 |
 |
 |
| |
Hepatitis B Virus (HBV) Dynamics in HIV-HBV Co-infected Patients: Significant Difference in the Viral Activity of Adefovir (ADV) or Tenofovir (TDF)
|
| |
| |
Reported by Jules Levin
CROI, Feb 2007, Los Angeles
K Lacombe,1,2,3 PY Boelle,1,2,4 J Gozlan,5,6 G Pialoux,7 C Lascoux-Combe,8 JM Molina,9 P Miailhes,10 A Boyd,2,3 and PM Girard,2,3
1Inserm, UMRS707, Paris F-75012 France; 2Universite Pierre et Marie Curie - Paris VI, Paris F-75012, France; 3AP-HP, service de maladies infectieuses et tropicales, Hopital Saint-Antoine, Paris F-75012, France;
4AP-HP, service de sante publique, Hopital Saint-Antoine, Paris F-75012, France; 5AP HP, Hopital Saint-Antoine, Service de bacteriologie-virologie, Paris, F-75012 France; 6Universite Pierre et Marie Curie - Paris 6, UMR
7079, Les Cordeliers, Paris, F-75006 France; 7AP-HP, service de maladies infectieuses et tropicales, Hopital Tenon, Paris F-75020, France; 8AP-HP, service de medecine interne, Hopital Saint-Louis, Paris F-75010, France;
9AP-HP, service de maladies infectieuses et tropicales, Hopital Saint-Louis, Paris F-75010, France; 10Hospices civils de Lyon, Hopital de l'Hotel-Dieu, Service d'Hepatologie, Lyon, F-69288 France
ABSTRACT
Background: It is not known whether the early HBV plasma dynamics in HIV-HBV co-infected patients is different according to treatment with TDF or ADV
Methods: HIV-HBV co-infected patients recruited in the HIV-HBV French cohort study were prospectively followed and assessed for biochemical and virological responses to ADV or TDF. Patients were HBV treatment-naive at inclusion, except for past or current Lamivudine, as part as antiretroviral regimen. Plasma Viral dynamics were modelled using random-effects, mixed-linear models
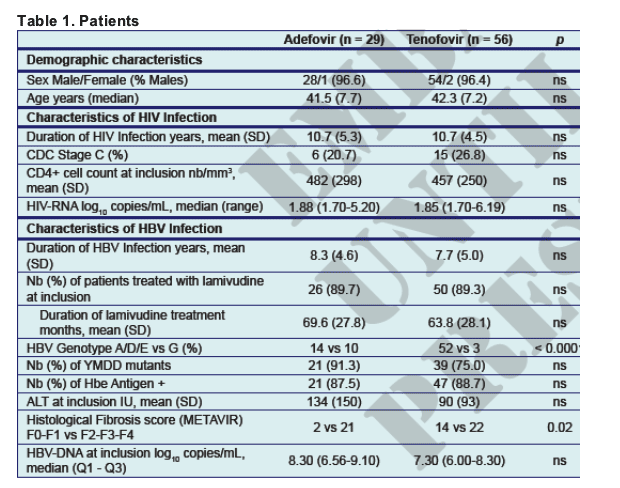
Results: A total of 85 patients were included, 29 in the ADV arm and 56 in the TDF arm. Both groups did not differ at inclusion, except for HBV genotypes (41.7% of genotype G in ADV group vs 5.4% in TDF group, p<0.003). During the first 6 months, there was an overall decrease of HBV load by 1 log unit (CI 95% [0.9, 1.2]) per month. Individuals in the TDF group presented a more rapid decline, with a declining slope estimated at 1.1 (CI95% 0.9-1.3) compared to 0.8 (CI 95% 0.6-1.0) in the ADV group. (p=0.036).
After 12 months of treatment, the HBV-DNA load, although similar at baseline (7.98log and 7.30log in the ADV and TDF groups respectively), declined to a
lower level in patients treated with TDF (2.45log vs 3.97log, p<0.0001).
A marked decrease in the rate of transaminases was noted in the TDF group but not in the ADV group (mean ALT (TDF) = 46 UI/mL vs mean ALT(ADV) = 83 UI/mL, p=0.001). The proportion of patients with HBe seroconversion was identical in both groups (4/29 with ADV vs 7/56 with TDF).
Conclusions: The HBV-DNA declined less rapidly under ADV than TDF treatment. The use of TDF was associated with a better biochemical and antiviral response. Viral HBV dynamics under both treatments are in favour of using TDF rather than ADV when controlling the HBV replication in HIV-infected patients is concerned.
Background
Roughly 6-9 % of HIV-infected individuals in the Western world are also infected with Hepatitis B virus (HBV) and liver-related mortality significantly increases with co-infection1
Because of its dual activity, lamivudine has been widely used to treat both HIV and HBV, but HBV resistance occurs in 90% of patients after 4 years2 and needs for new molecules are wide.
Adefovir (ADV) and Tenofovir (TDF) are two reverse transcriptase nucleotide inhibitors that are effective in reducing HBV-DNA for both wild type and lamivudine-resistant HBV strains in HIV-HBV co-infection.
Recent data suggest that TDF is not inferior to ADV in inhibiting the HBV replication.3
However, it is not known whether early viral dynamics of HBV differ according to the treatment with TDF or ADV.
Objective
The aim of the present study is to study the biological changes during treatment with either ADV or TDF and to compare the HBV dynamics in HIV-HBV co-infected patients.
Methods
Study Design
Non-randomized, open-label, multi-center prospective study on the HBV-DNA viral dynamics during the course of ADV or TDF treatment
Patients
Patients followed-up in the 2002 - 2006 French HIV-HBV Cohort Study were enrolled in the present study according to the following inclusion criteria:
- HIV enzyme linked immunoabsorbant assay (ELISA) positivity confi rmed by a Western-Blot
- HBsAg seropositivity
- Prior HBV treatment limited to lamivudine
- Baseline HBV-DNA viral load of at least 1000 copies/mL (c/mL)
- Initiation of a therapeutic regiment containing either ADV 10 mg QD or TDF 300 mg QD, either associated with or without lamivudine
Modeling HBV DNA Dynamics
The first six months of viral dynamics were modeled using mixed linear analysis, where the log viral load in treatment group i, individual j and time tij was:
- log(CVijk) = _i + _itjk + aj + bjtjk
- and _i and _i are the treatment-dependant, population-averaged intercept and slope of viral load decrease, and aj, bj are random subject-dependent intercept and slope with joint, binormal distribution. Censoring due to viral loads below the detection level was taken into account
Undetectable HBV Viremia during Follow-up
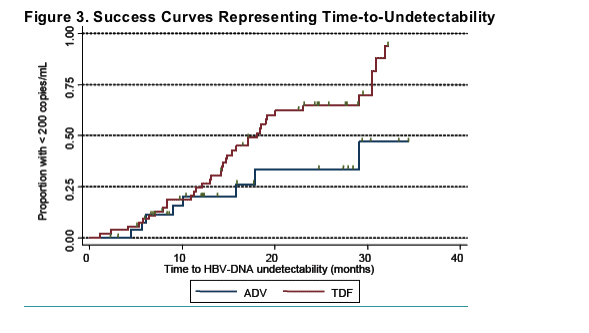
Using the time to HBV-DNA undetectability (< 200 c/mL) as the clinical endpoint, treatments were compared by Kaplan-Meier curves. The hazards rate ratio, comparing the rates of undetectable HBV-DNA between treatments, was calculated using Cox proportional hazard model, then subsequently adjusted for the factors signifi cantly associated with time to undetectability in univariate analysis.
RESULTS
Biological Changes over 12 Months
4 patients (14%) in the ADV had lost HBeAg at 12 months, and 7 (12.5%) in the TDF group.
Only TDF demonstrated a significant decrease in ALT and AST levels from baseline to 12 months (avg ALT: 90 to 46 UI/l; paired t-test, p = 0.001 and avg AST: 66 to 33 UI/l, paired t-test p = 0.001).
No significant changes in biochemical markers were noted in the ADV group.
Although there was no difference between TDF and ADV on ALT and AST at baseline, ALT and AST levels were lower with TDF than ADV (46 vs 83UI/L, p = 0.003, and 33 vs 72UI/L, p = 0.01).
HBV Viral Changes during Follow-up
The median duration of therapy was 775 days (IQR: 396 - 1008) for ADV and 1050 days (IQR: 797 - 1196) for TDF.
At 12 months, the HBV-DNA load, although equivalent at baseline (avg HBV-DNA c/mL: 7.91 log and 7.27 log in the ADV and TDF groups respectively), declined to a lower level in patients treated with TDF (avg HBV-DNA c/mL: 2.45 log vs 3.87 log, p = 0.0001).
A smaller number of patients had a substancially stable HBV-DNA level in the TDF group compared to the ADV group: 4 patients (7.1%) on TDF vs 7 patients (24.1%) on ADV had a change of less that -2 log10 c/mL HBV-DNA (Pearson's chi-square p = 0.03) 66 % (37/56) of TDF-treated vs 28 % (8 of 29) ADV-treated patients reached undetectable HBV.
HBV Dynamics Modeling
Figure 2. Modeling of HBV-DNA Decrease between Inclusion and 6 Months according to Treatment
--The lines represent the fit of the mixed linear model according to treatment
--In patients treated with TDF, HBV-DNA declined more rapidly, with a slope of
decline estimated at 1.1 (95% CI [0.9 - 1.3]) compared to 0.8 (95%CI [0.6 - 1.0]) in patients treated with ADV (p = 0.0036).

Impact of treatment on time to HBV DNA < 200 c/mL
The Kaplan-Meier analysis showed a significant difference in the impact of TDF vs ADV on time to undetectability, in favor of TDF (log-rank test, p = 0.04, HR: 2.22 [95%CI:1.03-4.77])
In univariate analysis, only HBeAg status, HBV-DNA level and ALT at baseline had an impact on HBV time-to-undetectability (no influence of YMDD, CD4 cells count, fibrosis score, co-treatment with Lamivudine, alcohol consumption).
After adjusting for the presence of HBeAg, log HBV DNA at baseline, and ALT levels divided at the median, TDF was still significantly associated with a higher rate of HBV DNA undetectability (HR = 2.79; 95 % CI : 1.14 - 6.84, p = 0.025).
Conclusions
In the context of HIV-HBV co-infection, when compared to ADV:
-- TDF had a more marked action on transaminases normalization;
-- TDF was associated with a more rapid decline in HBV-DNA during the first 6 months of treatment
-- TDF-treated patients reached undetectable levels of HBV-DNA at a significantly higher rate
-- After adjustment of HBeAg status, HBV-DNA at baseline and level of ALT at baseline, TDF was still associated with a higher rate of patients achieving undetectable HBV-DNA levels
However, randomized control trials, specifi cally designed to test drug superiority, will be necessary in order to clearly confirm a treatment difference.
|
| |
|
 |
 |
|
|