 |
 |
 |
| |
10% Resistance Rate in USA, Most Common Mutations in Naive Newly Diagnosed: CDC Report
|
| |
| |
Reported by Jules Levin
CROI 2007, Los Angeles
Antiretroviral Drug-resistance Mutations and Subtypes in Drug-naive Persons Newly Diagnosed with HIV-1 Infection, US, March 2003 to October 2006
W Wheeler1, K Mahle1, Ulana Bodnar*2, R Kline2, I Hall2, M McKenna2, and the US Variant, Atypical and Resistant HIV Surveillance (VARHS) Group
1Northrop Grumman Corp, Atlanta, GA, US and 2CDC, Atlanta, GA, US
Most Common HIV Drug Mutations Observed in Cases Newly Diagnosed with HIV
K103N:
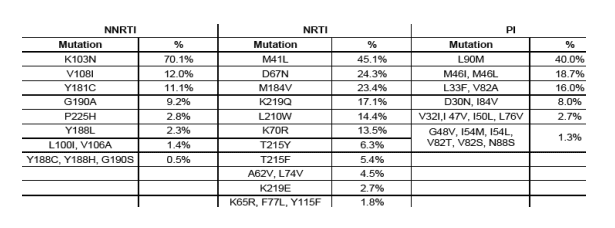
* The following major mutations were not seen (NNRTI): V106M, Y181I, P236L, (NRTI): 69Insert, K70E, V75I, F116Y, Q151M, M184I, (PI): I47A, I50V, V82F, V82L
ABSTRACT
Background: Recent studies indicate that transmission of HIV-1 with antiretroviral drug-resistance mutations may be increasing in the United States and that newly diagnosed persons may have increased risk of treatment failure. Additionally, increased travel and migration may influence the prevalence of HIV subtypes not commonly found in the United States. Using data from the U.S. Variant, Atypical, and Resistant HIV Surveillance system, we estimated the prevalence of antiretroviral drug resistance mutations and the distribution of subtypes.
Methods: In 11 states (409 sites), PR and RT sequence data were identified from specimens drawn from March 2003 to October 2006 from newly diagnosed, confidentially tested, and drug-naive persons (diagnosis dates: January 2003 to October 2006). Major antiretroviral drug resistance mutations were defined according to the International AIDS Society (IAS) -USA guidelines, and subtypes were derived using the Stanford online HIV Drug Resistance Database. These data were merged with demographic and clinical data reported to the national HIV case surveillance system.
Results: Of 3130 specimens suitable for analysis, antiretroviral drug resistance mutations were observed in 327 (10.4%) persons. The range in participating states was 6.3 to 13.0%. Drug-resistance mutations to nucleoside reverse transcriptase inhibitors (NRTI), non-NRTI (NNRTI), and protease inhibitors (PI) were found in 111 (3.6%), 217 (6.9%), and 75 (2.4%) persons, respectively. The predominant mutations were M41L for NRTI (45.1%), K103N for NNRTI (70.1%), and L90M for PI (40.0%).
Multi-drug resistance mutations were noted in 60 (1.9%) persons, ranging from 0 to 0.3% across states, with 44 (1.4%) persons having drug-resistance mutations to at least 1 drug in 2 classes and 16 (0.5%) having 3 class antiretroviral drug resistance mutations. Subtype B was observed in 2971 (94.9%) persons and non-B subtypes or recombinant forms were found in 158 (5.1%) persons. Among the latter, subtype C and CRF02_AG were most prominent accounting for 62 (2.0%) and 45 (1.4%) persons, respectively. The range of non-B subtypes and recombinant forms by state varied from 0 to 1.6%. Antiretroviral drug resistance mutations were observed in 13 (8.2%) persons with non-B subtypes or recombinant forms and in 314 (10.6%) persons with subtype B, though this difference was not statistically significant.
Conclusions: These data represent the largest and most diverse U.S. population-based sample reported to date, and have implications for initial treatment regimens for persons newly diagnosed with HIV. These findings also emphasize the importance of ongoing molecular HIV surveillance in the United States and will be useful for future evaluations of trends associated with the transmission of drug-resistance mutations and subtype distribution.
BACKGROUND
Variant, Atypical and Resistant HIV Surveillance System (VARHS)
The Centers for Disease Control and Prevention (CDC) maintains the HIV/AIDS reporting system (HARS) that provides data on the HIV epidemic to be used for national, state, and local public health HIV/AIDS prevention program planning and evaluation. In 2004, VARHS was introduced into routine HIV surveillance in selected areas.
The primary objectives of VARHS are to:
- Amplify and sequence the pol region (protease [PR] and reverse transcriptase [RT] genes) from remnant HIV diagnostic specimens from persons newly diagnosed and reported to HARS;
- Estimate the prevalence of mutations associated with HIV drug resistance, resistant patterns, and trends in dissemination of drug resistant strains;
- Determine the prevalence of genetic subtypes to gain insight into the genetic diversity of HIV-1 strains and the distribution of B and non-B variants among populations;
- Provide HIV-1 drug resistance and subtype data to assist local HIV treatment, prevention, and program planning and evaluation.
MATERIALS
3130 sequences were available from confidentially tested (non-anonymous testers), drug naive persons newly diagnosed and reported from March 2003 to October 2006 at 409 sites in 11 surveillance areas. Specimens were taken from remnant diagnostic serum or plasma drawn at participating public and private facilities.
Specimens were collected from the following surveillance areas: Colorado, Illinois, Louisiana, Maryland, Massachusetts, Michigan, Mississippi, North Carolina, Seattle/King County, South Carolina and Virginia.
Specimens were collected in public and private facilities from the following facility types: STD clinics (33.2%), counseling and testing sites (24.7%), private physicians (7.8%), hospitals (6.8%), and other facility types (6.1%). Data on facility of diagnosis were unavailable for 22% of cases.
Of those for whom information was available, 680 (23.7%) were female, 2073 (72.2%) were male; 498 (19.9%) were <25 years old, 1516 (60.6%) were 25-44 years old, 486 (19.4%) were >44 years old; non-Hispanic Black, non-Hispanic White, and Hispanic race constituted 1469 (46.9%), 701 (22.4%), and 293 (9.4%), respectively. Primary HIV transmission categories included men who have sex with men (MSWM), 816 (51.3%), and heterosexual contact, 328 (19.7%).
METHODS
Sequencing was performed on the HIV pol region (protease and the first 900 base pairs of the reverse transcriptase genes) by four laboratories: Stanford University, Palo Alto, CA; University of Washington, Seattle, WA; Maryland
Department of Health and Mental Hygiene, Baltimore, MD; and Michigan Department of Community Health, Lansing, MI.
Pol sequences were aligned using the Clustal W 1.83 multiple sequence program and applied for phylogenetic analysis using Neighbor- Joining method (Phylip version 3.5c) with and without bootstrapping included in the GeneStudio Package (http://www.genestudio.com).
Nucleic acid sequences were translated into amino acids for evaluation of drug resistance mutations using programs from the Stanford online HIV Drug Resistance Database (http://hivdb.stanford.edu).
Major mutations conferring drug resistance were defined according to the IAS-USA guidelines published in August 2006.
SUBTYPE RESULTS
B subtypes were observed in 2970 of 3130 (94.9%) persons.
Non-B subtypes and circulating recombinant forms (CRFs) were observed in 158 (5.1%) persons.
The percentage of non-B subtypes and CRFs ranged from 0 to 10.8% in participating surveillance areas.
The occurrence of non-B variants was significantly higher in non-Hispanic Blacks compared to non-Hispanic Whites (p<.008), cases of non-US origin compared to US origin (p<.0001), females compared to males (p<.0001), and males with heterosexual contact as the reported transmission category compared to MSWM (p<.0001).
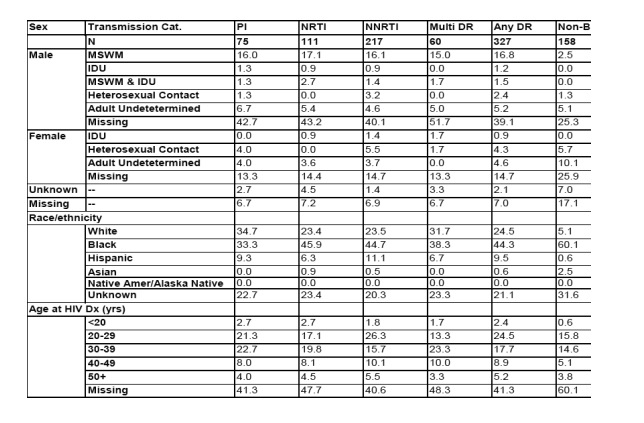
Percentage of newly diagnosed cases with drug resistant HIV and non-B subtypes, demographic characteristics.
Blacks had more NNRTI resistance (44% vs 23%), PI resistance was 34% in White, 33% in Blacks, Among White 34% had PI resistance, 23% NNRTI resistance. Among Blacks, 33% had PI resistance, 44% NNRTI resistance. Regarding NRTIs, among Blacks 45% had resistance, 23% among Whites. Resistance was more common among younger ages 20-40 yrs 15-24% compared to 40-49 yrs (8-10%) and 50+ (3-5%).
|
| |
|
 |
 |
|
|