it is not clear that there really was no limb fat loss. In addition, Emma Hammond from the Australian group (David Nolan) reported additional data from previous reports that AZT & d4T decreased mtDNA (subcutaneous fat biopsies) and this correclated with fat loss measured by DEXA. There was no significant mtDNA depletion associated with abacavir. For patients who remained on nelfinavir throughout the study there was a significant loss in limb fat. For patients remaining on efavirenz there was no loss of limb fat compared to baseline. Although limb fat increased from baseline to week 16 and then decreased back to baseline by week 48 & stayed there through week 144. Dube said, after adjusting for NRTI backbone there was an additional decrease in limb fat from week 32 to week 144 of 8.7% per year in the combined NFV & NFV/EFV groups compared to EFV alone MMANOVA p=0.03); limb fat decreased from week 32 to 144 on nelfinavir by 7.9% per year (p=0.02), but increased slightly in the EFV group (+2.7% per year).
Mike Dube reported today on DEXA outcomes in a substudy of ACTG 384. DEXA scans were performed every 16 weeks on a subset of 175 ART-naive patients who were randomized to receive nelfinavir, efavirenz, or both, combined with AZT+3TC or ddi+d4T. Subjects with any available data after baseline up to week 144 contributed to this as-treated analysis. 157 patients had baseline DEXA. 65 had baseline & week 144 DEXA. 20% still on original ART at week 144. So a lot of patients dropped off the followup due to therapy switches.
RESULTS
Limb fat increased similarly in all treatment arms during the first 32 weeks. 65 subjects had week 144 DEXA. Among 13 subjects who continued to receive ddI+d4T at week 144, the median change from baseline in limb fat was -32.5% (p=0.04 from baseline) and among 19 subjects on AZT+3TC, the median change was +1.4% (p=1.0 from baseline).
After week 32, limb fat changed by -1.7% per year for AZT/3TC and -19.0% per year for ddI+d4T (MMANOVA p<0.0001).
Among 7 subjects receiving nelfinavir without efavirenz at week 144, the median change from baseline limb fat was -23.8%; among 14 receiving both nelfinavir+efavirenz -21.4% (p=0.05 from baseline for pooled data from all nelfinavir recipients). and among 11 subjects on efavirenz without nelfinavir, the median change was +2.4% (p=0.90 from baseline).
Focusing on subjects receiving AZT/3TC, after week 32 limb fat changed by +2.7% per year with efavirenz and -7.9% per year for the combined nelfinavir and nelfinavir+efavirenz group (MMANOVE p=0.03).
Trunk fat increased similarly in all treatment arms during the first 32 weeks, tending to remain above baseline over time. There were marginal differences between NRTIs after week 32 (MMANOVA p=0.05), with trunk fat changes +7.0% for AZT/3TC and -3.4% per year for ddI/d4T. Among those subjects receiving AZT+3TC, after week 32 trunk fat changed by +18.9% per year with efavirenz and -3.5% per year for the combined nelfinavir and nelfinavir+efavirenz group (MMANOVA p<0.0001).
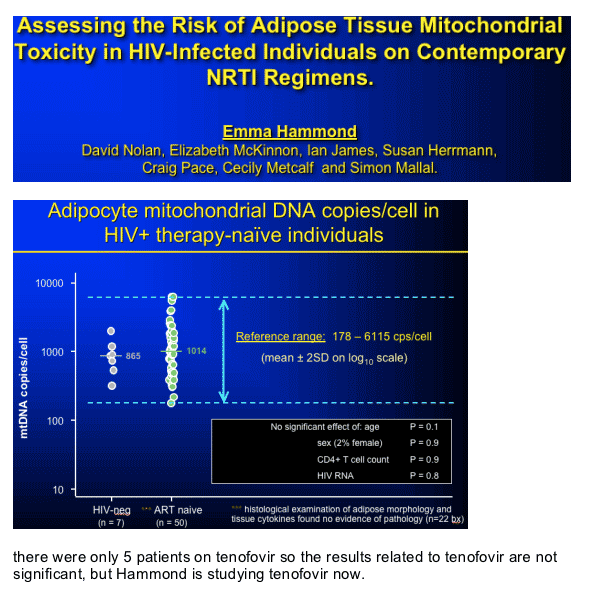
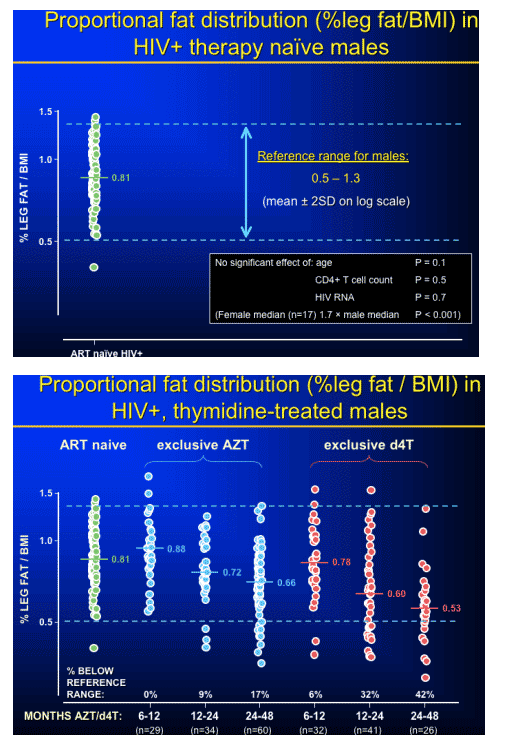
mtDNA changes in patients on AZT, d4T, & abacavir compared to controls; and correlation with DEXA changes: %leg fat/BMI, presented by Emma Hammond, Royal Perth Hospital, Perth, Australia
Questions study asked included:
what is the normal range of mtDNA values among therapy naive individuals, and what is the variation on specific therapy.
More specifically, does variation, in the level of depletion within groups on therapy predict lipoatrophy onset or severity.


|
| |
|
 |
 |
|
|