 |
 |
 |
| |
Two New HIV Drugs: NNRTI TMC278 and CCR5 Antagonist Maraviroc
|
| |
| |
CROI, Feb 2007, Los Angeles
Reported by David Margolis, MD, University of North Carolina
Advantage to the new NNRTI on the block
The primacy of efavirenz (Sustiva) was challenged by a new diarylpyrimidine non-nucleoside reverse transcriptase inhibitor (NNRTI), in the Tibotec stable of compounds, TMC278 (Abstr. 144LB;
http://www.retroconference.org/2007/Sessions/040.htm).
Pozniak reported the findings of a 48-week comparative study in ART-naive patients, which pitted TMC278 against efavirenz (EFV). In the lab TMC278 is highly active against wild type and drug-resistant HIV-1 in vitro, and it has demonstrated potent short-term antiviral activity in patients. It half-life is 48 hours, similar to EFV, and doses of 25-150 mg qd were active in phase I studies.
Three blinded, once-daily doses of rilpivarine (if you don't like to say TMC278) were compared with open-label EFV, all in combination with Combivir or Truvada. The primary endpoint was the proportion of patients with confirmed viral load <50 copies/mL (TLOVR definition, non-completer = failure) at 48 weeks.
89 patients received EFV and 279 were dosed with TMC278 at 25, 75, or 150 mg once daily. Combivir was used by 76% of patients and Truvada by 24%. A third of the patients were women, with a mean age of 35 years. Baseline median log10 viral load was 4.85 copies/mL and median CD4 cell count was 203 cells/µl.
At 48 weeks, there was was no statistically significant difference in the efficacy results between any of the treatment arms. The most common adverse events similar in all arms, but nervous system disorders and rash were less frequent with TMC278 (28% and 8%, respectively) compared with EFV (48% and 19%, respectively). Elevations of total and LDL cholesterol were less with TMC278 (mean 5 mg/dL and 0 mg/dL with TMC278 versus 31 mg/dL and 16 mg/dL).
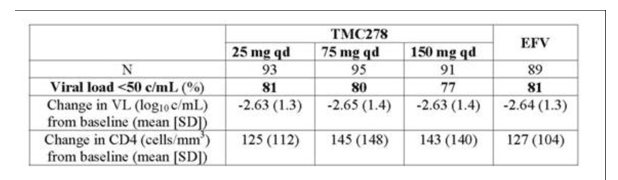
As seen in this table viral load reductions from baseline were -2.63 to -2.65 in all treatment arms.
Earlier, Bauman (abstr. 88;
http://www.retroconference.org/2007/Sessions/026.htm)
had presented elegant structural rationale for the superiority of TMC278 with respect to drug resistance. TMC278 retains activity in vitro against NNRTI-resistant HIV-1 strains, including L100I/K103N and K103N/Y181C double mutants, which are resistant to all approved NNRTI. The concept of structural flexibility in overcoming the effects of drug-resistance mutations evolved from systematic structural studies throughout the drug-development process.
These laboratory studies created HIV RT mutations based on other HIV-1 strains, and then built and rebuilt RT inhibitor molecules to select drugs with enhanced contacts to the viral enzyme. By creating pure crystals of drug and RT enzyme in the lab, the scientists could take pictures of the drug binding the enzyme at a resolution of 2.0-Angstroms (two 10-millionths of a millimeter). This process, called "crystal engineering" by the scientists, allowed the desing of an RT inhibitor with a finger-like structure to fit precisely into a tunnel-like groove in the RT enzymes active site, an interaction that is difficult for an enzyme mutation to break up. Although TMC278 seems, preliminarily, at least the equal of efavirenz with perhaps fewer side effects, the "resistance-to-resistance" of this new candidate will require more validation.
Another HIV entry inhibitor comes of age: Maraviroc, CCR5 blocker
Chemokine receptors (CCRs) serve to regulate cellular responses by signaling in response to chemokines, the hormones of the immune system signals. However, in addition to binding a CD4 receptor to infect a cell, HIV also requires interaction with a CCR. CCRs, particularly CCR5 as it is the CCR most-often used by HIV, are attractive drug targets for several reasons. There is a track record in the pharmaceutical industry of successful drug development of inhibitors of the G protein-coupled receptors, of which CCRs are a member. A proportion of the population has a mutation of CCR5 (delta 32), which renders the receptor nonfunctional, is protective against HIV-1 transmission, but confers no physiologic abnormality. Therefore long-term, chronic blockade of CCR5 by an antiviral drug is hoped to be safe.
Just as integrase inhibitors have had to cross many hurdles in the long road to licensure, so too have CCR antivirals. But the success of the MOTIVATE trials of the Pfizer CCR5 antiviral antagonist Maraviroc (MVC) was another benchmark at the 2007 CROI meeting, signifying the arrival of another potent drug that can contribute significantly to therapy in the face of multidrug (MDR) resistant HIV. As with integrase inhibitors, later studies may also lead to new cocktails and treatment sequences using CCR inhibitors.
MOTIVATE 1 and 2 were presented by Mayer (Abstract 144bLB) and van der Ryst (Abstract 144aLB) in a tag team format
(http://www.retroconference.org/2007/data/files/
webpage_for_CROI.htm). These phase 2b/3 studies were performed in North America, Europe and Australia as double-blind, placebo-controlled studies to assess the safety and efficacy of MVC in treatment-experienced HIV-infected patients. The primary endpoint of mean change in viral load at 24 weeks was presented.
Patients with 3-class drug exposure, therapy failure with VL > 5000 copies/ml, and R5-tropic HIV-1 as measured by the Monogram Trofile assay were randomized 1 : 2 : 2 to receive placebo, MVC once daily or MVC twice daily with optimized background therapy (OBT). OBT was allowed to be 3 to 6 antiretroviral drugs with or without low-dose ritonavir. MVC was given at a dose of 300 mg (qd or bid), unless a protease inhibitor (PI) other than tipranavir and/or delavirdine was given. Due to MVC boosting effects, in these cases MVC 150 mg once or twice daily was given. The analysis was stratified by baseline viral load over or below 100,000 c/ml and by enfuvirtide use.
1076 patients entered the two studies, and after Trofile testing, 585 received therapy in MOTIVATE 1 and 464 in MOTIVATE 2. Baseline characteristics were similar in both studies, with median CD4 count at entry in the 3 arms of both studies about 165 and mean HIV-1 RNA of about 4.8 logs/ml. 65-75% of patients had 2 or fewer active drugs in OBT, as per baseline resistance testing. Adverse events, severe adverse events, AIDS-defining events, and laboratory (including liver enzyme) abnormalities occurred with similar frequency across groups.
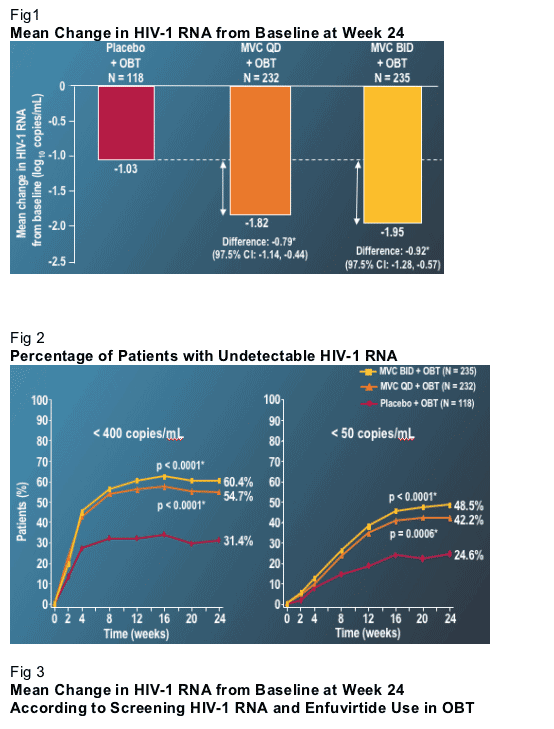
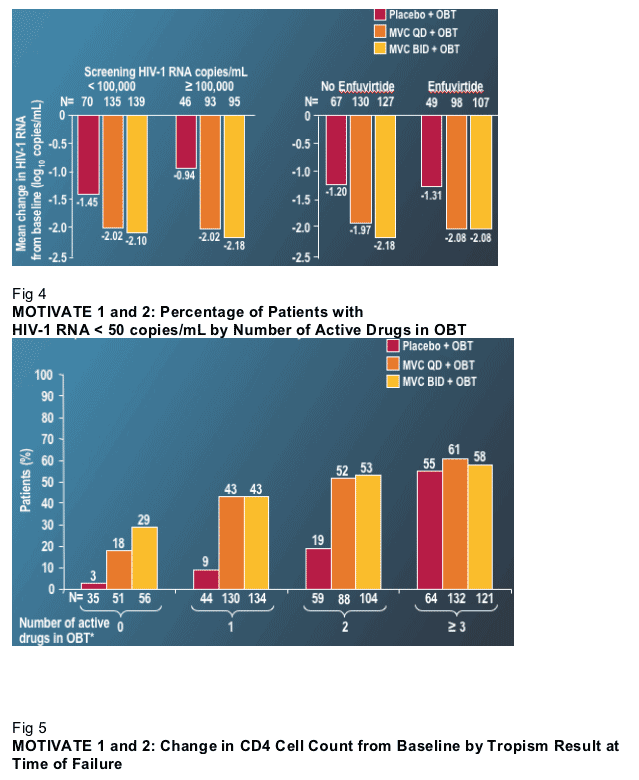
Patients receiving MVC had almost a 1 log greater drop in VL than patients receiving OBT and placebo (Fig. 1). There was little difference between the MVC bid and qd arms. At least to this 24-week point in the analysis, there seems to be no advantage to the higher bid dose. Nearly twice as many patients achieved VL below 400 or 50 copies if they received MVC (Fig. 2). VL declines were equally robust in the high and low VL strata, and with and without the use of enfuvirtide (Fig. 3). (note from Jules Levin: the same was observed from initial results with TMC114 but after further analysis Fuzeon improved response in patients who were sensitive to fewer ART drugs at baseline. So further analysis should be conducted by Pfizer's Maraviroc team).
This last observation was of interest, if disappointing. Relatively comparable VL declines in the patients who used or did not use enfuvirtide most likely reflect that in these studies providers used enfuvirtide conservatively. That is to say that enfuvirtide was likely used when few other active agents were available. One of the disappointing aspects of this result is that it suggests that there is no clinical synergy between enfuvirtide and MVC. It has long been hoped that two agents that impede viral entry may have a synergistic clinical effect. Although not a direct test of this question, the lack of a marked improvement in response in those patients that received enfuvirtide with MVC provides no evidence that potent synergy exists (note from Jules: further evaluation of this interesting questyion should be conducted by Pfizer). Finally, similar to the integrase studies, MVC improved response when 2 or fewer active agents were present in OBT (Fig 4), but MVC added little to a regimen that contained 3 or more active agents.
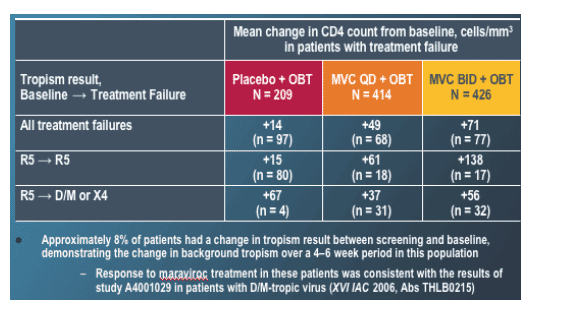
Of the patients who failed MVC, roughly half (Fig. 5) were found to have HIV at failure that used the CXCR4 receptor (X4-tropic) or both the CCR5 and CXCR4 receptor (dual/mixed X4/R5) as measured by the Trofile assay. It was mentioned that 8% of patients had virus that "changed tropism" between screening and entry. However this may simply be a reflection of the level of sensitivity of the Trofile assay for the detection of lower or fluctuating levels of X4 or DM virus within a population at the time of sampling.
To this point, poster 680 (from the Pfizer group) (http://www.retroconference.org/2007/Abstracts/28434.htm)
used a new technology call "ultradeep sequencing" to measure the frequency of X4 or DM virus. A high-throughput mass DNA sequencing machine called the 454 can simultaneously sequence hundreds of thousands of clones within a single sample using a chip-type method to amplify and immobilize DNA fragments spanning the gene of interest. This method should therefore detect individual clones present in a heterogeneous virus pool at a frequency of <1%.
Over 100,000 envelope sequences in the region that determines R5 or X4 use were examined in plasma samples from 2 patients. These patients had been identified as having only R5 HIV at screening, but CXCR4-using virus was detected following 10 days of monotherapy with MVC. At baseline, a single X4-using envelope sequence was found in more than 8000 sequenced. After 10 days of MVC monotherapy (in an earlier phase 1 study) this number had increased to 20 individual envelops predicted to use X4. This suggests that X4 viruses do not appear upon MVC therapy, but are selected from a tiny pre-existing population. The Trofile assay may therefore identify patients from whom MVC is not suitable by identifying those in whom this DM/X4 population is large enough to be readily detected.
Motivate 1 Study: Efficacy and Safety of Maraviroc plus Optimized Background Therapy In Viremic, ART-Experienced Patients Infected With CCR5-Tropic HIV-1: 24-Week Results of Phase 2b/3 Studies (03/05/07)
Maraviroc Combined Analysis & MOTIVATE 2 (Europe, Australia, USA): 24 week Results (03/01/07)
48-week primary analysis of trial TMC278- C204: TMC278 demonstrates potent and sustained efficacy in ARV-naive patients (03/01/07)
|
| |
|
 |
 |
|
|