 |
 |
 |
| |
HIV Pathogenesis in Humans & Monkeys
|
| |
| |
CROI, Feb 2007, Los Angeles
Reported by David Margolis, MD, University of North Carolina
Guts and Monkeys and Monkey's guts: HIV pathogenesis seen through an endoscope:
Most immune cells live in tissue, only occasionally migrating to perambulate in the bloodstream. And 98% our immune cells live in the tissue where we contact the outside world: in the skin and the mucosal tissue, most notably the roughly 30-foot long stretch of our small and large intestine that contains 50% of our immune cells.
Some years ago Ron Veazey observed a prompt and profound depletion of CD4 cells in the intestinal mucosa of SIV-infected monkeys. The recognition in the last 2 years or so that the human immune system takes a tremendous slug in the gut in the first few weeks of infection has completely revised current paradigms of the progression of HIV infection to AIDS.
Gut CD4s recover after ART:
Shenefelt presented a study of the recovery of CD4 cells in gut-associated lymphoid tissue in patients given ART (abstr. 28;http://www.retroconference.org/2007/Abstracts/29705.htm). Previous studies suggest that reconstitution of CD4+ T cells in antiretroviral therapy (ART)-treated individuals is less complete in GALT than in peripheral blood.
14 drug-naive patients were started on ART containing nevirapine, abacavir, AZT, and 3TC. CD4 cell counts at initiation were < 100/µl in all. Of the 14 enrollees, only 3 women and 5 men remained in the study, 6 others were lost to follow-up. Viremia was suppressed to <50 copies/ml in all but one subject at week 48, who still had low-level viremia. CD4 cell counts increased in all to ca. 200 cells/µl.
Differing with prior studies of GALT and the effect of ART, 4-um sections of GALT biopsies taken from the colon were stained with CD3 and CD4 antibodies, and counted by immunofluorescent microscopy. Numbers of lamina propria CD4+ T cells/mm3 were calculated. Mean GALT CD4+ T cells/mm3 were 279 at baseline, as expected higher than that in peripheral blood. Mean GALT CD4+ T cells increased through week 24 to 890 cells/mm3. This increase was greater than that observed in peripheral blood. Mean GALT CD4+ T cells decreased to 759 cells/mm3 at week 48, but remained significantly different than baseline and still reflected a larger increase than in peripheral blood. These increases in CD4+ T cells are significantly larger in GALT than peripheral blood, particularly during the first 24 weeks of therapy, a finding of immune reconstitution that has not been seen in other studies.
However, during the Q&A the study was criticized on methodological grounds. Mehandru from the Aaron Diamond pointed to a study their group had just published (Mehandru S et al. Lack of Mucosal Immune Reconstitution during Prolonged Treatment of Acute and Early HIV-1 Infection. PLoS Med. 2006; 3:e484), in which patients treated as early as 2 weeks after acute HIV infection, and for up to 7 years did not have full CD4 reconstitution as measured by both microscopy and flow cytometry of colonic GALT biopsy samples.
Most gut CD4s do not recover after most ART:
A similar study presented by Estes (Abstr. 67) presented a more nuanced picture. This group compared lymphocytes populations in inguinal lymph nodes and ileal biopsies in 32 HIV-infected and 11 uninfected volunteers. After ART was given, second biopsies were obtained after 6 months in 15 of the HIV+ patients. Total, naive, central and effector memory CD4 populations in were measured in peripheral blood, lymph nodes, Peyers patches, and lamina propria using flow cytometry and quantitative microscopy.
As expected, CD4 cells were depleted wherever Estes and colleagues looked prior to the initiation of ART. There were significant increases in CD4+ cells after 6 months of ART in peripheral blood and lymph node, but not in the ileal lamina propria. However, in Peyers patches, the inductive site where naive and central memory CD4+ T cells in GALT reside, there was a significant increase in central memory CD4+ T cells unless ART had been delayed until the patient had late stage disease (stated as clinical AIDS, but clinical details not presented). On the other hand, if therapy was started in the acute/early increases in central memory CD4+ T cells were the greatest. As clinical details were not given, it was unclear to this author how much reconstitution could be expected in GALT central memory cells if ART was initated according to current guidelines.
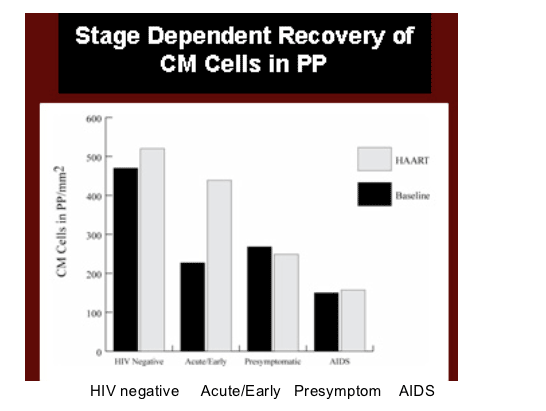
Estes said that lymphatic tissues do not uniformly reconstitute CD4+ T cells with ART. Lymph node and Peyers patches are capable of limited reconstitution. This was most striking for central memory CD4 cells in Peyers patches.
Why do some monkeys and some people not get AIDS?:
A large group of investigators led by Guido Silvestri at UPenn (abstr. 69) studied SIV infection in sooty mangabeys. Naturally exposed sooty mangabeys do not develop immunodeficiency after SIV infection despite chronically high viral loads. However, CD4 cells in these SIV-infected monkeys have a short half-life, as is seen in HIV and pathogenic SIV infection.
Silvestri and colleagues infected 5 sooty mangabeys with the SIVsmm strain that was derived from a naturally SIV-infected sooty mangabey, and had not been cloned or manipulated in the laboratory. They found a variable but significant depletion of intestinal GALT CD4+ T cells, as well as depletion of CD4+ cells in rectal mucosa, broncho-alveolar lavage, lymph nodes and peripheral blood. While only a minor decline in the level of CD4+ T cells in peripheral blood and lymph nodes was observed, all animals underwent a rapid, significant, and persistent depletion of CD4+ T cells in both rectal mucosa and broncho-alveolar lavage. This depletion involved all subsets of CD4+ T cells, including the CD28-CD95+ "effector" T cell subtype, thought to be important in recognition and clearance of HIV-infected cells. This depletion was temporally associated with a moderate increase in systemic and mucosal immune activation. The time course of this depletion was similar to that seen in other monkey species in which SIV infection can lead to AIDS. However, this activation in circulating blood cells and rectal biopsy tissue was found to decrease 30 to 50 days after infection.
All mangabeys remained asymptomatic. An early increase in plasma lipopolysaccharide (LPS) levels was seen in two of the infected mangabeys that also had the most profound CD4+ T-cell depletion in rectal mucosa. It was felt that this might indicate a transient loss of mucosal immune barrier integrity. In humans and other monkey species, it has been proposed that chronic immune activation induced by microbial products such as LPS occurs after HIV/SIV-induced mucosal damage, and is a key factor in progressive immune depletion. However, the innate inflammatory response of mangabey cells to bacterial byproducts was significantly reduced compared to to that of rhesus macaques. The authors concluded that sooty mangabeys might not develop AIDS, while rhesus macaques do develop AIDS after SIV infection, because mangabeys have an attenuated inflammatory response to the systemic translocation of intestinal microflora that follows SIV infection.
Observations reported by Saurabh Mehandru from the Markowitz group at the Aaron Diamond (abstr. 318) appear to fit the same general model of HIV pathogenesis. The group reported findings in 11 long-term non-progressors (LTNP; patients remaining healthy for several years without CD4 depletion and without the need for ART) and spontaneous controllers (SC; patients soon after infection with low viral loads). Findings in blood and rectal mucosal GALT biopsies were compared to those in HIV-uninfected controls (n = 18), primary HIV infection (PHI; n = 32), and chronic HIV infection (CHI; n = 10) served as controls. Mucosal lymphocytes were assessed both by flow cytometry and microscopy. Cellular HIV DNA was quantitated by PCR, and viral expression was examined using an ultrasensitive RNA PCR.
CD4+ T cells were more depleted in the rectal mucosa of LNTP and SC than in controls without HIV infection (38.7 vs 56.4% CD4 cells, or 8.4 vs. 11.1 absolute CD4+ cells/unit area). During PHI, a high frequency of proviral DNA+ cells was observed in the 2 SC patients studied (34% and 32% proviral DNA+ cells), suggesting that initial control of HIV replication in the GALT was not uniquely controlled in SC. However, among this group of LTNP and SC, the percentage of rectal GALT activated memory CD8+ T cells and CD4+ T cells were comparable to those of HIV- patients, and significantly lower than those of PHI who were not SC, and of CHI patients.
Such LNTP and SC patients might then be thought of as similar to sooty mangabey monkeys, in whom SIV replicates well in the GALT and results in cell activation, and ultimately destruction. However, for unknown reasons, in those monkeys or people not fated to develop AIDS, T cell production overcomes T cell destruction, initial cellular activation does not persist, and later CD4 depletion does not develop. Whether host factors, viral factors, or both contribute to this phenomenon is not yet known. The GALT is not a well-understood place. It will take more work to piece the observations made so far by different techniques, in different parts of the GI tract, in different animal models, and in small human cohorts, into a coherent picture of AIDS pathogenesis.
Clinical
Clinical markers to predict CD4 decline or clinical AIDS: describing different ends of the elephant:
Rodiguez and colleagues recently analyzed the ability of HIV viral load to foreshadow a decline of CD4+ T cells (Predictive value of plasma HIV RNA level on rate of CD4 T-cell decline in untreated HIV infection. Rodriguez B, et al. JAMA 2006; 296:1498-506). This report generated wide interest, as the self-titled "Bad Boys of Cleveland" found that plasma HIV-1 RNA explained less than 10% of yearly CD4 cell decline. This result led the researchers to suggest that factors other than viremia drive CD4 cell losses in HIV infection.
This led John Mellor's (abstr. 139) to re-evaluate the predictors of CD4 cell decline and the occurance of AIDS-defining events among 1679 HIV-1+ men enrolled in the Multicenter AIDS Cohort Study (MACS). Viral load is well correlated with mean CD4 cell decline (Fig 1). But there is a great deal of variation in CD4 cell decline from patient to patient, and the quantity of plasma viremia does not explain this variation (Fig. 2) as illustrated by the fact that the correlation statistic (called R-squared) is low.
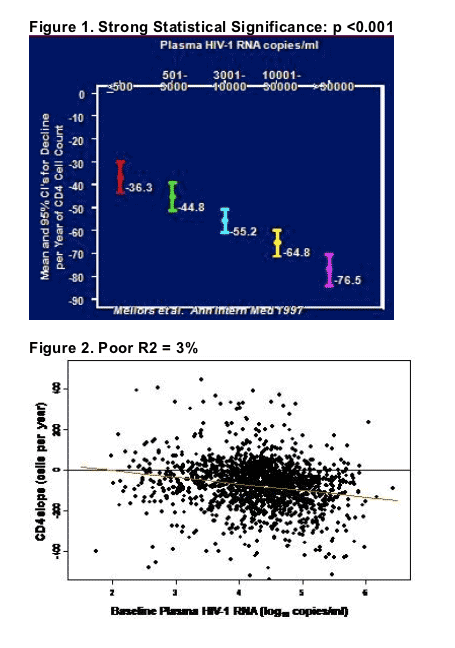
However, Mellors found that overall, a single HIV-1 RNA measurement was the best predictor of clinical AIDS events, explaining 45% of the variability in time to AIDS, compared with only 20% for CD4 cell count. The addition of baseline CD4 to viral load improved the prediction of AIDS somewhat (51%). A cellular activation marker (CD38+) was predictive of AIDS, but strongly correlated with HIV-1 RNA.
In the end, it seems that both groups have made and re-made important points. Due to the variability of CD4 counts, HIV RNA is the best predictor of clinical progression. Baseline CD4 cell count adds to this. HIV RNA by itself is not an excellent predictor of CD4 cell loss, and so better predictors of short-term CD4 cell loss would be useful to define. And clearly, both viremia and host immune parameters play a role in HIV pathogenesis.
|
| |
|
 |
 |
|
|