 |
 |
 |
| |
Entecavir HBV Therapy: does it have HIV activity?
|
| |
| |
Reported by Jules Levin
CROI, Feb 2007, LA
At CROI, Chloe Thio (Johns Hopkins Univ) reported that entecavir (ETV), a drug approved for HBV therapy and not HIV therapy, was found by her to be active against HIV in 3 HBV/HIV coinfected patients. She reported the 2 patients had a 1 log reduction in HIV viral load while on ETV monotherapy and the third perhaps more. She detected the M184V HIV drug resistance mutation in one patient, and claims that ETV has HIV activity. She has not studied resistance in the other 2 patients. To demonstrate HIV activity in vitro by ETV, Thio used what appears to be as far as I know a relatively new and not widely used assay developed by I think Robert Siliciano also at Johns Hopkins, and following her talk, Thio added all drug discovery initiatives should use a sensitive assay to test for HIV activity. ThioŐs report appears to be a first, as far as I know. Following ThioŐs talk Richard Colonno from BMS said at the microphone that they conducted extensive and comprehensive research over 10 years on entecavir and did not find HIV activity. ColonnoŐs full comments at the mirophone can be read in the text of my report below. At the microphone following ThioŐs talk Ray Schinazi and Rich Colonno questioned the validity and reliability of the assay. So, this question remains contentious and unresolved. Presumably a number of doctors have used ETV monotherapy in HBV/HIV coinfected patients, but apparently none have reported any reductions in HIV viral load experienced by patients.
Thio, BMS, and the FDA would like doctors who have used ETV monotherapy to contact them with any information they have that can help in this inquiry. In the Dear Doctor letter BMS advises doctors to use caution in using ETV in coinfected patients while they try to resolve this question. BMS is considering to quickly conduct a small clinical study in patients to answer the question.
It is important to resolve this question quickly because ETV is a potent drug and is an important option for therapy, and at the most recent AASLD liver meeting in Nov 2006 it was reported that after 3 years no drug resistance was found associated with ETV use in treatment-na•ve patients. In coinfected patients ETV has been used in one of 2 ways, either starting HAART plus ETV simultaneously or a patient could start ETV monotherapy and defer HAART for a later time. Every patient has a unique situation and it is important for patients to utilize the best therapy. BMS says caution should be considered in deciding upon therapy for coinfected patients. Excerpts from the BMS Dear Doctor Letter follow the report below.
"The Anti-Hepatitis B Drug Entecavir Inhibits HIV-1 Replication and Selects HIV-1 Variants Resistant to ARV Drugs"
Chloe Thio reported on 3 HBV/HIV coinfected patients who started entecavir therapy for HIV without being on HAART simultaneously and she reported all 3 patients were seen to have a 1 log drop in HIV viral load at the same time that they had large drops in their HBV DNA (viral load). Patient 1 was on AZT/3TC and NVP for <1 year previously but started ETV monotherapy due to high HBV DNA & needed to be treated. His HBV DNA dropped from a high level of 9 to 5 log IU/mL and his HIV viral load, which Thio said had been stable, dropped from 30-50,000 about 1 log. Subsequently his HIV increased & then decreased & settled out now about 0.5 log greater than nadir. Patient 2 was coinfected and also had similarly high HBV DNA & due to these high levels received entecavir monotherapy for 6 weeks during which his HBV DNA dropped dramatically but also his HIV RNA dropped about 1 log. His HIV RNA started to go up so his providers were concerned he might develop HIV drug resistance to they placed him on He was placed on HAART with TDF/FTC + EFV and Thio said his HBV DNA HIV RNA both Ôresponded nicelyŐ to this combination. Patient 3 was a little more complicated Thio said. He had been intermittently on ATZ/3TC previously. His HIV RNA had gone up and down. He had more advanced liver disease as shown by liver biopsy. Since his HIV did not need treatment at the time he was placed on Peg IFN to treat the HBV. He had somewhat of a response for about 6 months but was taken off due to noncompliance. While on Peg his HIV RNA dropped. For about 6 months prior to ETV monotherapy he was off all medications. He started ETV monotherapy because his liver disease clinically worsened. His HBV DNA dropped dramatically. His HIV RNA dropped about 1 log during the first 2 months and subsequently during the next 2 months dropped 2 logs to 130 copies/ml.
Thio said due to these cases we wanted to test in vitro if we could find activity of ETV against. They used a single cycle infectivity assay, which she said is "commonly used today to look for drug resistance". She said they also performed a mutation analysis from patient 1.They extracted his HIV RNA, amplified the polymerase with RT-PCR, cloned the products, and then multiple clones from each time point were sequenced and a phylogenetic tree was constructed.
Thio then showed a slide of the results from the infectivity assay. On the y axis (in log scale, see picture of slide below) is the percent of maximal infection relative to no drug or the amount of replication that occurs. She said here are typical dose response curves for AZT & 3TC. With ETV at very low doses she said we see there is a decrease in replication. She showed where the line was for 50% inhibition & said the IC50 ranged from 0.1 to 10 nanomolars. The IC50 was about 500 times lower than the IC50 of AZT and 50 times lower than the IC50 for 3TC. She said that with AZT & 3TC the more drug you give, with this assay, the greater the inhibition you get but with ETV with doses more than 10 nonomolars the response plateaus out. Thio claims that this is why ETVŐs HIV activity may have been missed, that the asay she used was more sensitive, and that a less sensitive assay might miss ETVŐs HIV activity.
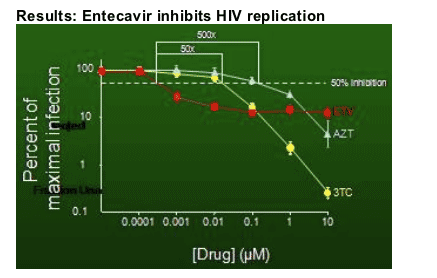
Thio then reported results from use of the single infectivity assay results suggesting that ETV inhibits reverse transcription saying ETV "likely works at the point of reverse transcription or perhaps could work earlier than that".
Regarding whether drug resistance was being selected on ETV, Thio then reported the results from the clonal analysis. At time point zero from patient 1 none of the clones had the M184 mutation (3TC mutation), at 4 months 61% of clones had M184V, and at 6 months 96% of clones had M184V. The only mutation found when they sequenced was M184V. Thio said the phylogenetic tree results suggest the M184V evolved from ETV therapy.
She then showed a slide of the results from putting the virus with the M184V mutation back into the infectivity assay along with wild type HIV and shows decreased inhibition of replication was seen from ETV and 3TC while with wild type virus 3TV & ETV inhibit replication.
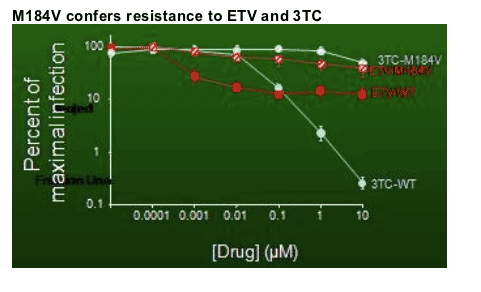
Thio summarized:
"WeŐve shown ETV inhibits HIV replication in vivo and in vitro and likely works at the step on reverse transcription. This activity against HIV is strong enough or potent enough that it can select for the M184V. If we put this M184V mutation back into our system ETV can no longer inhibit replication. So, these data support the fact that ETV should not be used in HIV/HBV coinfected individuals who are not on a fully suppressive HIV regimen without careful consideration of their other options. And a sensitive assay should be used to study other drugs for potential HIV activity before licensure. We would be interested in hearing from other people if they have cases, we would be interested in looking for other resistance mutations that might have occurred on ETV monotherapy therapy. Also, if you have cases where you donŐt find declines in HIV RNA it would be interesting to sequence the virus to see what mutations the virus has that might prevent ETV from inhibiting replication. You can contact Thio. Both the FDA and BMS are interested in having cases reported to them".
Chloe Thio: cthio@jhmi.edu
FDA 1-800-FDA-1088
BMS 1-800-321-1335
Ray Schinazi, a noted basic scientist/researcher in HIV, was the first to the microphone and said "in our hands ETV in PBMCs in a 5-day assay has no antiviral activity up to 100 micromolars. WeŐve also tested against HIV-2 and find no activity there too, so we differ from your.... I know your assay is a specialized assay but we use the regular standard assay, the same assay we used to discover 3TC/FTC in our lab. So I think we do know how we do things in our lab. The question for you is, have you tried to select resistant virus using your assay that you described today or any other method, because in theory if you are right about the selection in patients you should be able to reproduce this in cell culture. Have you done this experiment".
Thio responded:
"We have started doing this experiment. I can tell you it looks like they would select for the M184I, so as most of you know in vivo the M184VI usually occurs before the M184V. So we are continuing this process and hopefully we should know in a couple of weeks if we get the M184V but so far it looks like we have the M184I".
Vincent Soriano asked about resistance in patients 2 and 3, and in response Thio said they are in the process of studying patient 3 now but are unable to study patient 2 because he started on HAART before they knew about the patient so they cannot look for resistance and he did not have stored blood from before starting HAART..
Rich Colonna from BMS went to the microphone:
"For the intention of fair balance weŐd like to point out that weŐve studied ETV for over 10 years. WeŐve assayed this compound against HIV over that 10 year period against HIV probably dozens of times. WeŐve done single cycle, multiple cycle pseudotype assays. WeŐve done in vitro biochemical assays using ETV triphosphate against reverse transcriptase of HIV. In no case do we see activity against HIV below micromolar concentrations. The only time we can see any activity, about 10-fold more than IŐm talking about, submicromolar, is when we use extremely low viral inoculants. In which case we get flat curves similar to what weŐve seen here in your slide but we find those assays are very variable, not reproducible and clearly not anything close to what we would normally get with the titration of all the other antiretroviral drugs. Again, just for fairness I would just like to point out we have done an extensive study of this drug in HIV, a comprehensive study and we have not seen any activity against HIV".
In response to a question from Norbert Brau regarding testing telbivudine against HIV, Thio said adefovir at high doses has activity against HIV but at 10 mg is not active against HIV; we do have some telbivudine, which we will test.
Excerpts from BMS Dear Doctor letter:
"....Current treatment guidelines recommend BARACLUDE as an option for treatment of HBV in the HIV/HBV co-infected adult patient who does not qualify for HAART; however, in light of the newly-reported case history provided below, BMS advises caution if BARACLUDE is used in this setting.....
Details of the newly-reported case history are provided below:
- A 31-year-old HIV/HBV co-infected male received combination zidovudine, lamivudine, and nevirapine for less than 1 year in 2000. HAART was discontinued and the patient remained clinically stable with respect to his HIV. In early 2006, with a CD4+ of >500 cells/mm3 and an HIV-1 RNA of approximately 35,000 copies/mL, BARACLUDE monotherapy was initiated for the treatment of HBV. Within 2 months, the HBV DNA decreased by approximately 5.5 log10 IU/mL, and the HIV RNA decreased to approximately 2,000 copies/mL, subsequently remaining below baseline levels. HIV resistance testing at the start of BARACLUDE therapy did not show resistance, but the M184V substitution was detected following 6 months of therapy with BARACLUDE.
This patient is one of three HIV/HBV co-infected patients not receiving HAART in whom a 1-log10 reduction in HIV RNA has been noted while receiving BARACLUDE as treatment for chronic HBV infection.
Bristol-Myers Squibb has assessed the activity of BARACLUDE against HIV-1 in vitro: the EC50 for laboratory strains NL4-3, BRU and LAI was >1 uM in cell culture assays.In addition, BMS has evaluated the use of BARACLUDE 1 mg in the HIV/HBV coinfected population that was receiving simultaneous HAART. These data were from a single randomized, double-blind, placebo-controlled evaluation of the activity of BARACLUDE in 68 HIV/HBV co-infected patients who entered the study with stable HIV-1 RNA <400 copies/mL (mean CD4+ count of 511 cells/mm3). Patients continued their lamivudine-containing HAART regimen (lamivudine dose 300 mg/day) and were assigned to add either BARACLUDE 1 mg once daily (51 patients) or placebo (17 patients) for 24 weeks followed by an open-label phase for an additional 24 weeks where all patients received BARACLUDE (entecavir). At Week 24, BARACLUDE-treated patients had a mean reduction in HBV DNA of -3.65 log10 copies/mL compared with an increase of 0.11 log10 copies/mL in the placebo arm. In this setting, no difference in HIV RNA or CD4+ was noted between the BARACLUDE and placebo treatment groups.4,5
Bristol-Myers Squibb has collaborated closely with the U.S. Food and Drug
Administration on this issue, and the following changes to the package insert are in process of being made:
Under section MICROBIOLOGY/ Antiviral Activity:
"The EC50 value of entecavir against human immunodeficiency virus (HIV) type 1 laboratory strains NL4-3, BRU, and LAI was >1 _M in cell culture assays."
Under section INDICATIONS AND USAGE/ Description of Clinical Studies/ Special Populations:
"BARACLUDE has not been evaluated in HIV/HBV co-infected patients who were not simultaneously receiving effective HIV treatment."
The revised package insert will be available on www.baraclude.com. If you have any questions about this new information or require additional medical information, please contact Bristol-Myers Squibb at 1-800-321-1335.
If you have had a patient who experienced an adverse event following, or coincident with the use of BARACLUDE, please contact Bristol-Myers Squibb at 1-800-321-1335 or the FDA MedWatch program by phone at 1-800-FDA-1088, by fax at 1-800-FDA-0178, or by mail (MedWatch, 5600 Fishers Lane, Rockville, MD 20852-9787).
|
| |
|
 |
 |
|
|