 |
 |
 |
| |
The DIRECT Trial (Daily-Dose Consensus Interferon and Ribavirin:
Efficacy of Combined Therapy): treatment of non-responders to previous pegylated interferon plus ribavirin: sustained virologic response data
|
| |
| |
Reported by Jules Levin, NATAP
AASLD, Nov 2-6, 2007, Boston, MA
BR Bacon, A Regev, RH Ghalib, G Morelli, KD Rothstein, ML Shiffman, T Hassanein, JMJ Hammond
AUTHOR SUMMARY
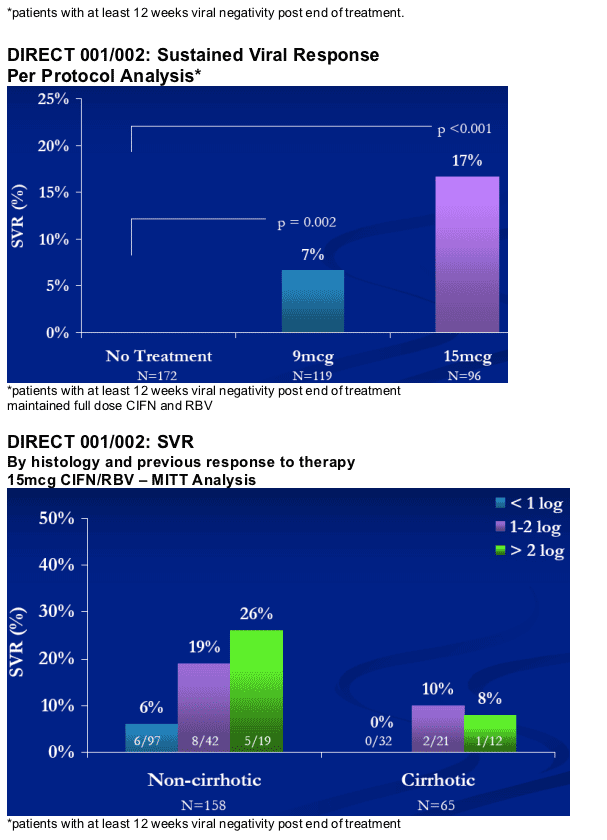
Patients with lower fibrosis stages experienced improved responses with CIFN.
Non-cirrhotic patients who were sensitive to PegIFN/RBV therapy had the best response.
15 mcg group consistently experienced a better response than the 9 mcg group.
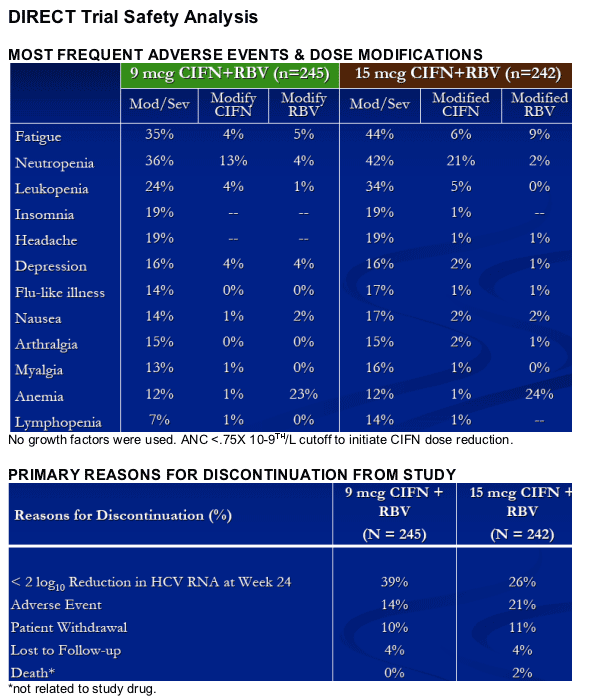
CIFN/RBV had an acceptable tolerability profile.
AUTHOR CONCLUSIONS
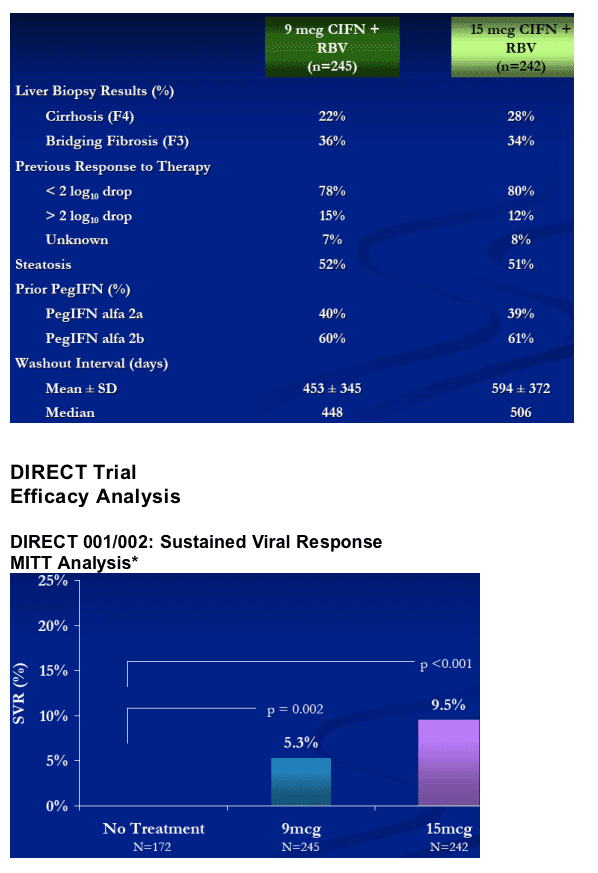
Patients who did not modify their CIFN/RBV dosages achieved up to a 17% SVR.
Approximately 33% of non-ciirhotic patients (15 mcg group) who were sensitive to PegIFN/RBV and did not modify their CIFN/RBV dosages achieved an SVR.
Daily CIFN in combination with RBV was well tolerated at doses up to 15 mcg.
SEE TABLES/GRAPHS BELOW.
Background: Consenus Interferon
10--100 fold more potent in 100 fold more potent in vitro activity than other vitro activity than other interferon alphasinterferon alphas
1 amino acid difference amino acid difference between between IFN _-2_--and and IFN _--2b
19 amino acid difference amino acid difference between between IFN _--2a and and CIFN
18 amino acid difference amino acid difference between between IFN _--2b andandCIFN
DIRECT: Study Design
Randomized, Open Label Study in Patients who did not respond to previous combination therapy with pegylated interferon and ribavirin.
--patients received either 9 mcg CIFN qd plus RBV or 15 mcg qd CIFN plus RBV or no treatment

KEY INCLUSION CRITERIA
Documented compliance during previous PegIFN alfa-2a or 2b plus RBV or 2b regimens (80/80/80)
Patients must have had a documented viral nonresponse to PegIFN alfa-2a or 2b plus RBV defined as:
-- < 2 log10 reduction in HCV RNA from baseline at any time between 12 and 24 weeks of therapy or
-- Detectable HCV RNA at end-of of-treatment (EOT) after completing at least 24 weeks of therapy
All fibrosis stages as determined by a biopsy within 3 years prior to screening
KEY EXCLUSION CRITERIA
History of decompensated liver disease
Patients who prematurely discontinued, interrupted, or dose reduced prior PegIFN/RBV therapy
Depression: major depressive episode within 2 years prior to screening
Patients who had been treated for HCV within 3 months prior to screening
Known HIV infection or chronic HBV infection
KEY STUDY FEATURES
CIFN dose reduction initiated at ANC <.75 x 10-9th/L
-- 15 mcg down to 9 mcg and then to 6 mcg
-- 9 mcg down to 6 mcg
RBV dose reduction at Hb <10 g/dL
-- 1,000-1,200 mg/d to 600 mg/d
Study was not powered to detect significant differences between the 9 mcg and 15 mcg arms
No use of growth factors
|
| |
|
 |
 |
|
|