 |
 |
 |
| |
Long-Term Follow-Up of Entecavir Treated Protocol-Defined 'Non-Responders' in Rollover Study ETV-901
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
M Sherman1, M Rizzetto2, CL Lai3, YF Liaw4, A Gadano5, I Jacobson6, E Schiff7, J Yang8, R Colonno8, B Kreter9
1Toronto General Hospital, Toronto, Canada; 2Ospedale Molinette divisione di gastroenterologia, Turin, Italy; 3Queen Mary Hospital, The University of Hong Kong, Hong Kong; 4Linkou Chang Gung Memorial Hospital, Taoyuan, Taiwan; 5Hospital Italiano, Buenos Aires, Argentina;
6Department of Medicine, Weill Medical College of Cornell University, New York, USA; 7Center for Liver Diseases, University of Miami School of Medicine, Miami, USA; 8Bristol-Myers Squibb, Wallingford, CT, USA; 9Bristol-Myers Squibb, Princeton, NJ, USA
AUTHOR CONCLUSIONS
Fewer than 5% of nucleoside-naive patients treated with entecavir in studies ETV-022 and ETV-027 met protocol-defined criteria of 'Non-Response'
Most 'Non-Responders' who continued to receive ETV treatment in rollover study ETV-901 achieved HBV DNA <300 copies/mL and ALT normalization
4 patients experienced virologic breakthrough during treatment in ETV-901; however, genotypic and phenotypic analysis failed to show evidence of ETV
resistance
Treatment compliance should be evaluated in patients with non-response
Introduction
Entecavir's (ETV) efficacy in nucleoside-naive HBeAg(+) [ETV-022] and
HBeAg(-) [ETV-027] patients with chronic hepatitis B (CHB) has been previously demonstrated1,2
Protocol-defined patient management criteria dictated that 'Non-Responders' in studies ETV-022 and ETV-027 discontinue study therapy
However, 'Non-Responders' who entered rollover study ETV-901 continued to be monitored. Results for patients initially treated with ETV are presented
Methods
Study population
679 nucleoside-naive HBeAg(+) and HBeAg(-) patients were enrolled and treated with 0.5 mg of ETV in studies ETV-022 and ETV-027
Based on protocol-defined patient management criteria, patients who failed to achieve HBV DNA <0.7 MEq/mL by bDNA at Week 48 or during yr-2 were classified as 'Non-Responders' and were to discontinue study therapy
Protocol-defined 'Non-Responders' were offered either continued treatment with entecavir or (at the descretion of the investigator) alternative off-study HBV therapy
Initially, due to ongoing blinding of studies ETV-022 and ETV-027, patients enrolling ETV-901 received a combination of ETV 1 mg and LVD 100 mg a day. Subsequently the protocol was amended for patients to receive monotherapy with ETV 1 mg
Efficacy and safety analyses
HBV DNA, ALT and HBV serology were evaluated during treatment in ETV-022 and ETV-901:
-- HBV DNA was measured by the Roche COBAS Amplicor PCR assay (version 2.0; lower limit of quantifi cation 300 copies/mL)
-- Serology tests were performed in a central laboratory in ETV-022 and in local laboratories in ETV-901
Safety evaluation include incidence of clinical adverse events and laboratory abnormalities
Resistance analyses
Resistance analyses were performed on all patients who entered ETV-901 from ETV-022 with a treatment gap of ≦35 days
Samples from all patients with HBV DNA >300 copies/mL at weeks 48, 96, 144, 192 or End of Dosing were genotyped, and phenotype determined for all emerging substitutions
Samples from all patients who experienced a virologic breakthough (confirmed
or last on-treatment ≥1 log10 increase from nadir) were also phenotyped, including those with no observed genotypic changes
Results
30 out of 679 patients treated with ETV in studies ETV-022 and ETV-027 met
protocol definition of 'Non-Response':
-- 27 patients from ETV-022 (19 at the end of yr-1 and 8 during yr-2)
-- 3 patients from ETV-027 at the end of yr-1
21 'Non-Responders' enrolled into ETV-901:
-- All patients were HBeAg(+) patients from ETV-022
-- 13 were classified as 'Non-Responders' at the end of yr-1 and 8 during yr-2
Baseline characteristics in ETV-022
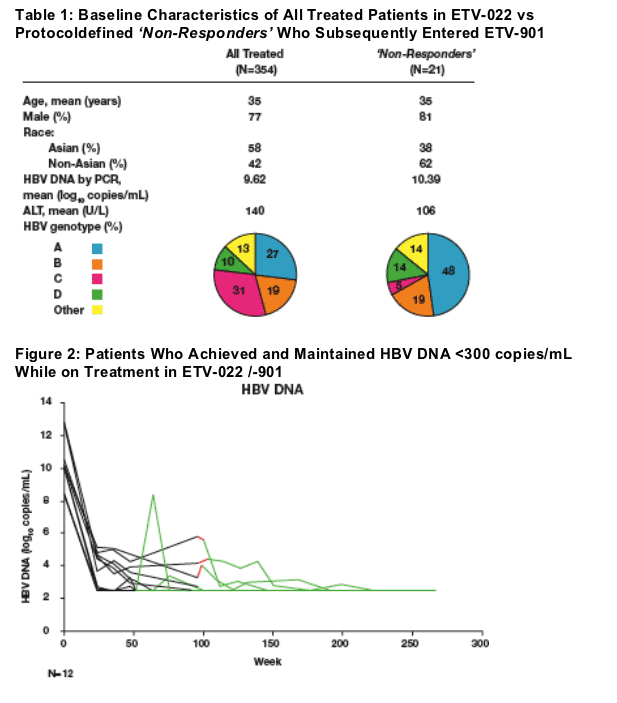
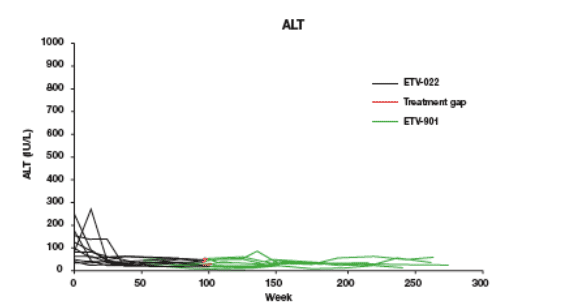
12 protocol-defined 'Non-Responders' achieved and maintained undetectable HBV DNA during ETV treatment:
Mean viral load at ETV-022 baseline = 10.2 log10 copies/mL
The majority of patients achieved and maintained ALT normalization while on treatment
5 patients had HBeAg seroconversion
1 patient had a virologic breakthrough at the last observation in ETV-022, followed by a treatment gap of 29 days between ETV-022 and ETV-901 and then became undetectable when re-treated with ETV during ETV-901
No patients had evidence of genotypic resistance
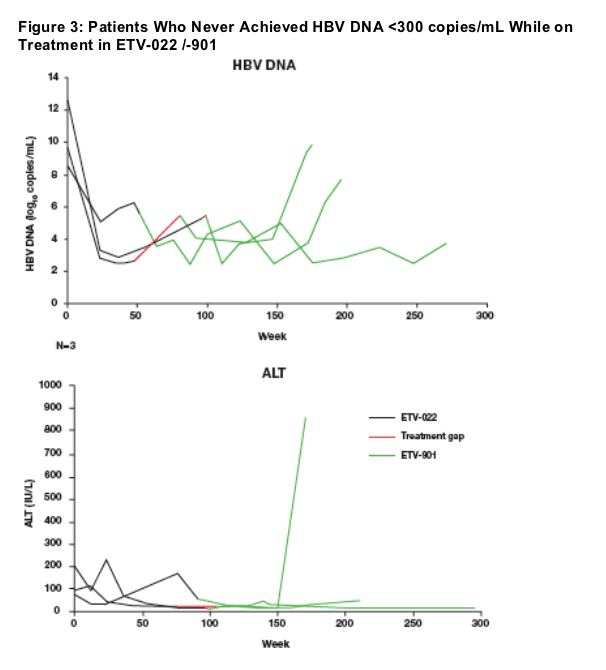
3 protocol-defined 'Non-Responders' achieved but did not maintain undetectable HBV DNA during ETV treatment
Mean viral load at ETV-022 baseline = 10.4 log10 copies/mL
2 patients normalized ALT; 1 patient with virologic breakthrough also developed ALT fl are
2 patients had HBeAg seroconversion
2 patients experience virologic breakthrough
No patients had evidence of genotypic resistance
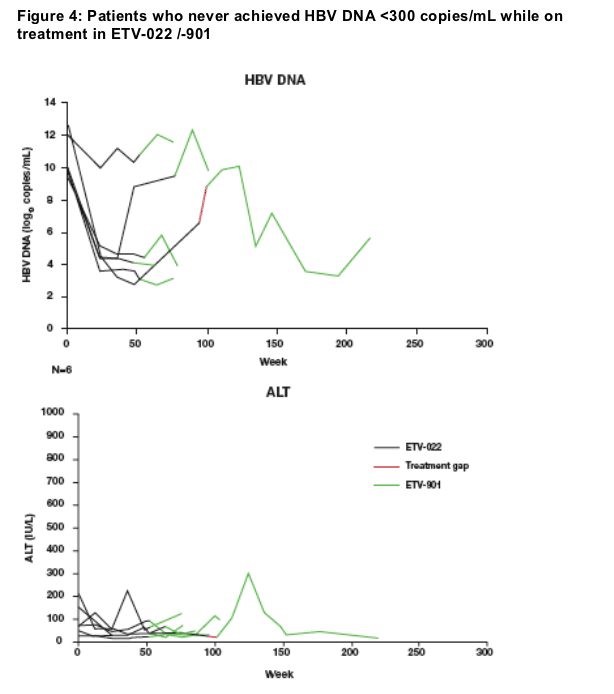
6 protocol-defined 'Non-Responders' failed to achieve undetectable HBV DNA during ETV treatment
Mean viral load at ETV-022 baseline = 10.7 log10 copies/mL
5 patients experienced an initial reduction in HBV DNA of 6.0 log10 copies/mL
ALT levels fluctuated between normal and abnormal in all patients
2 patients experience virologic breakthrough
No patients had evidence of genotypic resistance

References
1. Chang TT, et al. New Engl J Med. 2006; 354(10):1001-1010.
2. Lai CL, et al. New Engl J Med. 2006; 354(10):1011-1020.
|
| |
|
 |
 |
|
|