 |
 |
 |
| |
Resistance Determination in Patients Experiencing Virologic Breakthrough Following Telbivudine or Lamivudine Therapy in the International GLOBE Trial
|
| |
| |
Reported by Jules Levin
DDW, May 22, 2007, Wash DC
D. N. Standring1; A. Patty1; C. Chapron1; L. van Doorn2; B. Belanger1; N. A. Brown1; M. Seifer1
1. Idenix Pharmaceuticals, Cambridge, MA, USA.
2. DDL Diagnostic Services, Voorburg, Netherlands.
Introduction: Resistance to telbivudine and lamivudine was evaluated at week 48 of therapy in an international trial (GLOBE), involving 1367 patients with chronic hepatitis B who were randomized (1:1) to receive 600 mg telbivudine or 100 mg lamivudine daily. Here we report the genotypic analysis of patient samples showing viral breakthrough as outlined by EASL/EMEA guidelines (≥ log10 above nadir rebound in HBV DNA after a drop in viral load of at least 1 log10 copies/mL on drug therapy).
Methods: Amplification and direct sequencing of the 344 codon HBV RT domain were performed at screen for the ITT population. Patients experiencing breakthrough were re-sequenced at week 48 or post-breakthrough to identify resistance mutations.
Results: 131 patients met the breakthrough definition; 32 were on telbivudine and 99 on lamivudine. All but 6 lamivudine samples were amplifiable. Genotypic resistance, all based on rtM204 variants, was confirmed in 28/32 telbivudine and 75/93 lamivudine patients; the remaining genomes were essentially wild-type, presumably reflecting patient non-compliance. Of the 28 telbivudine resistant patients, 27 exhibited the M204I mutation alone (10) or in combination with rtL80I/V substitutions (17); 1 showed a mixed codon 204 variant (M204M/I/V). Of the 75 lamivudine resistant patients, 34 were due to M204I (most with rtL80 substitutions), 25 to M204V/L180M, and 16 were mixed M204M/I/V mutants. No telbivudine-related M204V or M204/L180M mutants or novel resistance mutants were seen in association with breakthrough, without a detectable M204I mutation. The proportions of telbivudine recipients with breakthrough versus genotypically confirmed resistance (latter in parentheses) were 5.9/(5.0)% in HBeAg-positive and 2.25/(2.25)% in HBeAg-negative patients, versus 15.3/(11.2)% and 12.5/(10.3)%, respectively, for lamivudine. Analysis of year 2 data is ongoing, but to date M204I-mutant HBV strains continue to be the basis of genotypic resistance with virologic breakthrough for telbivudine.
In vitro phenotypic testing revealed that M204I and M204I/L80I variants were resistant to telbivudine (>1300 fold), but sensitivity to adefovir or tenofovir was only 3 to 5 fold reduced.
Conclusions: In the large GLOBE trial, genotypic HBV resistance with virologic breakthrough on telbivudine is based on the M204I mutation, confirming our earlier findings (Standring et al., EASL 2005). Based on our in vitro data, the best agents for therapy of telbivudine resistant M204I and M20I/L80I mutant HBV strains would appear to be adefovir or tenofovir.
POSTER S1781
AUTHOR DISCUSSIONS and CONCLUSIONS
The resistance profile of telbivudine is well characterized.
_ The primary pathway to breakthrough and resistance for telbivudine is the signature M204I mutation
The clinical resistance profile reflects predictions from in vitro models.
_ Telbivudine is active against the M204V single mutation
Close HBV monitoring for lack of early virologic response at 24 weeks may identify patients who are at greatest risk for developing resistance to telbivudine monotherapy.
Preliminary clinical data on 21 patients suggest that adefovir may be a treatment salvage option for pa-tients experiencing viral breakthrough during telbivudine treatment (see also data presented by J. Heath-cote et al at this meeting [abstract 1776]).
Background
Telbivudine, an L-nucleoside with potent and specific activity against the hepatitis B virus (HBV), is approved in many countries worldwide for the treatment of compensated chronic hepatitis B (CHB).1 A large, 2-year, international, randomized clinical trial (GLOBE) compared the safety and efficacy of telbivudine and lamivudine in 1367 nucleos(t)ide-naive patients with hepatitis B e antigen (HBeAg) seropositive or HBeAg-seronegative CHB. After 1 and 2 years of treatment, telbivudine showed significantly greater efficacy vs lamivudine on all direct measures of antiviral efficacy in both HBeAg-positive and HBeAg-negative patients, with significantly less resistance.2,3 A quantitative association between serum HBV DNA reduction in the first 6 months of treatment and subsequent efficacy outcomes was also established.4,5 Here we report the in vitro phenotypic resistance and cross-resistance profiles of telbivudine. We also report the genotypic analysis of all patients with viral breakthrough, as defined by a ≥1 log10 copies/mL above nadir rebound in HBV DNA after a drop in viral load of at least 1 log10 while on drug therapy.
METHODS
Phenotypic In Vitro Resistance
Phenotypic in vitro resistance testing was performed in stably expressing HBV cell lines of HepG2 origin bearing known resistance mutations to lamivudine or adefovir. In addition, cell lines bearing the signature telbivudine resistance mutation M204I, alone or in conjunction with L80I/V, were used to evaluate the cross-resistance profiles of telbivudine, lamivudine, tenofovir, adefovir, and entecavir.
Clinical Study Design
Eligible male and female patients were between 16 and 70 years old with serum HBV DNA >6 log10 copies/mL, serum alanine aminotransferase (ALT) 1.3-10 times the upper limit of normal (ULN), positive serum hepa-titis B surface antigen (HBsAg), and compensated liver disease. Patients were randomized (1:1) to treatment with telbivudine (600 mg/day PO) or lamivudine (100 mg/d PO) (Figure 1). Treatment assignments were stratified by baseline HBeAg status (positive or negative) and baseline ALT levels (≦ or ≥ 2.5 x ULN). The study was sponsored by Idenix Pharmaceuticals, Inc., and Novartis Pharmaceuticals, Inc.
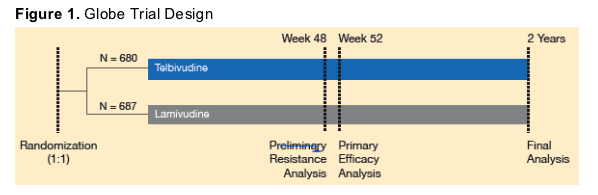
Resistance Determination in the GLOBE Trial
Viral breakthrough
_ Confirmed rebound of serum HBV DNA by ≥1 log10 copies/mL above nadir, after initial reduction of ≥1 log10.6
Resistance
_ Amplification and population sequencing were performed at screen for the full intent-to-treat (ITT) population (1367 patients). HBV genomes were amplified from sera of study patients by high fidelity polymerase chain reaction (PCR; or nested PCR for serum samples with low viral load). The 344_amino acid polymerase reverse transcriptase (RT) domain was sequenced as outlined in Figure 2 to detect all known resistance mutations, as well as any novel mutations. Blinded samples were sequenced by an independent, third party vendor (Delft Diagnostic Laboratories, Voorburg, The Netherlands).
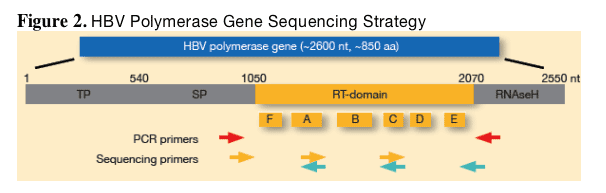
In patients experiencing a confirmed 1 log10 above nadir viral rebound, HBV polymerase genes were resequenced at week 48 or postbreakthrough to identify treatment-emergent resistance mutations via pair-wise comparisons. A preliminary resistance analysis (focused on codon 204 mutants) was done at week 92. Week 104 analysis is ongoing.
Primary treatment failure
-- Serum HBV DNA levels never below 5 log10 copies/mL after ≥24 weeks of treatment
RESULTS
In Vitro Drug Efficacy and Antiviral Activity Against Wild-Type Virus and Viruses Harboring Reported Resistance Mutations
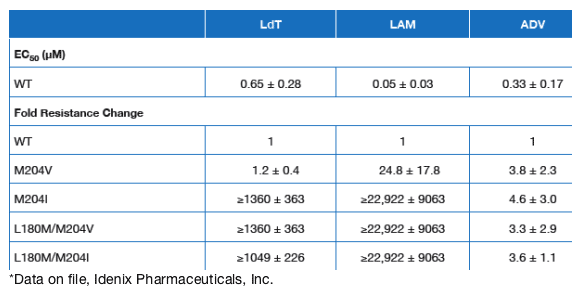
Telbivudine was active against viruses harboring the nucleoside resistance mutation M204V, whereas it was inactive (in vitro fold resistance >1000) against viruses in which the resistance mutations M204I, L180M/M204V, and L180M/M204I were present (Table 1).
Antiviral activity of lamivudine was reduced (M204V) or inactive (M204I, L180M/M204V and L180M/M204I) against viruses harboring the nucleoside resistance mutations (Table 1).
The nucleotide analog adefovir showed a mild decrease in antiviral activity against these mutations, but is known to be active against lamivudine-resistant mutants in the clinic (Table 1).7
Table 1. In Vitro Drug Efficacy and Fold Resistance Against Wild Type HBV and Virus Harboring Lamivudine Resistance Mutations*
Telbivudine was active or showed mildly reduced antiviral activity against viruses harboring the adefovir resistance mutations N236T or A181V (Table 2). Given the low level of decrease in susceptibility (Table 1), telbivudine is expected to be active against adefovir-resistant viruses. However, this needs to be clinically validated.
Conversely, although adefovir showed mildly reduced susceptibility (<5-fold) to N236T and A181V, both of these mutations are believed to lead to clinical resistance to adefovir (Table 2).8,9

The in vitro activity of various mucleosides and nucleotides was also tested (Table 3) against the M204I and M204I + L80I mutants selected by telbivudine in the clinic.
The fold resistance data suggest that nucleosides and nucleotides show different cross-resistance profiles (Table 3).
These data suggest that telbivudine-resistant HBV may be salvaged by nucleotide analogs tenofovir or adefovir.10

Resistance in the GLOBE Trial
At week 48, 131 HBeAg-positive and HBeAg-negative patients had viral
breakthrough (as defined above).
-- Telbivudine arm: n = 32
-- Lamivudine arm: n = 99
Sequencing analysis
-- Paired sequences obtained for all 32 telbivudine-treated patients:
31 at week 48
1 at week 16
-- Paired sequences obtained for 93 of 99 lamivudine-treated patients:
89 at week 48
4 at week 40
6 missing lamivudine sequences assumed to be wild type
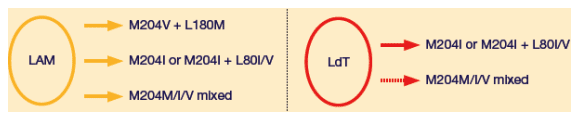
Telbivudine had a simplified pattern of resistance based primarily on the M204I mutant, as compared with three patterns of resistance for lamivudine (Figure 3).
Secondary mutations at codon 80 (L80I or L80V), or occasionally L80I/V + L180M, have been found in conjunction with M204I.
Variants at codons A181 and L229 were detected infrequently, but were not associated with viral break-through unless an M204I mutation was also present.
No novel or unique telbivudine signature resistance mutation has been found to date.
Figure 3. Summary of Clinical Resistance Profiles Observed in the GLOBE Trial With Lamivudine and Telbivudine
The absence of telbivudine resistance based on M204V variants is believed to reflect the activity of telbivudine against the M204V single mutant (Table 4).
During the development of clinical resistance to lamivudine, M204V precedes the emergence of the more robust M204V + L180M double mutant.

This analysis identified M204I as the signature telbivudine resistance mutation.
No M204V mutation was associated with viral breakthrough after 1 year of treatment with telbivudine.
Mixed codon 204 mutants were uncommon with telbivudine.
Significantly fewer M204 resistance mutations were associated with viral breakthrough after 1 year of treatment with telbivudine as compared with lamivudine.
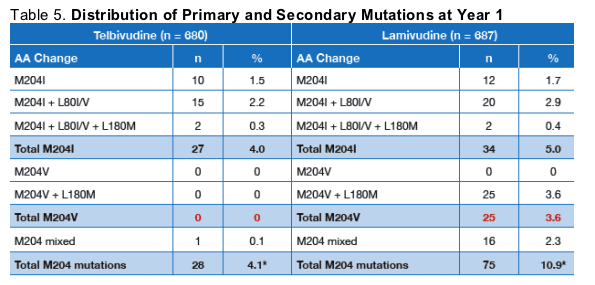
The lamivudine resistance mutation M204V is always accompanied by L180M.
In contrast, the signature telbivudine resistance mutation M204I is often accompanied by a secondary change at position L80 (usually L80I), which is thought to be a compensatory mutation.
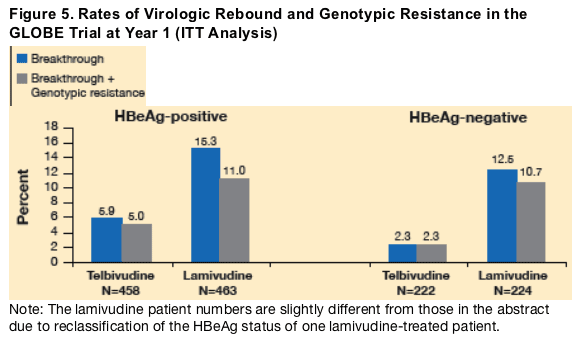
Note: The lamivudine patient numbers are slightly different from those in the abstract due to reclassification of the HBeAg status of one lamivudine-treated patient.
Resistance Rates According to Serum HBV DNA Level at Week 24 of Treatment
There was a low rate of resistance in patients who had undetectable DNA at week 24 (Figure 6).
In the same group of patients, the probabilities of achieving efficacy outcomes at 2 years were high for telbivudine recipients with PCR-nondetectable HBV DNA at week 24.11,12
A low level of HBV replication in the liver and/or low compliance may be responsible for the resistance rate observed in patients with undetectable serum HBV DNA.
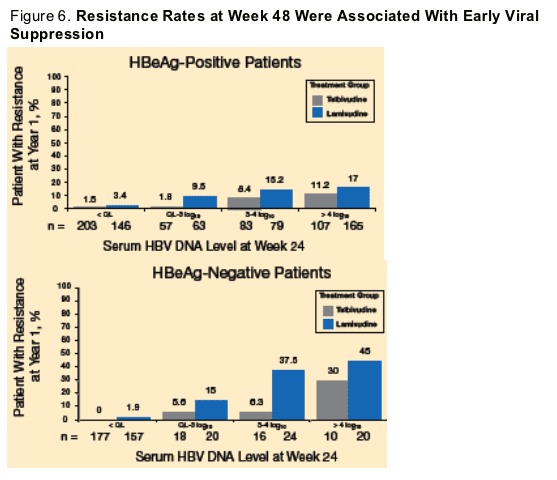
A preliminary resistance analysis was conducted at Week 92.
The resistance profile observed at week 92 (Table 6) was similar to that seen at Year 1.
No M204V-based telbivudine resistance was identified.
No novel telbivudine resistance mutation was seen.
A complete resistance analysis at week 104 is ongoing.

|
| |
|
 |
 |
|
|