 |
 |
 |
| |
Peginterferon Alfa-2a (40KD) Plus Ribavirin for 16 or 24 Wks in Pts With HCV Genotype 2/3 Infection and Advanced Fibrosis/Cirrhosis
|
| |
| |
Reported by Jules Levin
DDW, May 19-24, 2007, Washington DC
M. L. Shiffman1; F. Suter2; B. R. Bacon3; D. R. Nelson4; H. Harley5; R. Sola6;
S. D. Shafran7; K. Barange8; A. Lin9; G. Hooper10; S. Zeuzem11
1. Hepatology Section, Virginia Commonwealth University Medical Center, Richmond, VA, USA.
2. Divisione di Malattie Infettive, Ospedali Riuniti, Bergamo, Italy.
3. St Louis University, St Louis, MO, USA.
4. University of Florida, Gainsville, FL, USA.
5. Royal Adelaide Hospital, Adelaide, SA, Australia.
6. Liver Section, Minas Gerais Faderal University - School of Medicine, Barcelona, Spain.
7. University of Alberta Hospital, Edmonton, AB, Canada.
8. Hopital Purpan, Toulouse, France.
9. Roche, Nutley, NJ, USA.
10. Roche, Welwyn, United Kingdom.
11. Saarland University Hospital, Homburg/Saar, Germany.
Background: Chronic hepatitis C (CHC) pts with cirrhosis have a higher risk of developing severe complications and are in particular need of treatment. In the ACCELERATE trial of peginterferon alfa-2a plus ribavirin (RBV) in pts with HCV genotype 2 or 3 (G2/3) infection, cirrhosis was a significant negative prognostic factor of a sustained virologic response (SVR). We further analyzed the ACCELERATE database to compare the efficacy of treatment in pts with and without cirrhosis, and whether on-treatment virologic response would assist treatment optimization.
Methods: Pts (n=1469) received peginterferon alfa-2a (PEGASYS) 180mg/wk plus RBV (COPEGUS) 800mg/d for either 16 or 24wks. SVR (undetectable [<50IU/mL] HCV-RNA at 24wks post-treatment) was analyzed according the presence or absence of baseline cirrhosis in 1309 pts receiving treatment as defined by the protocol. Cirrhosis included advanced fibrosis/transition to cirrhosis (Knodell ≥3, Metavir ≥3, or Ishak modified HAI score of 4 [with nodules or >3 bridges] or ≥5). Additional analyses determined SVR rates in pts with and without rapid virologic response (RVR; undetectable HCV-RNA [<50IU/mL] at treatment wk 4).
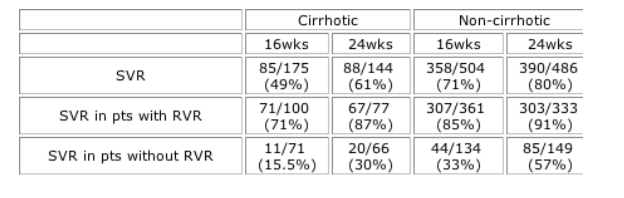
Results: Cirrhotic pts had lower SVR rates than non-cirrhotic pts; for both groups, SVR was higher with 24 vs. 16wks' treatment (Table). RVR was achieved by 56% of pts with cirrhosis and 71% of non-cirrhotic pts. Non-cirrhotic pts with RVR had SVR rates of 85% with only 16wks' treatment; extending treatment to 24wks resulted in an SVR rate of 91% (p=0.02; two-sided Fisher's exact test). In cirrhotic pts with RVR, 24wks' treatment resulted in higher SVR rates than with 16wks (87% vs. 71%; p=0.01). In pts without an RVR, both cirrhotic and non-cirrhotic, 24wks' treatment produced higher SVR rates than 16wks (p=0.04 and p<0.0001, respectively).
Conclusions: Higher SVR rates with peginterferon alfa-2a plus RBV therapy are achieved in G2/3 pts without cirrhosis, supporting early treatment before progression to advanced liver disease. In pts with RVR, 24wks' treatment resulted in SVR rates >87% in both cirrhotic and non-cirrhotic pts. Cirrhotic pts without RVR have a chance of SVR of only 30% with the currently recommended 24-wk regimen; in these pts, longer treatment durations might be beneficial. Assessing on-treatment virologic response at wk 4 thereby allows treatment optimization in both cirrhotic and non-cirrhotic pts with HCV G2/3 infection.
|
| |
|
 |
 |
|
|