 |
 |
 |
| |
Hepatic Steatosis (30%) and Liver Function Abnormalities among HIV-Infected Persons
|
| |
| |
Reported by Jules Levin
CROI, Feb 2007, Los Angeles
Nancy Crum-Cianfl one1,2, Charles Hames3, Sheila Medina1,2, Richard Campin4, Thomas Capozza3, Carolyn Brandt1,2, Dean Asher4,
Brad Hale1, and the TriService AIDS Clinical Consortium
1. TriService AIDS Clinical Consortium, Infectious Disease Clinical Research Program (IDCRP), USUHS, Bethesda, Maryland,
2. Infectious Disease Clinic, 3. Gastroenterology Department and 4. Radiology Department, Naval Medical Center San Diego, California
ABSTRACT
Background: Non-alcoholic fatty liver disease (NAFLD) and steatohepatitis (NASH) are increasingly described conditions in the general population, but there are currently no data regarding these conditions in HIV-infected persons without co-infection with hepatitis C virus.
Methods: Clinic patients from a military HIV clinic with low rates of hepatitis C were prospectively enrolled to examine the prevalence and predictors of steatosis and abnormal liver function tests (LFTs) among HIV patients. A questionnaire regarding medication use and social/behavioral habits was performed along with a BMI assessment, LFTs, and liver ultrasound (US). Those with abnormal LFTs had a full laboratory panel to include iron studies, ceruloplasmin, autoimmunity panel, alpha-1 antitrypsin, and serum alcohol level to ascertain the etiology of the abnormality. A liver biopsy was performed for those with steatosis or liver disease of unclear etiology. Statistical analyses included Fishers exact and rank sum testing; multivariate models utilized backward stepwise logistic regression.
Results: 60/200 (30%) had abnormal LFTs, most which were grade 1 (1.25-2.5x normal). The mean age of patients was 39 years (range, 20-68) with a mean CD4 count of 541 (18-1448 cells/mm3); 74% had received HAART. Of those with elevated LFTs, 7 (12%) were HbsAg positive, 2 (3%) hepatitis C
seropositive, and one (2%) had probable hemochromatosis. In the multivariate model, past HAART use (OR 5.6, p=0.002), alcohol use (OR 1.1, p=0.01), and increased BMI (OR 1.1, p=0.03) were predictive of abnormal LFTs. Thirty percent (44/148) had steatosis on US; 41/44 (93%) met the criteria for NAFLD.
Based on liver biopsy findings, US had a sensitivity of 83%, specificity of 90%, and PPV of 94% for detecting steatosis. 25% of patients had fibrosis on biopsy. Only 22% of those with steatosis had abnormal LFTs, but 90% had hepatomegaly.
Predictors of steatosis in the multivariate model included BMI (OR 1.92 per 5 unit increase, p=0.01) with a trend towards d4T use (OR 1.7, p=0.13). BMI and waist circumference were correlated (r=0.73, p<0.05).
Conclusion: Non-alcoholic hepatic steatosis is common (~30%) among HIV-infected patients. Hepatomegaly, but not abnormal LFTs, was predictive of NAFLD. LFTs were not a useful screening test for steatosis. Risk factors for NAFLD among HIV patients include increased BMI and/or waist circumference and perhaps d4T use.
Background
Liver enzyme elevations are common among human immunodeficiency virus (HIV)-infected persons. Early in the HIV epidemic, liver disease was often due to opportunistic infections such as Mycobacterium tuberculosis, Mycobacterium avium complex, and cytomegalovirus. With the advent of highly active antiretroviral therapy (HAART) in 1996, disease due to opportunistic infections and other AIDS defining conditions dramatically declined. However, HIV-infected patients now face substantial morbidity and mortality from non-AIDS defining conditions. Recent studies have shown that one of the most common causes of death among HIV-infected persons is liver disease.
In the post-HAART era, the cause of this liver dysfunction appears to be multifactorial. Antiretroviral medications such as protease inhibitors and nevirapine have been implicated. Given similar routes of transmission, concurrent viral infections with hepatitis B and/or C are associated with liver disease. Alcohol abuse and illegal drugs (cocaine, methamphetamines), hemochromatosis, Wilson's disease, autoimmune hepatitis, and alpha-1 antitrypsin deficiency are also causes of LFT aberrations.
Another potential cause of liver disease among HIV patients in the post-HAART era may be nonalcoholic fatty liver disease (NAFLD). NAFLD is estimated to occur in 30% of the general U.S. population and is the most common cause of elevated liver function tests. The prevalence of NAFLD is increasing in the U.S.,
reflecting the rising rates of obesity, hyperlipidemia, and insulin resistance/diabetes, which are known risk factors for NAFLD. HIV-infected persons also have high rates of these predisposing conditions; hence,
NAFLD may be playing a substantial role in liver disease among HIV patients. Data on NAFLD as an etiology of abnormal LFT's among the HIV positive population are limited.
We performed a prospective study among HIV-infected patients to determine the prevalence and causes of elevated liver function tests. In addition, we studied the prevalence of NAFLD in this population and examined the potential risk factors for its development.
METHODS
HIV-infected patients from a military clinic with low rates of hepatitis C were prospectively enrolled to examine the prevalence and predictors of steatosis and abnormal liver function tests (LFTs) among HIV patients.
After signing the informed consent, patients completed a questionnaire regarding demographic information, signs or symptoms of liver disease, medical history, current medications, history of antiretroviral therapy use, and alcohol/drug use. Data regarding medical illnesses, CD4 counts, HIV viral
loads, prior liver function test results, prior lipid panel, body mass index (BMI), and stage of HIV infection was obtained via review of the patient's medical record. Each patient had a waist and hip circumference performed.
Participants donated blood specimens (unless already drawn within the past month) for a complete blood count (CBC), and liver function testing to include alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase, lactic dehydrogenase, total bilirubin, and albumin. Women of childbearing age had a BHG (urine); those found to be positive were excluded from the study. Biochemical liver abnormalities will be graded as follows: 1.25-2.5x normal (grade 1), 2.6-5 x (grade 2), 5.1-10x (grade 3) and >10x (grade 4).
Those with normal liver function tests were retested for the same laboratories in 4-8 months. Those with abnormal LFTs at either time (defined as ALT>63 IU/L, AST>41 IU/L, Alk Phos>126 IU/L or a TBili>2 mg/dl) underwent additional blood tests to include serum iron, TIBC, ferritin, ANA panel, anti-smooth muscle antibody, hepatitis panel (including hepatitis B DNA and hepatitis C RNA viral level if CD4 count is below 200), TSH, PT/INR, ceruloplasmin, alpha-1 antitrypsin phenotype, alcohol level, fasting blood sugar, and a fasting lipid panel. Those participants who are hepatitis B core antibody positive and surface antigen negative had a hepatitis B DNA viral load performed.
All patients (including those with normal LFTs) had a modified right upper quadrant ultrasound performed in the Radiology Department to evaluate for steatosis and hepatomegaly.
Test results were reviewed by a gastroenterologist and those necessitating a liver biopsy on clinical grounds (either due to elevated LFTs, hepatomegaly on physical exam or steatosis/other hepatic abnormality on ultrasound) were offered this procedure.
Statistical analyses included calculation of the prevalence of liver abnormalities and NAFLD. Demographic and clinical data between those with and without liver function abnormalities utilized the Fisher's exact test for dichotomous variables and the rank-sum test for continuous variables. A logistic regression model was utilized for the multivariate analyses to compare those with and without liver disease by examining variables of interest including demographics, HIV clinical disease indicators, medical conditions, and medications (STATA software, College Station, Texas).
RESULTS
Demographics of the Study Population:
- The mean age of patients was 39 years (range, 20-68).
- 93.5% were male.
- Race was Caucasian in 50%, African American in 24.5%, Hispanic in 15%, and other for 10.5%.
- The mean CD4 count was 541 (18-1448 cells/mm3); 74% had received HAART.
Abnormal LFTs:
60/200 (30%) had abnormal LFTs, most which were grade 1 (1.25-2.5x normal).
- Of those with elevated LFTs, 7 (12%) were HbsAg positive, 2 (3%) hepatitis C
seropositive, and one (2%) had probable hemochromatosis.
- There was no difference in symptoms such as nausea, vomiting, abdominal
pain, change in stools, fatigue, sexual drive, appetite, or pruritis between those
with and without abnormal LFTs.
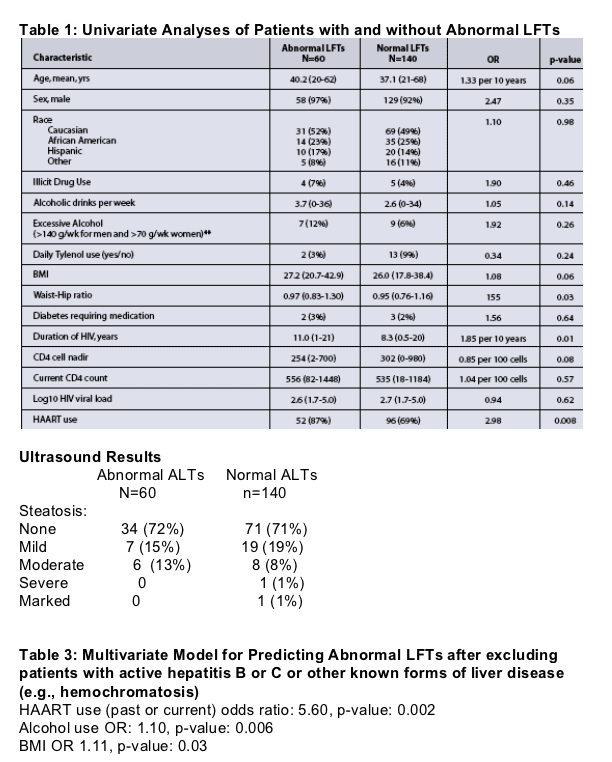
In the univariate analyses (Table 1), predictors of abnormal LFTs included an elevated waist-hip ratio, longer duration of HIV infection, and use of HAART. There were trends for increasing BMI, age, and the use of alcohol, but no associations with specific antiretrovirial medication, lipid levels, or diabetes.
Abnormal LFTs were not significantly associated with changes in liver size or with steatosis on ultrasound evaluation. Table 2
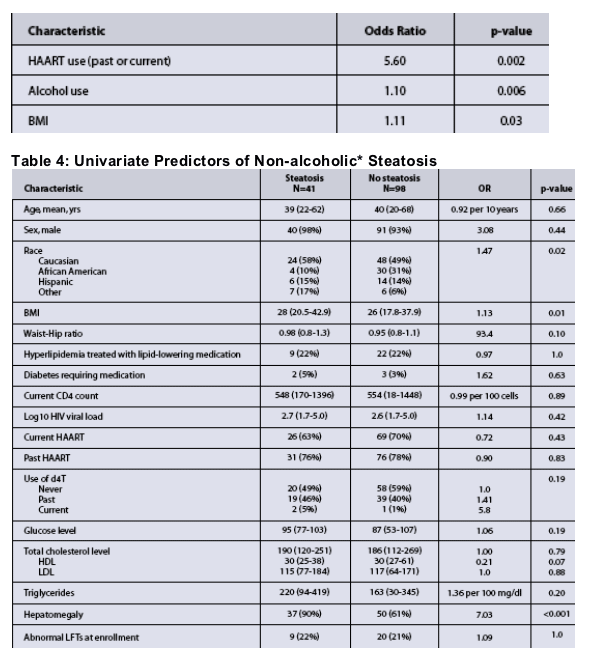
In the multivariate model, past or current HAART use (OR 5.6, p=0.002), alcohol use (OR 1.1, p=0.006), and increased BMI (OR 1.1, p=0.03) were predictive of abnormal LFTs (Table 3).
RESULTS
Hepatic Steatosis:
- Thirty percent (44/148) had steatosis on US; 41/44 (93%) met the criteria for NAFLD.
- Based on liver biopsy findings (figures 1 & 2). US had a sensitivity of 83%, specificity of 90%, and PPV of 94% for detecting steatosis.
- 25% of patients had fibrosis on biopsy.
- Only 22% of those with steatosis had abnormal LFTs, but 90% had hepatomegaly.
- NAFLD was not associated with specific symptoms attributed to liver disease
- Predictors for Steatosis are shown in tables 4.
- Hepatomegaly was the most predictive finding (OR 1.6 overall, p<0.001).
- Excluding hepatomegaly from the model, predictors of steatosis in the multivariate model included BMI (OR 1.92 per 5 unit increase, p=0.008) with a trend towards d4T use (OR 1.7, p=0.13).
- Although the sample size was small, a low HDL was also associated with NAFLD (OR 0.72, p=0.04).
- Elevated LFTs were not a good indicator of NAFLD.
CONCLUSIONS
Non-alcoholic hepatic steatosis is common (~30%) among HIV-infected patients.
- Hepatomegaly is a common finding among patients with NAFLD.
- LFTs are not a useful screening test for steatosis (sensitivity of 20%).
- Compared to liver biopsy results, ultrasound detection of steatosis has a good sensitivity (83%) and specificity of (90%).
- Risk factors for NAFLD among HIV patients include increased BMI and/or waist circumference and perhaps d4T use and a low HDL level.
|
| |
|
 |
 |
|
|