 |
 |
 |
| |
Increased SVR Rate with 48 Wks' Treatment and Higher RBV Dose in HCV Genotype 2/3 Pts Without a Rapid Virologic Response (RVR) Treated with Peginterferon Alfa-2a (40KD) (PEGASYS) Plus RBV (COPEGUS)
|
| |
| |
Reported by Jules Levin
DDW May 20, 2007
B. Willems1; S. J. Hadziyannis2; T. R. Morgan3; M. Diago4; P. Marcellin5; D. E. Bernstein6; P. J. Pockros7; A. Lin8; M. L. Shiffman9; S. Zeuzem10
1. Hopital Saint-Luc-Centre Hospitalier de l'Universite de Montreal, Montreal, QC, Canada.
2. Department of Medicine and Hepatology, Henry Dunant Hospital, Athens, Greece.
3. Gastroenterology-11, VA Medical Center, Long Beach, CA, USA.
4. Hospital General de Valencia, Valencia, Spain.
5. Hopital Beaujon, Clichy, France.
6. North Shore University Hospital, Manhasset, NY, USA.
7. Division of Gastroenterology/Hepatology, Scripps Clinic, La Jolla, CA, USA.
8. Roche, Nutley, NJ, USA.
9. Virginia Commonwealth University Medical Center, Richmond, VA, USA.
10. Department of Internal Medicine, Saarland University Hospital, Homburg/Saar, Germany.
Background: ACCELERATE, a large-scale (n=1469) prospective study recently showed that, overall, peginterferon alfa-2a (40KD) plus ribavirin (RBV) for 24wks was superior to 16wks in hepatitis C virus (HCV) genotype 2/3 pts (EASL 2006:A734). In this study, two-thirds of pts achieved a rapid virologic response (RVR; HCV-RNA <50IU/mL at wk 4). These early-responder pts had a >80% probability of achieving an SVR with 16wks of therapy, suggesting that this shortened treatment regimen may be a reasonable customized option for RVR pts who cannot tolerate a full course of therapy. Conversely, pts without an RVR had an SVR rate of only 49% with 24wks' treatment, suggesting that these pts may benefit from more intensive treatment regimens. To determine whether an intensified regimen of peginterferon alfa-2a plus RBV may be beneficial in genotype 2/3 pts without an RVR, we retrospectively examined available data from other pivotal clinical studies.
Methods: SVR and relapse rates following peginterferon alfa-2a (PEGASYS) plus RBV (COPEGUS) were analyzed in HCV genotype 2/3 pts who did not achieve RVR in two studies (NV15942, NV15801). In NV15942 (Hadziyannis. Ann Int Med 2004), pts were randomized to 24 or 48wks of peginterferon alfa-2a 180µg/wk plus RBV 800 or 1000/1200mg/d. In NV15801 (Fried. NEJM 2002), pts received 48wks of peginterferon alfa-2a plus RBV 1000/1200mg/d.
Results: RVR was achieved by a high proportion of genotype 2/3 pts (75% and 59% in NV15942 and NV15801). In pts in NV15942 who did not achieve an RVR, the lowest relapse rate and the highest SVR rate (76%) was achieved with 48wks of peginterferon alfa-2a plus RBV 1000/1200mg/d (Table). This compared with 61% for 48wks' peginterferon alfa-2a plus RBV 1000/1200mg/d in NV15801 and 49% for 24wks' peginterferon alfa-2a plus RBV 800mg/d in ACCELERATE.
Conclusions: Genotype 2/3 pts who do not achieve an RVR could receive added benefit if treated for 48wks with a higher dose of RBV (1000/1200mg/d).These results merit further investigation in a prospective controlled study to fully elucidate how treatment customization can benefit pts.
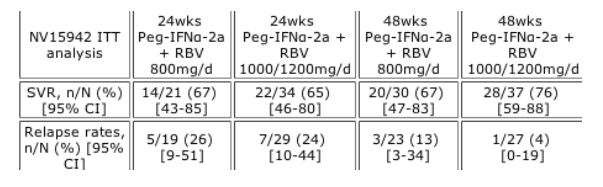
Pts without follow-up data were considered not to have achieved an SVR.
|
| |
|
 |
 |
|
|