 |
 |
 |
| |
Switching to lopinavir/ritonavir (LPV/r) Tablets once daily (TAB-QD) from soft-gel capsule dosed BID/QD (SGC-BID/SGC-QD) led to significant improvements in tolerability, diarrhea, antidiarrheal medication use, and satisfaction
|
| |
| |
CORRECTED VERSION
Reported by Jules Levin
8th Intl Workshop on Adverse Drug Reactions & Lipodystrophy in HIV
S Schrader1,SK Chuck2, LW Rahn2, KG Emrich2, PS Parekh21The Schrader Clinic, Houston, TX, USA,2Abbott Laboratories, Abbott Park, IL, USA; poster 83
AUTHOR CONCLUSIONS
In this US survey of 41 HIV-infected patients, significant improvements were reported after switching from LPV/r SGC dosed BID/QD to Tablets dosed QD.
-- 80% of respondents indicated no or rare antidiarrheal use
-- Differences in GI side effects seen with SGC dosed QD vs. BID appear to be lessened with the LPV/r Tablet formulation
-- Significant improvements in satisfaction for 38% of respondents
-- Significant improvements in overall tolerability for 25% of respondents
-- Adherence improved from 93% to 97% and 15% more respondents had no missed doses after switching from SGC-BID to Tablets dosed QD
The LPV/r Tablet benefits most often cited by respondents were:
-- I can take it just once a day (75%), I take fewer pills (73%), I don't have to take it with food (68%), I don't have to refrigerate it (65%)
These results suggest that LPV/r Tablets dosed QD provides multiple benefits relative to SGC with patients valuing QD, fewer pills, and no food or refrigeration requirements. Additional assessments of QD dosing of LPV/r Tablet's tolerability profile are warranted.
BACKGROUND
Kaletra (lopinavir/ritonavir, LPV/r) Tablets were FDA approved in October 2005.
Short-term results in HIV-negative, healthy volunteers suggest improved tolerability, but data in HIV-infected patients have not been previously reported.
[Klein C, et al. EACS, 2005, PE4.3/2]
Compared to twice daily (BID) dosing, once daily (QD) dosing of LPV/r Soft Gel Capsule (SGC) was associated with an increased rate of Grade 3+ diarrhea from 5% vs. 16%. [Johnson MA, et al. JAIDS. 2006]
Features of LPV/r Tablets compared to Soft Gel Capsule (SGC)
[Kaletra US Prescribing Info. 10/05]
-Based on novel Melt-Extrusiontechnology
-No oleic acid or sorbitol
-Contains 200 mg of lopinavir and 50 mg of ritonavir
-Daily pill count decreased from 6 to 4 for same daily dose of 800/200 mg
-No refrigeration; no need for dosing with food-Less pharmacokinetic
variability
STUDY OBJECTIVES
To assess patient self-reported differences between LPV/r SGC dosed BID (SGC-BID) or QD (SGC-QD) and LPV/r Tablet formulation dosed QD (TAB-QD).
Satisfaction
Tolerability
-Overall
-Frequency and severity of select adverse effects
-Diarrhea & antidiarrheal use
Adherence
-Missed doses, fewer pills, food requirement with SGC
-Reasons for missed doses
Benefits
Quality of life
To determine patient preference between LPV/r SGC-BID or SGC-QD and LPV/r TAB-QD.
METHODS - Survey Design
Self-reported, anonymous, multiple-choice survey in English & Spanish
Addresses satisfaction, overall tolerability, adverse effects, adherence, perceived benefits, formulation preference, and quality of life
SGC and Tablet surveys had identical questions with 4 additional comparative questions (SGC vs. Tablets) in the Tablet survey
Respondents were asked to think back over the last 4 weeks and indicate in a typical week the frequency & severity of side effects
Adherence was reported on based on the last week of dosing
Questions written at grade 6 level
METHODS - Survey Distribution
52 out of 65 US physicians contacted distributed surveys to patients; a small payment to physicians were made for efforts related to distribution and handling of surveys with a maximum of 25 patients per site allowed; patients received no compensation
Physicians provided surveys to the patients while on LPV/r SGC and LPV/r Tablets dosed at 400/100mg BID after a minimum of 4 weeks on each formulation.
Patients completed the surveys in waiting area at their routine scheduled visits.
Patient privacy was maintained by having patients seal completedsurveys into envelopes prior to providing survey to clinic staff for mailing to research company managing the project.
October 2005 through May 2006
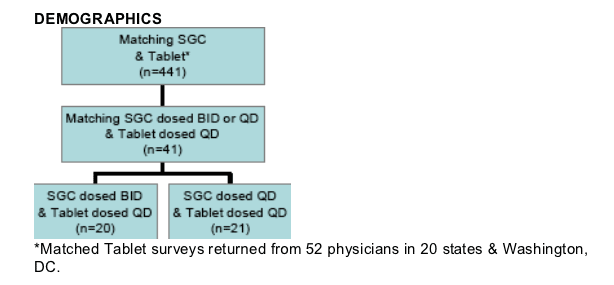
The 41 respondents were mostly males (76%) with diverse ethnicity.
- 41% Black non-hispanic
- 10% Hispanic
- 49% White non-hispanic
Majority of respondents were > 35 years old:
- 45yrs (34%), 35-44yrs (39%), < 35yrs (27%)
Duration of antiretroviral therapy:
- 5yrs (41%), 1-5yrs (49%), < 1yr (10%)
- The SGC-BID and SGC-QD groups had similar durations of therapy.
Duration of LPV/r therapy:
1 year LPV/r SGC (80%) and <3 months LPV/r Tablet (81%)
RESULTS
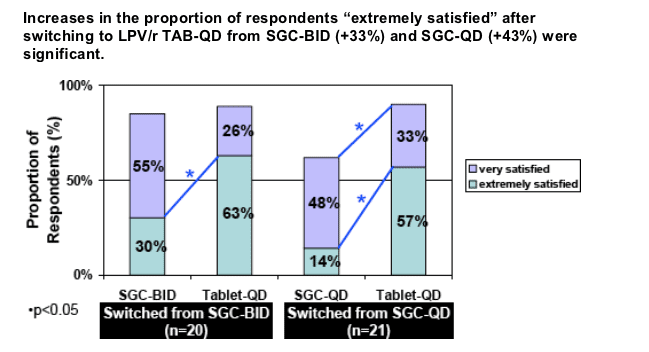
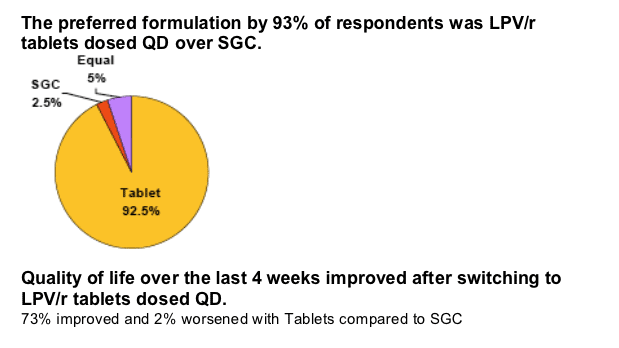
A similar proportion of respondents were "extremely" satisfied with TAB-QD, independent of switching from SGC-BID (63%) or SGC-QD (57%).
Overall 90% of respondents (n=41) were "very" or "extremely" satisfied on Tablets dosed QD (vs. 73% on SGC).
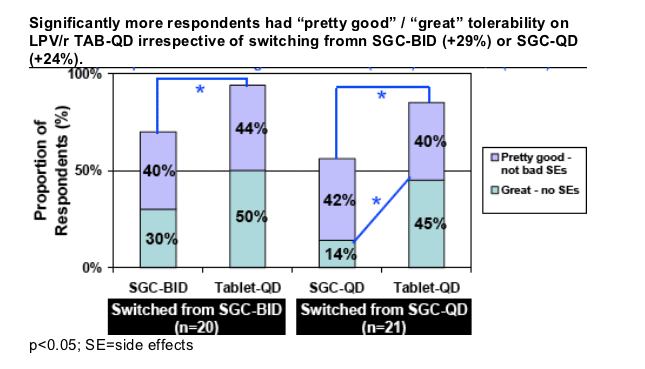
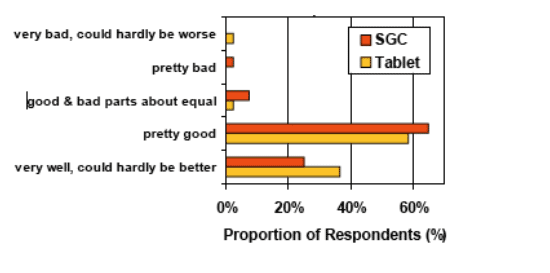
Overall 90% of respondents (n=41) felt they had "pretty good" or "great" tolerability on Tablets dosed QD (vs. 64% on SGC).
Greater improvements were noted when switching from SGC-QD vs. SGC-BID.
Respondents had improvements in diarrhea prevalence, severity & antidiarrheal use after switching to LPV/r Tablets dosed QD
Overall, 78% reported no diarrhea or had improvements in diarrhea
--Improvements were greater when switched from SGC-QD vs. SGC-BID
Proportion of respondents reporting no diarrhea after switch to TAB-QD
--SGC-BID (n=20): doubled (p<0.10)
--SGC-QD (n=21): more than quadrupled (p<0.05)
No reports of "severe"diarrhea on TAB-QD (vs. 8% for SGC, p<0.10)
Antidiarrheal use of 1+ times per wk decreased from 44% to 20% (p<0.05)
--80% of respondents indicated no or rare antidiarrheal use (vs. 56% for SGC)
Bloating, pain, or gas in stomach & nausea were decreased with no "severe" episodes on LPV/r Tablets dosed QD
Bloating, pain, or gas in stomach prevalence decreased and those who experienced episodes had diminished frequency on Tablets dosed QD
Nausea prevalence decreased from 27% to 5% (p<0.05)
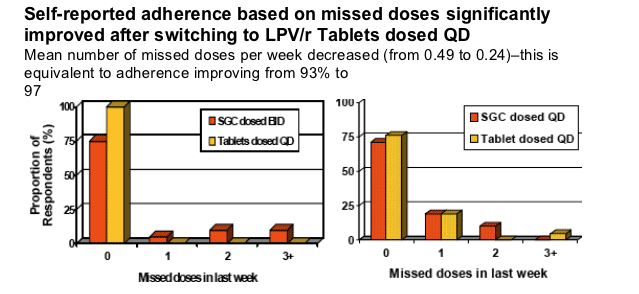
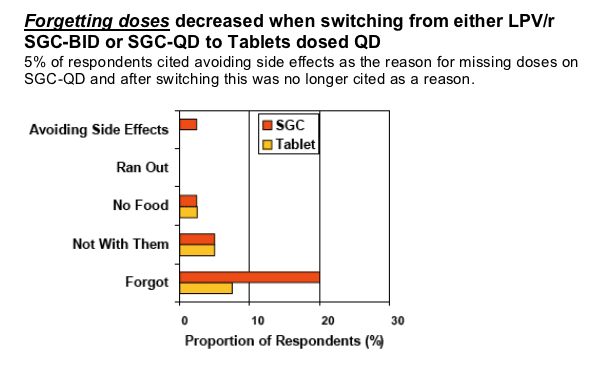
More respondents (n=41) indicated "not missing doses"(i.e. 100% of doses were taken) in the last week after switching to TAB-QD when measured with yes/no response (+10%) or indicating the number of missed doses (+20%)
--75% on SGC-BID vs. 90% on TAB-QD reported no missed dose
LPV/r Tablet benefits cited by respondents related to refrigeration, pill count, and food requirement
Respondents cited the following as benefits they "liked"
- I can take it just once a day (75%)
- I take fewer pills (73%)
- I don't have to take it with food (68%)
- I don't have to refrigerate it (65%)
68% of respondents cited the lack of dietary restriction as a benefit. This is only partially explained by the 34% of respondents non-adherence to LPV/r SGC's food requirement.
-- 34% of respondents indicated at least 1 dose of LPV/r SGC was taken without food in the last week
-- On average of 17% (1.6/7) of QD doses and 22% (2.4/14) of BID doses of LPV/r SGC were taken without food.
|
| |
|
 |
 |
|
|