 |
 |
 |
| |
Simpler genotype score to predict response to tipranavir
|
| |
| |
CROI, Feb 2007, Los Angeles
With virologist colleagues, Boehringer researchers famously advanced a mutation score involving substitutions at 16 protease positions to predict response to tipranavir/ritonavir in PI-experienced people [3]. At the Retrovirus Conference Anne-Genevieve Marcelin (Pitie-Salpetriere Hospital, Paris) and colleagues at other centers detailed a score with substitutions at only five protease positions that may make predicting response easier [4]. Marcelin and coworkers cautioned, though, that their new mutation formula must be validated in other cohorts.
The new score rests on analysis of 143 people who had tried a median of four PIs (interquartile range three to five) and had a median of three major and nine minor mutations in protease according to the IAS-USA scheme. Everyone had a viral load above 1000 copies after at least 3 months of treatment with tipranavir/ritonavir and no other PIs.
Defining virologic response as at least a 1-log (10-fold) drop in viral load by treatment month 3 or a load below 200 or 50 copies at month 3, Marcelin counted 79 people (55%) with a virologic response to tipranavir/ritonavir. Mutations that appeared in more than 10% of pre-tipranavir genotypes and correlated with response at a P value less than 0.10 underwent further statistical analysis to pick the mutation cluster most strongly tied to virologic response. This statistical test correlated the mutations M36I/L/V, Q58E, H69I/K/N/Q/R/Y, and L89I/M/R/T/V with a worse virologic response and F53L/W/Y with a better response.
The earlier Boehringer score included three of those mutations, at positions 36, 58, and 69 [3]. Mutations at position 89 imperiled virologic response in a data set analyzing phenotype/genotype correlations [3]. And the F53L mutation turned up in a Virco analysis of mutations linked to tipranavir response [5].
Marcelin combined these mutations to yield a score of 36 - 53 + 58 + 69 + 89. All 7 people with a score of -1 were 3-month virologic responders, compared with about 80% of 33 people with a score of 0, 60% of 63 people with a score of 1, 40% of 24 people with a score of 2, 20% of 14 people with a score of 3, and neither of 2 people with a score of 4 (P = 1.47 x 10[-7]). Multivariate analysis determined that a mutation cutoff score of 2 correlated with virologic response at an odds ratio of 6.8 (P < 0.001). Previous enfuvirtide independently raised the risk of nonresponse 3.99 times (P = 0.0015), regardless of enfuvirtide use in the tipranavir regimen. Combining efavirenz with tipranavir independently lowered odds of virologic failure 86% (odds ratio 0.14, P = 0.035).
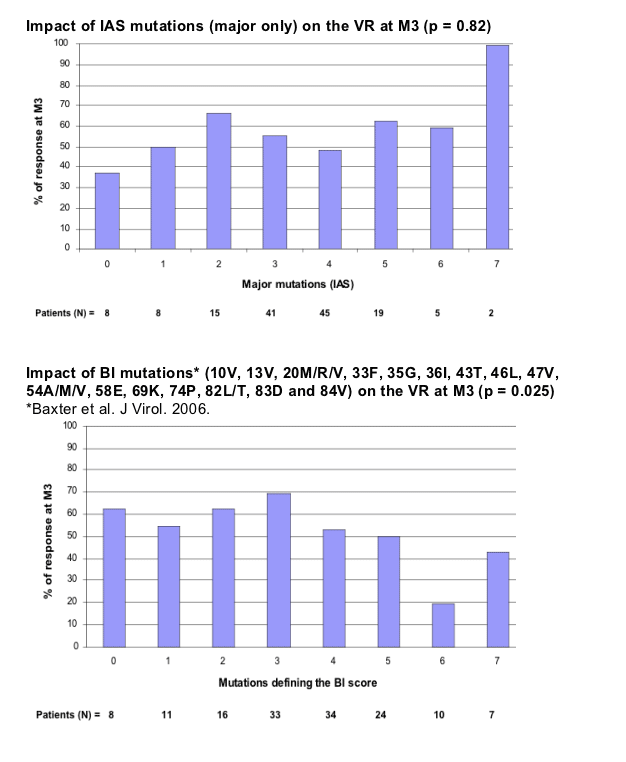
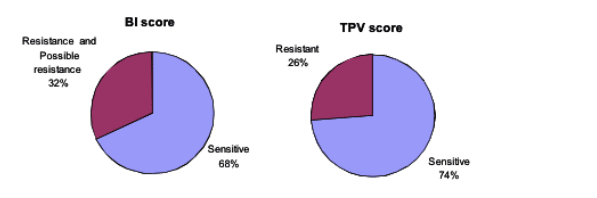
According to the Boehringer score, four or more mutations at the 16 protease positions mean virus is resistant or possibly resistant to tipranavir/ritonavir. Fewer than four mutations at those mutations predict the virus will be sensitive to tipranavir/ritonavir. Comparing the new score with the Boehringer score in 473 people with virologic failure in 2006, Marcelin found that the new score calls 74% of the viral samples sensitive to tipranavir and 26% resistant. The Boehringer score calls 68% of the same isolates sensitive to tipranavir and 32% resistant or possibly resistant. Thus the new score is simpler and eliminates the gray zone of "possible resistance." Lack of any major PI mutations in the new score, Marcelin proposed, may explain why tipranavir retains activity against viruses containing major mutations.
Besides Marcelin's group, Boehringer, and Virco, the FDA also recently proposed a pre-tipranavir resistance score to predict response to tipranavir/ritonavir [6]. Analyzing genotypes from the RESIST trials, the FDA found that five or more mutations at positions 13, 32, 36, 47, 58, 60, 82, and 84 foretold a worse 24-week response to tipranavir/ritonavir. People with five or more of these mutations and not combining enfuvirtide with tipranavir had a median 24-week viral load drop of 0.86 log, compared with more than a 1.5-log drop with five or more of these mutations in people who took both enfuvirtide and tipranavir.
Mutations at only one protease position, 36, overlap in the FDA and Marcelin recipes. These divergent results underline the difficulty of predicting response with mutation scores, which vary considerably with the patient populations and statistical methods used to derive them.
References
3. Baxter JD, Schapiro JM, Boucher CAB, et al. Genotypic changes in human immunodeficiency virus type 1 protease associated with reduced susceptibility and virologic response to the protease inhibitor tipranavir. J Virol. 2006;80:10794-10801.
4. Marcelin AG, Masquelier B, Descamps D, et al. Mutations associated to response to boosted tipranavir in HIV-1 infected protease inhibitor experienced patients. 14th Conference on Retroviruses and Opportunistic Infections. February 25-28, 2007. Los Angeles. Abstract 612.
5. Bacheler L, Vermeiren H, Winters B, et al. Clinically relevant phenotypic resistance and cross resistance to tipranavir among recent routine clinical isolates. 4th European HIV Drug Resistance Workshop. March 29-31, 2006. Monte Carlo. Abstract 40.
6. Naeger LK, Struble KA. Food and Drug Administration analysis of tipranavir clinical resistance in HIV-1-infected treatment-experienced patients. AIDS. 2007;21:179-185.
"Mutations associated to response to Boosted Tipranavir in HIV-1 Infected Protease Inhibitor Experienced Patients"
AG Marcelin1, B Masquelier2, D Descamps3, J Izopet4, C Charpentier5, C Aloui6, G Peytavin3, M Lavignon7, P Flandre1, V Calvez1 on behalf the ANRS TPV study group
1 CHU Pitie-Salpetriere, 2 CHU Bordeaux, 3 CHU Bichat-Claude Bernard, 4 CHU Toulouse, 5 CHU Georges Pompidou, 6 CHU Avicennes, 7 Boeringher-Ingelheim, France.
Author Conclusions :
"This study based on genotype/ VR correlation identified a genotypic score for TPV/r which was shown to be more predictive than taking into account only IAS mutations or a score based on genotype/phenotype correlation mixed with genotype/VR correlation. Thus, it could be taken into account in genotypic interpretation systems rules and will need further external validation. This work identified mutations not previously described to be associated with a strong impact with PI resistance, suggesting that TPV has a unique resistance profile. This mutation profile helps to explain its activity against viruses harbouring classical PI resistance mutations.
DISCUSSION
A TPV/r mutation score could be constructed based on clinical virologic data from a multi PI-experienced patient cohort. Among the amino acid positions identified in this score: 36 - 53 + 58 + 69 + 89
- Some of these mutations have been previously identified in the BI score (36, 58, 69) (Baxter et al. J Virol 2006).
- The 89M/V mutation has been previously described in dataset analysing phenotype/genotype correlation (Baxter et al. J Virol 2006).
- The F53L mutation has been previously identified by Virco as being associated with increased susceptibility to TPV (L Bacheler et al. Clinically Relevant Phenotypic Resistance and Cross Resistance to Tipranavir among Recent Routine Clinical Isolates. 4th European HIV Drug Resistance Workshop, Monte Carlo, Monaco. 29-31 March 2006).
- None of these mutations are considered as major IAS mutation, thus probably explaining the activity of TPV on viruses harbouring such mutations
- Although the previous BI score has been shown to be predictive of the VR in this dataset, it is not possible to determine a clinical cut-off using this score.
- It seems that the VR to TPV/r was lower in patients infected with non clade B viruses in univariate analysis, but not in multivariate analysis. This should be further investigated including more patients infected with non clade B viruses."
Background
The aim of this study was to identify mutations associated with virological response to tipranavir/ritonavir (TPV/r)-based regimen in protease inhibitor (PI)-experienced patients
Patients and Methods
Inclusion criteria:
- PI-experienced patients
- Baseline HIV-1 RNA >1000 copies/ml
- TPV/r as the sole PI during at least 3 months
- Detectable TPV plasma levels at M3
Virological methods:
- RT and protease resistance genotypic analysis were determined from plasma samples drawn at baseline TPV/r
- Reported mutations as listed at www.iasusa.org (update September 2006)
Statistical methods:
- Virological response (VR) was defined as at least 1 log10 of HIV-RNA decrease from baseline or HIV-RNA < 200 copies/ml or < 50 copies/mL at month 3.
- Association between each protease mutation (codons 1 to 99) and VR was studied using Fisher's exact test (Step 1 analysis). Mutations with prevalence >10% and p-value <0.10 were retained in the first step of the analysis . Mutations at positions 1-9, 18, 21, 22, 23, 25-31, 38-40, 42, 44, 45, 49-52, 56, 59, 65, 67, 68, 70, 75, 78-81, 83, 86-88, 91, ,92, 94-99 in the protease gene could not be evaluated because their prevalence was < 10%.
- Cochran-Armitage (CA) test was used to select the combination of mutations most strongly associated with VR, with a removing procedure technique.
- Multivariate analysis (logistic model) was performed to account for confounding variables
143 Patients were included in the analysis:
Baseline Characteristics (median): an average of 3 major PI mutations (0-7); 9 minor mutations; 85% male; 89% subtype B; HIV RNA: 4.7 log; CD4: 115; previous treatment: 11 ARTs, 6 NRTIs, 4 PIs, 24% NNRTIs, 28% Fuzeon; TPV/r co-treatment: 97% NRTIs, 15% NRTI+NNRTI, 55% NRTIs+Fuzeon.

RESULTS
Virological response to TPV/r-containing regimen:
- 55% of patients achieved a VR at M3
- HIV RNA reduction at M3 = -1.16 log copies/ml (- 3.82 to 1.16)
- HIV RNA reduction at M6 = -0.83 log copies/ml (-4.00 to 0.58)
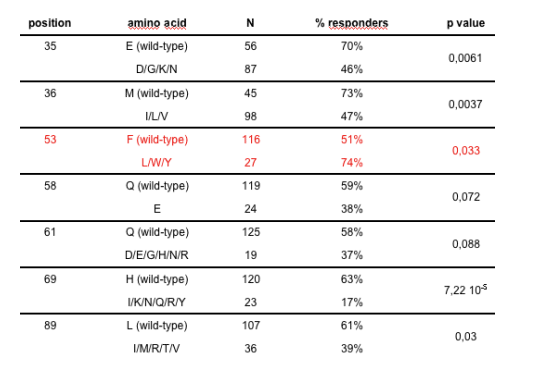
Mutations associated with the VR at M3
All listed mutations are associated with a worse VR, except for F53L/W/Y in red (better VR).
TPV genotypic score
The CA procedure did not retain mutations at codons 35 and 61 and led to select the following genotypic score: 36 - 53 + 58 + 69 + 89, as providing the strongest association with VR.
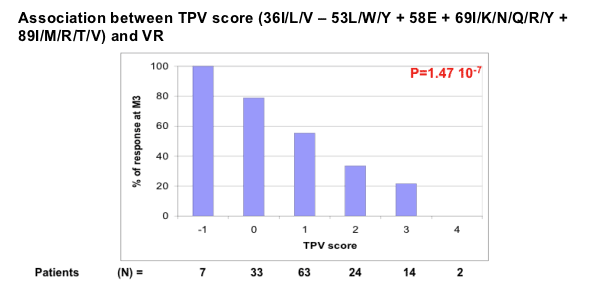
Multivariate analysis
Variables included in the logistic model were: T20 in past regimen, Clade B vs. Non Clade B, EFV in the background regimen, TPV score (score < or ≥ 2)
In the final model, were retained as independently associated to the VR:
- T20 past regimen OR=3.99 (p=0.0015) for all patients, as well as in patients that went on to receive T20 with TPV, to a poorer VR
- EFV background regimen OR=0.14 (p=0.035) to a better VR
- TPV score (score < or ≥ 2) OR=6.8 (p<0.001)
Evaluation of the BI score (www.hivfrenchresistance.org) and the new TPV score in the Pitie-Salpetriere database of treated patients experiencing virological failure (473 virological failure in 2006)
BI score:
- TPV Resistant or Possible resistant: >/= 4 mutations among 10V, 13V, 20M/R/V, 33F, 35G, 36I, 43T, 46L, 47V, 54A/M/V, 58E, 69K, 74P, 82L/T, 83D and 84V
- TPV sensitive: < 4 mutations among 10V, 13V, 20M/R/V, 33F, 35G, 36I, 43T, 46L, 47V, 54A/M/V, 58E, 69K, 74P, 82L/T, 83D and 84V
TPV score:
- TPV Resistant: at least a score of +3 among: 36I/L/V - 53L/W/Y + 58E + 69I/K/N/Q/R/Y + 89I/M/R/T/V
- TPV sensitive: a score of -1 to +2 among: 36I/L/V - 53L/W/Y + 58E + 69I/K/N/Q/R/Y + 89I/M/R/T/V
|
| |
|
 |
 |
|
|