 |
 |
 |
| |
Effect of Famotidine 20 and 40 mg Dosing Regimens on the Bioavailability of Atazanavir with Ritonavir in Combination with Tenofovir in Healthy Subjects
|
| |
| |
"Effect of Famotidine 20 and 40 mg Dosing Regimens on the Bioavailability of Atazanavir with Ritonavir in Combination with Tenofovir in Healthy Subjects" Download PDF Here
Reported by Jules Levin
CROI, Feb 2007, Los Angeles
Sangeeta Agarwala*, A Persson, T Eley, M Child, D Filoramo, T Li, X Xu, and R Bertz
Bristol-Myers Squibb Pharma Res Inst, Princeton, NJ, US
STUDY OBJECTIVES
Primary:
To assess the pharmacokinetics (PK) of ATV, and identify one or more dosing regimens of ATV/RTV/TDF when dosed with FAM that would result in ATV exposures similar to those when ATV/RTV/TDF 300/100/300 was dosed without FAM
Secondary:
To assess the PK of RTV and tenofovir when co-administered with ATV in the presence and absence of FAM
To assess the safety and tolerability of ATV/RTV/TDF in the presence and absence of FAM
Background: Previously, a reduction of 18 to 28% in atazanavir (ATV) exposures was noted when the H2-receptor antagonist, famotidine (FAM) 40 mg every 12 hours was administered with ATV/ritonavir (RTV) 300/100 mg in healthy subjects; the effect of lower doses of FAM and combination with tenofovir (TDF) have not been studied. TDF decreases ATV exposures when coadministered with ATV/RTV by ~25 to 30%. The objective was to evaluate dosing strategies for FAM to maintain ATV exposures when coadministered with RTV and TDF.
Methods: Healthy subjects (n = 40) received 300/100/300 mg ATV/RTV/TDF once daily for 10 days (Trt A). Subjects were then randomized into 2 cohorts (n = 20). Cohort 1 received FAM 20 mg every 12 hours, morning FAM was co-administered with ATV/RTV/TDF for 7 days (Trt B), followed by FAM 20 mg every 12 hours, morning FAM was given 2 hours after ATV/RTV/TDF for 7 days (Trt C). Cohort 2 received FAM 40 mg every afternoon, 12 h from ATV/RTV/TDF for 7 days (Trt D), followed by FAM 40 mg every 12 hours temporally separated as in Trt C (Trt E). Intensive pharmacokinetic was evaluated on days 10, 17, and 24. In all Trt, ATV/RTV/TDF was administered in the AM with a light meal. Adjusted geometric mean ratios (GMR) and 90% confidence intervals (CI) for ATV pharmacokinetics were estimated by general linear mixed effects models with Trt A as the reference.
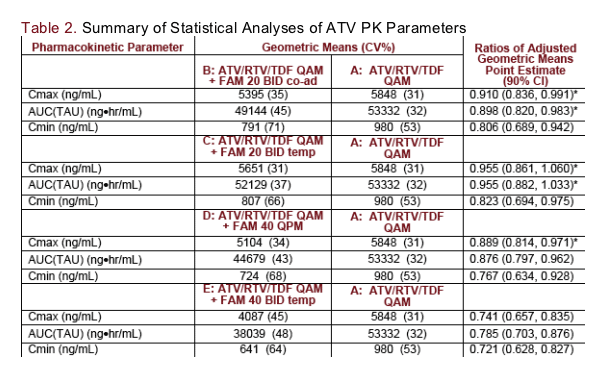
Results: GMR of ATV pharmacokinetics for each of Trt B-E relative to Trt A with 90% CI are shown: Similar ATV exposures were noted for Trt B, C, and D when compared to Trt A; 90%CI were within 0.80 to 1.25 for ATV Cmax in Trt B, C, and D and AUC in Trt B and C. AUC in Trt D fell just below 0.80. Cmin was 18 to 23% lower for Trt B, C, and D. In contrast, decreases in ATV Cmax, AUC, and Cmin of 26%, 21%, and 28% were noted with Trt E. RTV and TDF exposures were similar with FAM. No unexpected adverse events were noted.
Conclusions: This study demonstrates several possible ways to administer ATV/RTV with both TDF and FAM. Relative to ATV/RTV 300/100 mg + TDF, ATV exposures were similar when FAM was given as 20 mg twice daily simultaneously administered or temporally separated, or temporally separated as 40 mg every afternoon.
When FAM 20 mg twice daily was administered either temporally separated (10 h before and 2 h after) or simultaneously with ATV/RTV and TDF, Cmax and AUC were not affected and an estimated <20% decrease in Cmin was observed
FAM 40 mg administered QPM (12 h prior to ATV/RTV and TDF in the AM) produced similar exposures to both FAM 20 mg BID regimens. Cmax and AUC were minimally affected (about 12% reduced). Cmin was 23% lower relative to the control treatment
__
FAM 40 mg administered twice daily temporally separated from ATV/RTV and TDF (10 h before and 2 h after) resulted in decreases in ATV Cmax, AUC and Cmin of 26%, 21% and 28%, respectively
This study demonstrates several ways to administer ATV/RTV with
both TDF and an H2 receptor antagonist such as FAM
Pharmacokinetics:
Atazanavir
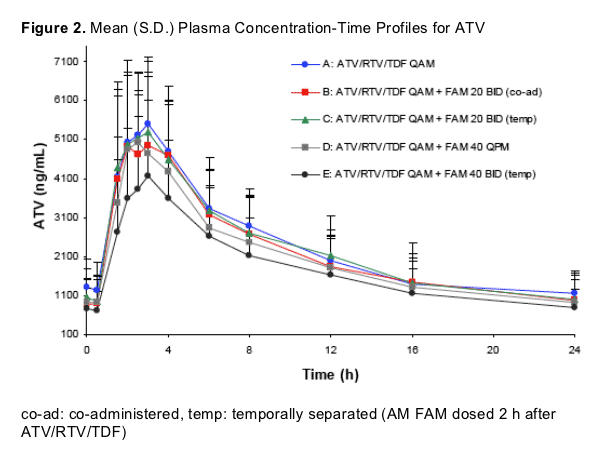
# Subjects/Treatment: A: 39, B: 18, C: 18, D: 20, E: 18
Median Tmax ranged from 2.0 - 2.75 hours
co-ad: co-administered, temp: temporally separated (AM FAM dosed 2 h after ATV/RTV/TDF)
* Met the criteria for no-effect: 90% CI within 0.80 - 1.25
ATV exposures in the control treatment (ATV/RTV/TDF QAM) were comparable to historical data5
The data suggest a dose dependent effect of FAM on ATV exposures. ATV exposures were reduced with FAM 40 mg BID as compared to FAM 20 mg BID (co-administered or temporally separated)
The administration of FAM 20 mg BID (either temporally separated or co-administered with ATV/RTV/TDF) did not affect ATV AUC(TAU) or Cmax and estimated reductions in ATV Cmin of <20% were observed
FAM 40 mg once daily 12 hours after ATV/RTV/TDF minimally reduced ATV AUC(TAU) and Cmax (approximately 12% or less) relative to ATV/RTV/TDF without FAM. A reduction in ATV Cmin of approximately 23% was observed
FAM 40 mg BID temporally separated from ATV/RTV/TDF resulted in 21% and 26% reductions in ATV AUC(TAU) and Cmax respectively. A 28% reduction in ATV Cmin was observed
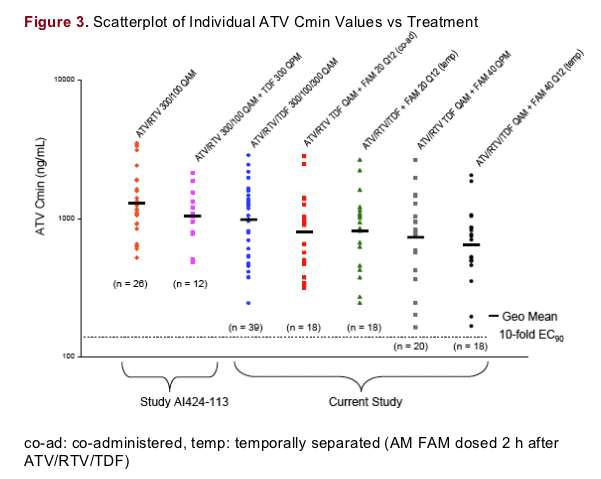
As demonstrated in Figure 3, the administration of ATV/RTV 300/100 mg with TDF 300 mg (temporally separated by 12 hours) in a previous study, AI424-113, yielded an approximate 20% reduction in ATV Cmin relative to ATV/RTV without TDF4
The geometric mean ATV Cmin (980 ng/mL) after the simultaneous administration of ATV/RTV 300/100 mg with TDF 300 mg in the current study was comparable to that observed in study AI424-113 (1031 ng/mL), even though TDF was administered 12 hours after ATV/RTV
No individual Cmin value from the healthy subjects in any of the treatment groups in the current study fell below the 10-fold protein binding adjusted EC90 for wild-type HIV (140 ng/mL). Note the data were collected in healthy subjects; ATV exposures would be expected to be somewhat lower in HIV-infected subjects, however Cmin values would still be expected to be well above the median wild-type EC90 (14 ng/mL)
There appears to be a trend toward lower ATV Cmin values as FAM dose is increased from 20 mg BID to 40 mg BID
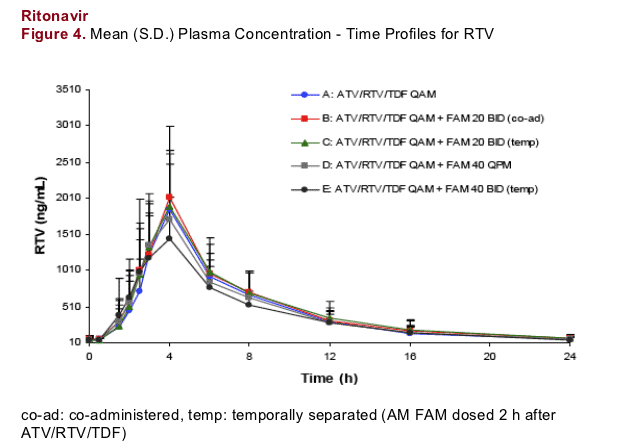
As noted in Figure 4, RTV exposures are largely unaffected by FAM; a finding that is consistent with previous results2
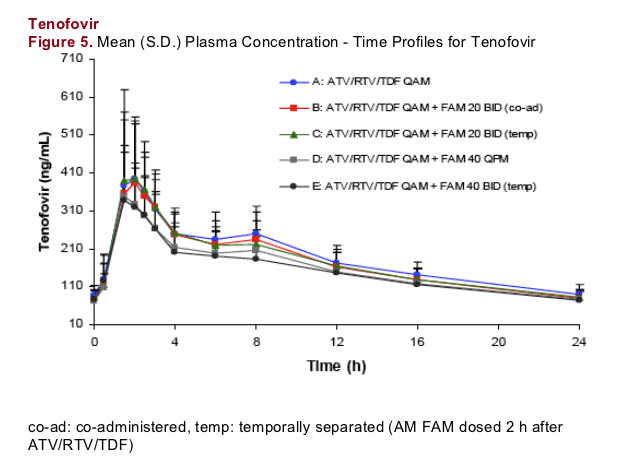
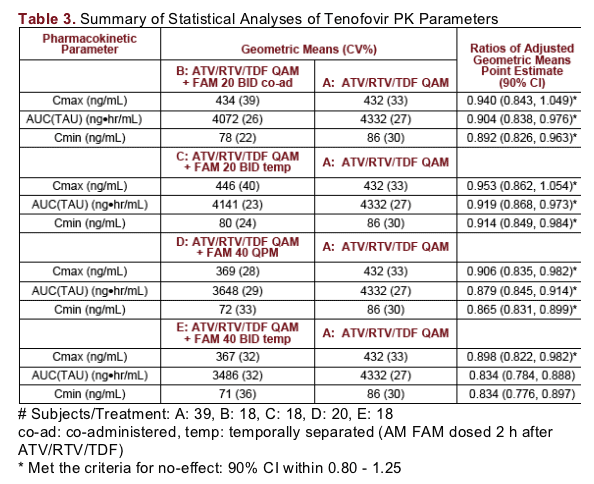
Tenofovir exposures were similar across treatments. Cmax and AUC were within approximately 10% and 17%, respectively, of control values for all treatments
Famotidine:
Sparse sampling was collected for FAM and no PK parameters were calculated, however concentrations in each regimen were as expected
FAM 20 mg BID regimens produced maximum concentrations that were approximately 50% of those observed with FAM 40 mg BID dosing, suggesting linear pharmacokinetics
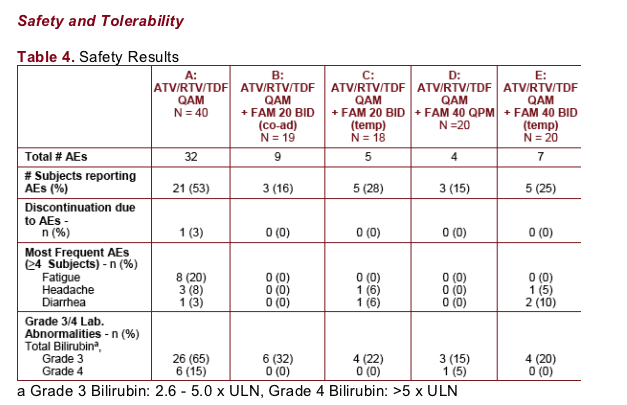
There were no reported deaths or serious adverse events in this study. One subject discontinued prior to randomization due to the AE of vomiting
ATV is a reversible inhibitor of UGT1A1 that is primarily responsible for glucuronidation of bilirubin. Inhibition of UGT1A1 by ATV is associated with benign, reversible hyperbilirubinemia and has not been linked to the development of hepatotoxicity. Elevated bilirubin levels decreased to normal range upon cessation of study drug
Co-administration of ATV/RTV/TDF and FAM up to 40 mg twice daily was generally safe and well tolerated
|
| |
|
 |
 |
|
|