 |
 |
 |
| |
Tipranavir & TMC114 Not Cross-Resistant in Clinical Isolates Study: An Investigation into the influence of the Tipranavir-associated V82L/T mutations on the susceptibility to darunavir and brecanavir
|
| |
| |
Reported by Jules Levin
14th Conference on Retroviruses and Opportunistic Infections
25-28 February 2007, Los Angeles, CA.
RC Elston1, DR Kuritzkes2, R Bethell1
1Boehringer Ingelheim (Canada) R&D, Laval, Canada; 2Brigham & Women's Hospital, Boston, MA, USA
".....The object of this analysis was to investigate the change in phenotypic susceptibility to DRV and BCV of clinical isolates of HIV-1 from patients who experienced virological rebound while on a TPV-based regimen...... Cross resistance is a critical issue when optimizing a treatment regimen for a multi-PI experienced patient. Previously it was reported that the majority of patients who are susceptible to TPV at baseline, but received DRV and experienced virological failure on a DRV based regimen would remain susceptible to TPV [9]. In this analysis, we have confirmed that this is equally true if patients received a TPV based regimen and experienced virological rebound. No isolate that was fully susceptible to DRV (<10-FC) prior to TPV therapy developed resistance (>40FC) to DRV irrespective of the resistance mutations selected by TPV. The majority (84%) remained fully susceptible....The observation that the majority of isolates remained susceptible to DRV after treatment with TPV is consistent with earlier reports that these two agents have dissimilar resistance profiles....."
Introduction
The development of cross-resistance between HIV-1 protease inhibitors (PI) following virological failure of multiple PI-containing regimens poses a significant problem when constructing subsequent regimens. Two recently approved PIs, Tipranavir (Aptivus�) and Darunavir (Prezista) have improved activity against
multi-PI resistant viruses and thereby offer realistic sequencing options.
Tipranavir (TPV) is structurally distinct from earlier PIs and requires multiple PI mutations before significant levels of resistance occur [1,2]. Virological rebound on a TPV-based regimen is most commonly associated with the development of the V82L or V82T mutation.
Darunavir (DRV) is chemically related to the previously approved PI, amprenavir and also to brecanavir (BCV) whose clinical development was recently discontinued (Figure 1) [3]. The improved potency of DRV and BCV relative to APV is due, at least in part, to the addition of a bis-tetrahydrofuran moiety [3]. The improved potency results in reduced impact of PI-associated mutations, although it has been reported that mutations associated with APV resistance are also associated with a reduced virological response to DRV [4].
Significantly, TPV and DRV are structurally diverse and have limited overlap in the mutations used to predict virological response (overlapping mutations: L33F, I47V, I54M, I84V) [5,6]. Furthermore mutations selected by TPV are mostly not selected by DRV and vice versa [5,6]. Consequently resistance testing should guide physicians to choose between the two PIs in order to construct the optimal
regimen to treat a patient harboring multiple-PI resistant virus. The object of this analysis was to investigate the change in phenotypic susceptibility to DRV and BCV of clinical isolates of HIV-1 from patients who experienced virological rebound while on a TPV-based regimen.
Methods and materials
Matched baseline and virological failure samples for isolates developing protease resistance mutations following virological rebound on a TPV-based regimen were evaluated for susceptibility to TPV, DRV and BCV using the PHENOSCRIPT� Assay (Eurofins Viralliance, Paris). Where two on-therapy plasma samples existed, the later sample was analyzed. Samples were provided by patients participating in the Phase III RESIST 1 (1182.12 conducted in the US, Canada, Australia) and RESIST 2 (1182.48 conducted in Europe and South/Central America) clinical trials. Genotypes were obtained by commercially available population sequencing. Phenotyping (Antivirogram assay) had previously been performed; however, this assay never included DRV or BCV in the protease panel. DRV and BCV were synthesized by Boehringer Ingelheim and supplied in a blinded fashion to Eurofins Viralliance. Fold change (FC) in susceptibility of viruses to drugs was determined relative to HIV-1NL4-3. Clinical cut-offs for DRV have been previously established for the Antivirogram Assay [7). For purposes of example these cut-offs were used in Figure 2.
Results
Patient accountability
Sixty-two matched patient samples were included in this analysis representing patients identified as experiencing virological rebound whilst receiving a TPV-based regimen at the first data cut-off. In this analysis, 41/62 matched patient samples were selected for further phenotypic evaluation to determine the susceptibility to DRV and BCV following virological rebound of a TPV-based regimen. Data was successfully generated for 39/41 matched patient samples.
In order to fully understand changes in susceptibility caused by the development of TPV resistance, samples from the remaining 21 isolates (21/62) were excluded from this analysis due to the absence of development of protease mutations and/or decreases in TPV susceptibility, the presence of mixtures at position V82, or the baseline presence of V82T/L (Table 1).
Failure of a TPV containing regimen was most often accompanied by the development of the TPV signature mutations V82L or V82T (29/62, 47%). In the absence of development of V82L/T mutations, failure was accompanied by either no genotypic changes (6/62, 10%) or development of alternative resistance pathways not involving V82T/L (27/62, 43%). Of note, the possibility that the V82L/T mutation would have developed had additional on-therapy time points been available cannot be excluded.
Summary of protease mutations developing or lost following
virological rebound of a TPV-based regimen
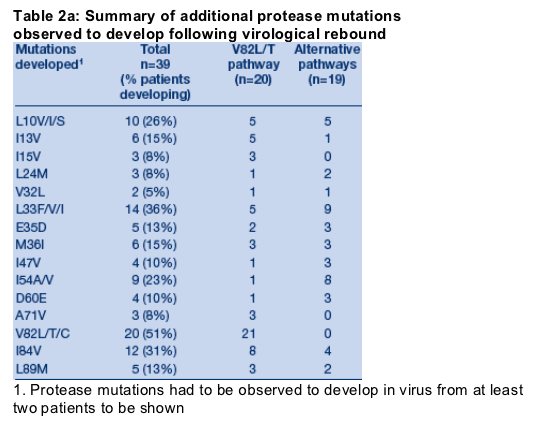
Virological rebound while the patient was receiving a TPV containing regimen was often accompanied by the development (Table 2a) and loss (Table 2b) of multiple protease mutations. Of note, patients failing therapy with virus containing the V82L/T mutations had lower incidence of the I84V mutation at baseline (V82L/T: 2/21, 9%; non V82L/T: 11/19, 58%). In the absence of selection of V82L/T, the most commonly selected mutations were L33F and I54V/A.
Interestingly, mutations L10F, L24I, I50V, L76V and V82A if present at baseline, were subsequently lost in the majority of cases upon virological rebound.

Baseline and on-therapy changes In susceptibility to PIs including TPV, DRV and BCV following virological rebound
-- Analysis of baseline phenotypic susceptibility
- Isolates had lower fold change to TPV (2.1-fold) compared to other PIs including DRV (12-fold) and BCV (11-fold). This is consistent with a previous report [8].
- Median fold changes in susceptibility to APV, DRV and BCV were very comparable.
- Higher baseline resistance to protease inhibitors including TPV was observed in isolates that went on to develop TPV resistance in the absence of V82L/T mutations.
-- Susceptibility of isolates to TPV after virological failure
- As expected, virological failure while on a TPV containing regimen led to substantial decreases in susceptibility to TPV (FC 2.1 to 28).
- Virological rebound with development of the V82L/T mutation was associated with greater reductions in susceptibility to TPV.
-- Susceptibility of isolates to other PIs after virological failure of TPV
- Overall, failure of a TPV containing regimen was not associated with decreased susceptibility to any other protease inhibitor with the possible exception of ATZ. Modest increases in susceptibility to LPV, APV, DRV and BCV were observed.
- With the exception of ATZ, development of the V82L/T pathway mutations led to increased susceptibility or no change in susceptibility to all other PIs including DRV and BCV.
- Virological rebound in the absence of V82L/T led to increases in susceptibility to all protease inhibitors.
Table 3: Summary of median phenotypic susceptibility (fold change) of clinical isolates before and after virological rebound of a TPV containing regimen.
Of note, in the table below TMC114 phenotypic resistance before viral rebound to TPV was 6-20 fold and after viral rebound to TPV fold-change in phenotypic resistance to TMC114 was 6-12 fold. In each of the 3 situations described in the table TMC114 phenotypic resistance improved or was the same after TPV rebound. Susceptibility to TMC114 varied depending on the presence of V82L/T. Susceptibility of isolates before and after viral rebound of TPV with development of V82L/T mutations were 6-fold and 6-fold, but without development of V82L/T susceptibility to TMC114 was 20-fold before TPV rebound and 12-fold after.

Detailed analysis of change in susceptibility to DRV following
virological bound of TPV
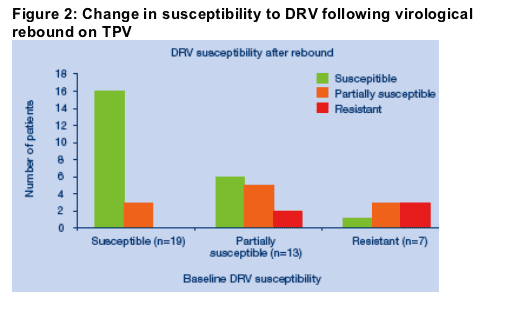
Using clinical cut-offs for DRV of <10FC for Susceptible and >40FC for Resistant [7] at baseline there were 19 isolates that were susceptible to DRV, 13 isolates that were partially susceptible to DRV and seven isolates that were classified as resistant to DRV. The change in susceptibility to DRV after failure of TPV in summarized in Figure 2.
Of the 19 isolates that were fully susceptible to DRV at baseline, following virological rebound on TPV, 16/19 (84%) remained fully susceptible. No isolate developed resistance to DRV.
Of the 13 isolates with partial susceptibility to DRV, six isolates (6/13, 43%) became fully susceptible to DRV after TPV therapy, whereas two isolates developed resistance.
Four of the six isolates becoming fully susceptible lost known DRV associated mutations (I50V, L76V) after virological failure of TPV.
Of seven isolates that were resistant to DRV at baseline, four developed increased susceptibility to DRV following virological rebound of TPV. Three of these viruses had lost known DRV associated mutations (I50V or I54L/M).
Correlation between susceptibility of TPV, APV, BRC and DRV
Overall there appeared to be minimal overlap between the resistance profile of TPV and DRV. To explore the correlation between reduced susceptibility to TPV, APV, DRV and BCV further, the FC of all baseline (n=39) and on-therapy (n=39) isolates were plotted for pairs of PIs (BCV:DRV, BCV:APV, APV:DRV, TPV:DRV;
Figure 3). No correlation was observed between TPV and either APV, DRV or BCV. A stronger correlation was observed between resistance to APV and resistance to DRV, than between APV- and BCV-resistance. This finding may reflect the higher degree of chemical similarity between APV and DRV than between APV and BCV. No correlation was observed between resistance to TPV and resistance to any other PI (data not shown).
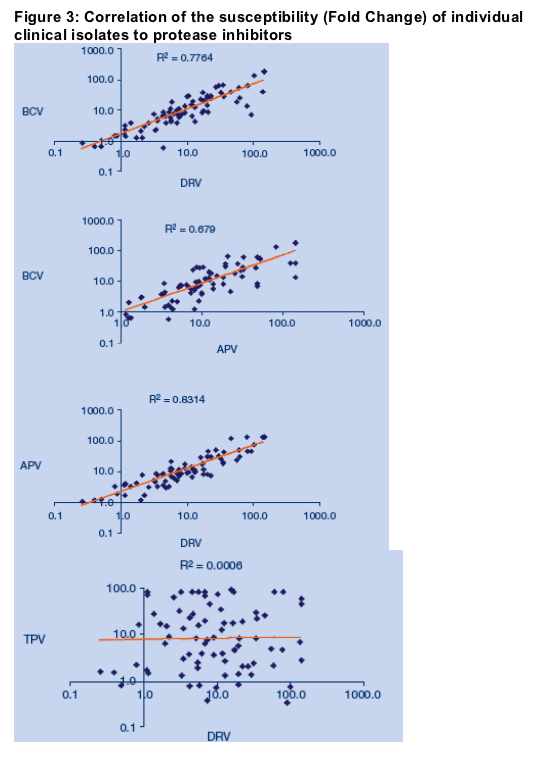
Discussion
Cross resistance is a critical issue when optimizing a treatment regimen for a multi-PI experienced patient. Previously it was reported that the majority of patients who are susceptible to TPV at baseline, but received DRV and experienced virological failure on a DRV based regimen would remain susceptible to TPV [9]. In this analysis, we have confirmed that this is equally true if patients received a TPV based regimen and experienced virological rebound. No isolate that was fully susceptible to DRV (<10-FC) prior to TPV therapy developed resistance (>40FC) to DRV irrespective of the resistance mutations selected by TPV. The majority (84%) remained fully susceptible.
Interestingly, virological rebound on a TPV based regimen often resulted in extensive changes in the viral genotype. Mutations were both selected and deselected from the predominating viral population. These changes led to resistance to TPV, but sometimes resulted in increased susceptibility to APV, DRV and BCV. It is likely that this arose through the loss of known APV/DRV associated mutations such as L10F, I54L, I50V, and L76V. The observation that
the majority of isolates remained susceptible to DRV after treatment with TPV is consistent with earlier reports that these two agents have dissimilar resistance profiles.
Optimizing the use of TPV and DRV in multi-PI experienced patients is critical to the successful use of these agents in this patient population. Given the apparent limited overlap in the resistance profiles of these agents it is likely that there will be times when one agent would be preferred over the other based upon resistance mutations present. Given the strong correlation between phenotypic susceptibility to DRV and APV, the impact APV mutations have on virological response to DRV [4], and the deselection of some of these mutations by TPV; the data suggest that patients harboring certain APV-associated mutations may benefit more from a TPV-based regimen than a DRV-based regimen.
Conclusions
- Overall failure of a TPV-based regimen had minimal consequences on the susceptibility to other PIs, including DRV.
- The majority (84%) of isolates that were susceptible to DRV at baseline remained susceptible after TPV therapy.
- In agreement with their chemical similarity, a high degree of cross resistance was observed between APV and DRV.
References
1. Larder et al (2000) AIDS 14:1943-1948
2. Doyon et al (2005) Antiviral Research 68:27-35
3. Ghosh et al (2006) ChemMedChem 1:939-950
4. De Bethune et al (2006) CROI, Feb 5-8th, Denver, USA
5. Aptivus Package Insert - TPV mutations Score
6. Darunavir Package Insert - DRV Mutation score
7. De Meyer et al (2006) Poster 73, 15th Int. HIV Drug Res. Workshop, 13-17th
June, Sitges, Spain.
8. Craig et al (2006) Poster THPE0023, 16th int. AIDS Conf. 13-16th 9. Aug,
Toronto, Canada.
9. De Meyer et al (2006) Poster 42, 4th European HIV Drug Res. Workshop,
29-31 Mar., Monte Carlo, Monaco.
|
| |
|
 |
 |
|
|