 |
 |
 |
| |
Effects of tipranavir/ritonavir (TPV/r) on the activity of hepatic and intestinal cytochrome P450 3A4/5 and P-glycoprotein (P-gp): implications for drug interactions
|
| |
| |
Reported by Jules Levin
CROI, Feb 2007, Los Angeles
Manoli Vourvahis1, Julie Dumond1, Kristine Patterson1, Naser Rezk1, Nicole White1, Steve Jennings1, Hsiao Tien1, John P Sabo2, Thomas R MacGregor2, Angela DM Kashuba1
1University of North Carolina, Chapel Hill, NC, USA; 2Boehringer Ingelheim Pharmaceuticals Inc, Ridgefield, CT, USA
AUTHOR DISCUSSION & CONCLUSION
After first dose, TPV/r moderately inhibited hepatic CYP3A4/5 and intestinal P-gp, and potently inhibited intestinal CYP3A4/5. Over time, TPV/r induced CYP3A4/5, and P-gp activity.
At steady-state, overall TPV/r effects were: moderate inhibition of hepatic CYP3A4/5, potent inhibition of intestinal CYP3A4/5, and minimal effects on P-gp activity.
These data demonstrate less of an inhibitory effect on CYP3A and P-gp activity than has been seen historically with RTV and other PIs, and may form the basis for understanding the interaction between TPV/r and amprenavir, lopinavir, and saquinavir.
Neither midazolam nor digoxin had any clinically relevant effect on the steady-state pharmacokinetics of TPV/r as measured by AUC0-12h, Cmax and Cp12h.
Abstract
Background Understanding antiretroviral drug interactions is a complex challenge. Phenotyping allows for a general signal of potentially important drug interactions. This is the first investigation to simultaneously evaluate TPV/rÕs effects on intestinal and hepatic CYP3A4/5 and P-gp under first dose and steady-state conditions.
Methods This open-label, cross-over study was performed in 16 healthy volunteers. Subjects were studied at baseline, and 10 h after a first dose and a steady-state dose of TPV/r 500 mg/200 mg BID. At each visit, subjects received single doses of IV and PO midazolam (CYP3A4/5) and IV and PO digoxin (P-gp). Plasma samples were collected over 48 hours and analyzed by validated LC/MS methods. PK parameters were calculated using a non-compartmental approach (WinNonLinPro 4.0.1). Statistics were performed using SAS. Data are presented as the geometric mean ratio [90% CI] (GMR = PK parameter at first dose or steady-state Ö PK parameter at baseline) or mean±SD.
Results Baseline PK parameters for all subjects (10M, 6F; 13 white, 3 black; 26.6±7.7 yrs; 24.2±3.4 BMI) were within expected values. PK parameter comparisons are summarized below.
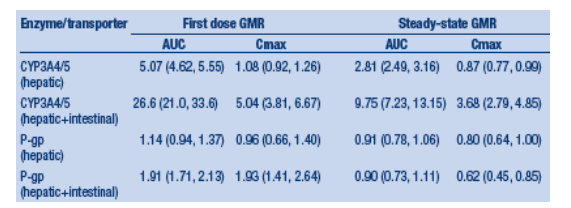
Conclusions After first dose, TPV/r moderately inhibited hepatic CYP3A4/5 and intestinal P-gp, and potently inhibited intestinal CYP3A4/5. Over time, TPV/r induced CYP3A4/5, and P-gp activity. At steady-state, overall TPV/r effects were: moderate inhibition of hepatic CYP3A4/5, potent inhibition of intestinal CYP3A4/5, and minimal effects on P-gp activity. These data demonstrate less of an inhibitory effect on CYP3A and P-gp activity than has been seen historically with ritonavir and other protease inhibitors, and may form the basis for understanding the interaction between TPV/r and amprenavir, lopinavir, and saquinavir. These data specifically, and this approach generally, assists with the understanding and prediction of complex drug-drug interactions.
Introduction
Tipranavir (TPV, Aptivus) is an approved novel protease inhibitor (PI) with potent activity against multiple PI-resistant HIV-1. To achieve effective plasma TPV concentrations and a twice-daily dosing regimen, co-administration of TPV with 200 mg of ritonavir (TPV/r) is essential. Ritonavir (RTV) inhibits hepatic CYP3A, intestinal P-glycoprotein (P-gp) and possibly intestinal CYP3A [1,2]. TPV/r is effective and well tolerated in PI experienced HIV+ patients [3,4].
Using the erythromycin breath test, TPV itself was shown to induce CYP3A, and this effect is reversed with RTV co-administration [1]. Subsequently, TPV/r was shown to decrease amprenavir, lopinavir, and saquinavir exposure by 50-80% [5].
A pharmacokinetic analysis of loperamide, a P-gp substrate, co-administered with TPV/r suggests that TPV/r induces efflux transporters in vivo [6]. Given that only 0.3% of loperamide is absorbed systemically, a more intensive evaluation of P-gp activity was warranted [7].
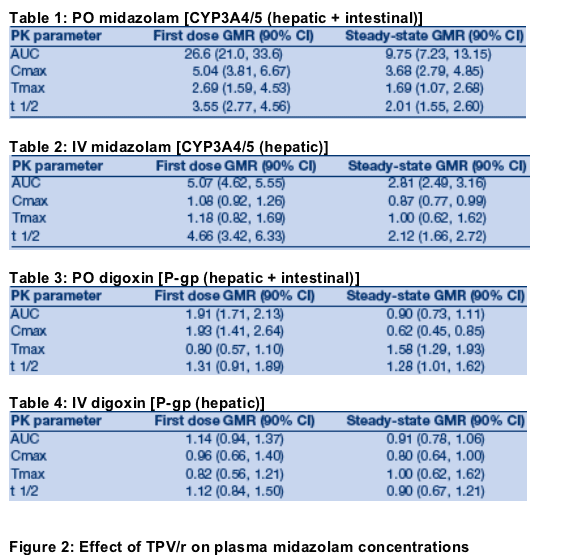
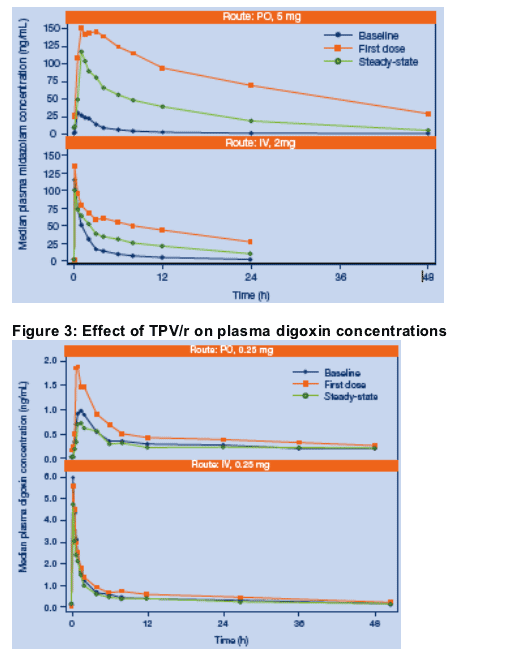
Midazolam (MDZ) and digoxin are FDA recommended phenotyping substrates for CYP3A4/5 and P-gp, respectively [8].
Methods
Study design
Single dose PK of oral MDZ (5 mg), IV MDZ (2 mg), oral digoxin (0.25 mg), and IV digoxin (0.25 mg) were assessed alone, after single dose TPV/r 500 mg/200 mg, and in combination with steady-state TPV/r 500 mg/200 mg BID. Subjects were randomized to receive either oral or IV digoxin under Ôat first dose TPV/r'conditions.

Bioanalytical Validated bioanalytical assays were used to determine the concentrations of TPV and RTV (BASi, West Lafeyette, Indiana, USA) and MDZ and digoxin (UNC CFAR Clinical Pharmacology and Analytical Chemistry Lab, Chapel Hill, North Carolina, USA), in plasma (EDTA anticoagulant) using liquid chromatography/tandem mass spectrometry (LC/MS/MS) and LC/MS technologies. The lower limit of detection was 25.0 ng/mL for TPV and RTV, 0.5 ng/mL for MDZ, and 0.1 ng/mL for digoxin.
Pharmacokinetics Plasma drug concentration-time profiles were analyzed with
WinNonlin Professional v4.0.1 (Pharsight Corporation, Mountain View, California, USA). The geometric mean ratio (GMR) and the associated 90% confidence interval (CI) were derived and evaluated for each primary pharmacokinetic parameter (Tmax, Cmax and AUC) using SAS Release 8 (SAS Institute, Cary, North Carolina, USA). Graphical summaries were produced with S-PLUS Professional 8.0 (Insightful Corporation, Seattle, Washington, USA).
The study was conducted in compliance with the Boehringer Ingelheim ethical study conduct SOPs, the institutional REB guidelines, ICH guidelines and the Declaration of Helsinki 1996.
Demographics
Sixteen healthy volunteers (10 male; 6 female) participated in this study. The mean age and body mass index (BMI) was 26.6 ± 7.7 years and 24.2 ± 3.4, respectively. Twelve (75%) subjects were of white race and four (25%) subjects were of black race.
Results
The steady-state pharmacokinetics of TPV and RTV in this study were consistent with previous studies in healthy volunteers and HIV+ patients [1,3,4].
References
1. MacGregor TR, Sabo JP, Norris SH, et al. Pharmacokinetic characterization of different dose combinations of co-administered tipranavir and ritonavir in healthy volunteers (BI1182.5). HIV Clinical Trials 2004; 5(6): 371-382.
2. Norvir (ritonavir capsules) Product Information. Abbott Laboratories. January, 2006.
3. Cahn, P. and the RESIST 2 Study Team. 24-week data from RESIST-2: Phase 3 study of the efficacy and safety of background therapy plus tipranavir/ritonavir (TPV/r) or optimized ritonavir-boosted standard-of-care (SOC) comparator PI (CPI) in a large randomized multicenter trial in treatment-experienced HIV+ patients. In 7th International Congress on Drug Therapy in HIV Infection. 2004. Glasgow, UK. Abstract PL14.3.
4. Hicks, C. and the RESIST 1 Study Team. RESIST-1: a phase 3, randomized, controlled, open label, multicenter trial comparing tipranavir/ritonavir (TPV/r) to an optimized comparator protease inhibitor/r (CPI/r) regimen in antiretroviral (ARV) experienced patients: 24 week data. in 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). 2004. Washington. Abstract H-1137a.
5. Walmsley S, Leith J, Katlama C et al. Pharmacokinetics and safety of tipranavir/ritonavir (TPV/r) alone or in combination with saquinavir (SQV), amprenavir (APV), or lopinavir (LPV): interim analysis of BI1182.51. XV Intl AIDS Conference, Bangkok. Abstract WeOrB1236.
6. Mukwaya G, MacGregor T, Hoelscher D, et al. Interaction of ritonavir-boosted tipranavir with loperamide does not result in loperamide-associated central neurologic side effects in healthy volunteers. Antimicrob Agents Chemother 2005;49: 4903-4910.
7. Heykants J, Michiels M, Knaeps A, et al. Loperamide (R18553), a novel type of anti-diarrheal agent: Part 5. The pharmacokinetics of loperamide in rats and man. Arneimittelforschung. 1974 Oct;24(10): 1649-53.
8. Food and Drug Administration: FDA Guidance for Industry: Drug interaction studiesÑStudy design, data analysis, and recommendations for dosing and labeling - Draft guidance. Rockville, MD: FDA, Sept 2006.
|
| |
|
 |
 |
|
|