 |
 |
 |
| |
Assessing HIV-1 Tropism in ACTG A5211: A Comparison of Assays Using Replication-competent Virus from Peripheral Blood Mononuclear Cells (PBMC) Versus Plasma-derived Pseudotyped Virions
|
| |
| |
Reported by Jules Levin
CROI, Feb 2007, Los Angeles
Noriaki Hosoya*1, Z Su2, T Wilkin3, R M. Gulick3, C Flexner4, M Hughes2, P R. Skolnik5, W L. Greaves6, E Coakley7, and D R. Kuritzkes1
1Brigham and Women's Hosp, Harvard Med Sch, Boston, MA, US; 2Harvard Sch of Publ Hlth, Boston, MA, US; 3Weill Med Coll of Cornell Univ, New York, NY, US; 4Johns Hopkins Univ, Baltimore, MD, US; 5Boston Med Ctr, MA, US; 6Schering Plough, Kenilworth, NJ, US; and 7Monogram Biosci, South San Francisco, CA, US
AUTHOR CONCLUSIONS:
Variable sensitivity for detecting CXCR4-using virus in subjects with a low proportion of D/M or X4 virus was noted for the Trofile and MT-2 cell assays.
Fluctuations in Trofile assay results near the threshold of detection of dual-tropic or X4 viruses are partly accounted by the low fraction of dual-tropic or X4 viruses in the population.
Detection of low-level D/M or SI virus at entry was clinically significant as judged by subsequent virologic failure and persistence of D/M (or X4) virus.
More sensitive assays for detection of minor populations of X4 variants in the HIV-1 quasi-species are needed.
ABSTRACT
Background: Switching from R5 to dual/mixed (D/M) virus is seen in ~10% of subjects screening for CCR5 inhibitor studies. To explore this change, we evaluated results from an MT2 cell assay and relative infectivity on U87-CD4-CXCR4 cells of D/M samples obtained at screening and/or entry using the Trofile assay (Monogram Biosciences) in ACTG A5211, a phase 2b trial of vicriviroc (VCV).
Methods: Plasma samples from subjects at screening and/or entry into A5211 were tested by Trofile. Luciferase activity measured as relative light units (RLU) on U87-CD4-CCR5 or U87-CD4-CXCR4 cells, or both, was used to classify pseudotyped viruses as R5, X4, or D/M, respectively. MT-2 cells were
inoculated with HIV-1 isolates and inspected for syncytium induction (SI); the same viruses were tested on U87-CD4-CCR5 and U87-CD4-CXCR4 cells.
Results: As reported (Wilkins et al, CROI 2006), 178/391 (46%) subjects had D/M virus at screening, and were excluded from A5211. Of 118 subjects with R5 virus who enrolled into A5211, 12 (10%) showed D/M virus at entry. The median RLU on CXCR4+ cells for these samples was significantly lower than that of samples with D/M virus at screening (295 vs 28,085; p<0.0001). To determine whether D/M virus could be detected in additional subjects by assay of infectious virus, MT-2 cell assays were performed using virus cultured from available cryopreserved PBMC obtained at baseline (n=106). Infectious HIV-1 was recovered from 55 cultures (52%). Virus from 5 subjects was SI and replicated on U87-CD4-CXCR4 cells. 2 of the 47 subjects (4%) who tested as R5 at entry had SI virus; both showed early emergence of X4 virus on study. In comparison 3 of the 8 subjects (38%) who tested as D/M at entry had SI virus. Of the 8 subjects who tested D/M by Trofile at entry, 7 had confirmed D/M tropism at later time points; 4 of these 7 were non-SI on MT-2 cells at entry. 5 subjects without X4 or SI virus at entry by either assay developed D/M virus on study. (Table.2)
Conclusions: Variable sensitivity for detecting CXCR4-using virus in subjects with a low proportion of D/M or X4 virus was noted for the Trofile and MT-2 cell assays. Fluctuations in Trofile assay results near the threshold of detection of dual-tropic or X4 viruses are partly accounted by the low fraction of dual-tropic or X4 viruses in the population.
BACKGROUND
ACTG A5211 is a randomized, placebo-controlled phase 2b trial of vicriviroc (VCV) in treatment-experienced subjects with R5 virus.
CCR5 inhibitor trials require screening for CCR5-using virus at entry.
192/391 (49%) subjects had R5 virus at screening [1]
12/118 enrolled subjects had D/M virus at entry [2]
Subjects with D/M virus at entry showed smaller reduction in HIV-1 RNA at week 24 on VCV:
R5 at entry (N=71): -1.33 log c/mL
D/M at entry (N=10): -0.77 log c/mL (p=0.01)
Commonly used assay (TrofileTM ; Monogram) detects D/M when X4 component >10%
About 10% of subjects with R5 virus at screening have D/M virus at entry.
OBJECTIVES
Compare relative light units (RLU) of X4 component of plasma virus population in samples with D/M virus at screening and entry.
Explore tropism of HIV-1 isolates at entry using MT-2 cell assay and infectivity on U87-CD4-CCR5 or U87-CD4-CXCR4 cells.
Compare results of infectious virus assays with Trofile assay (pseudovirions).
METHODS
Tropism of plasma virus at screening and entry measured by Trofile assay.
Pseudotyped viruses classified as R5, X4 or D/M based on luciferase production (RLU) on U87-CD4-CCR5 and U87-CD4-CXCR4 cells.
HIV-1 cultured from cryopreserved PBMC obtained at entry Syncytium induction (SI) assayed on MT-2 cells.
Infectivity on U87-CD4-CCR5 and U87-CD4-CXCR4 cells determined by p24 Ag production.
Chemokine receptor tropism assay (Trofile assay)
The HIV-1 envelope coding sequence from plasma viral RNA is amplified and cloned into an envelope expression vector. The envelope expression vector and an HIV genomic vector carrying a luciferase reporter gene are co-transfected into 293 cells. Pseudotyped HIV particles are harvested and used to infect U87 cells expressing CD4 and either CCR5 or CXCR4. Luciferase activity measured as relative light units (RLU) on U87-CD4-CCR5 or U87-CD4-CXCR4 cells, or both, is used to classify pseudotyped viruses as R5, X4, or dual or mixed coreceptor tropism for CCR5 and CXCR4, respectively.
HIV preparation from PBMC
Cryopreserved PBMC obtained at entry baseline were co-cultured with PHA-stimulated PBMC from seronegative donors to generate infectious stocks of HIV-1. Fresh PBMC for healthy donors, stimulated with PHA 2 days, in RPIM medium, supplement with 20% Fetal calf serum and IL-2, were added to the culture each week. Cultures were checked twice a week and the cells were resuspended in fresh culture medium. Virus production was detected with a HIV-1 p24 capture enzyme-linked immunosorbent assay. Infectious HIV-1 was recovered from 55 of 106 (52%) cultures as detected by p24 production.
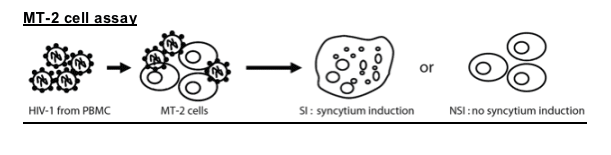
MT-2 cells were inoculated with HIV-1 isolates and inspected for syncytium induction (SI). Virus supernatant 50ml was added to 51000 MT-2 cells in duplicate wells of flat 96 well microtiter plates in final volume of 200ml/well. Cultures were every 3 days for synsytium formation by using inverted microscope until day14. If no syncytia were observed by the day of assay termination, the isolate was scored as NSI.
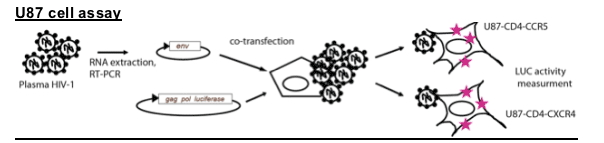
The same viruses were tested on U87-CD4-CCR5 and U87-CD4-CXCR4 cells. Virus supernatant 50ml was added to 51000 U87-CD4-CCR5 or U87-CD4-CXCR4 cells in 24 well plates in a final volume of 1ml/well. Cultures were every 3 days for virus production by p24 Ag ELISA.

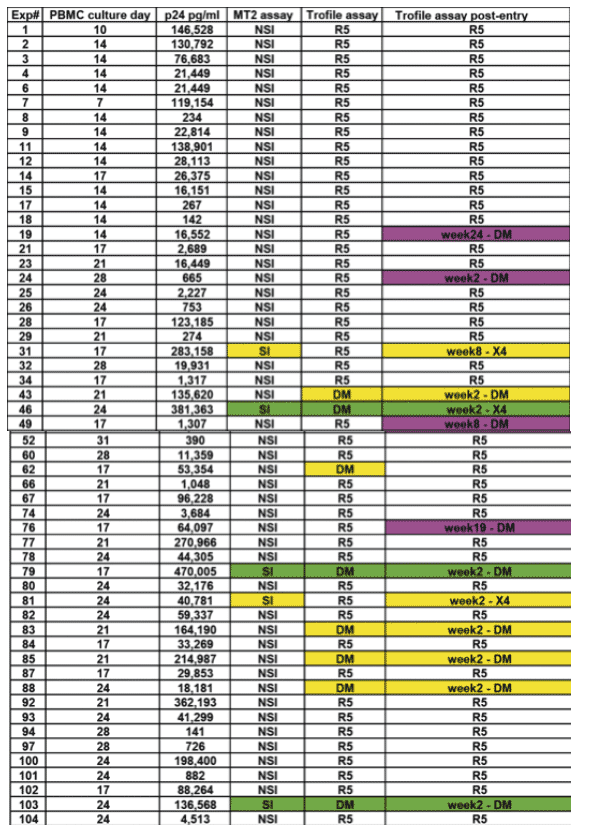
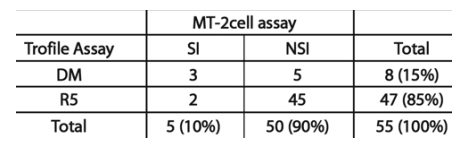
Both R5 subjects with SI virus at entry showed early emergence of D/M virus on VCV.
7 of 8 (88%) subjects with D/M virus at entry showed D/M or X4 virus at later time points.
5 subjects without either D/M or SI virus at entry showed emergence of D/M virus on VCV.
|
| |
|
 |
 |
|
|