 |
 |
 |
| |
Salvage Therapy With Adefovir for Virologic Breakthrough in Telbivudine-Treated Patients From the GLOBE Study
|
| |
| |
Reported by Jules Levin
DDW, May 22, 2007, Wash DC
J. Heathcote1; E. Gane2; C.L. Lai3; A. Min4; T. Poynard5; O. Ovunc Kurdas6; J.D. Grange7; N.A. Brown8
1Toronto Western Hospital, Toronto, ON, Canada; 2Middlemore Hospital, Auckland, NZ; 3University of Hong Kong, Hong Kong, China; 4Beth Israel Medical Center, New York, NY, USA; 5Groupe Hospitalier Pitie-Salpetriere, Paris, France; 6Haydarpasa Numune Hospital, Istanbul, Turkey; 7Hpital Tenon, Paris, France; 8Idenix Pharmaceuticals, Cambridge, MA, USA
In this nonrandomized, observational, post-hoc analysis of a small patient cohort failures on telbivudine in the Globe Study received a reduction in HBV DNA -4.3 log copies/ml by adding adefovir and -3.7 log copies/ml by switching to adefovir.
AUTHOR CONCLUSIONS
These data suggest that adefovir may be an effective salvage option for patients experiencing viral breakthrough during telbivudine treatment.
Adefovir and the adefovir + telbivudine combination were well tolerated during the period of this analysis.
Background
Telbivudine (_-L-2_-deoxythymidine) is an L-nucleoside.
The GLOBE study compared the efficacy of telbivudine vs lamivudine in patients with hepatitis B e antigen (HBeAg)-positive or -negative chronic hepatitis B (CHB).
M204I mutation is associated with telbivudine resistance; hepatitis B virus (HBV) with the M204I substitution remains susceptible to adefovir dipivoxil in vitro.
Adefovir restores adequate viral suppression in patients with lamivudine resistance.
We present here a preliminary analysis of an ongoing study.
The aim of this analysis was to evaluate the effectiveness and safety of adefovir as "salvage" therapy for telbivudine resistance in the GLOBE study.
METHODS
GLOBE Study Design
Phase III, randomized, controlled double-blind clinical trial-1367 enrolled patients, 680 randomized to telbivudine.
Telbivudine 600 mg/day PO vs lamivudine 100 mg/day PO.
Patients: HBeAg-positive or HBeAg-negative HBV, with:
- Baseline HBV DNA ≥106 copies/mL
- Baseline alanine aminotransferase (ALT) 1.3-10 x upper limit of normal (ULN)
- Compensated liver disease
After 2 years of treatment in GLOBE, eligible patients received continued telbivudine therapy in the 022 follow-on study.
Aim of Adefovir Salvage Therapy
Restoration of HBV DNA suppression following viral breakthrough on telbivudine
Normalization of ALT
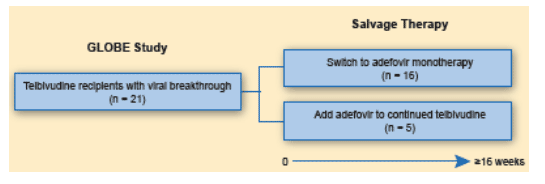
Patients for Adefovir Salvage Therapy
-- 32 patients randomized to telbivudine had viral breakthrough at 1 year
-- 21 patients with viral breakthrough on telbivudine received ≥16 weeks of adefovir at the discretion of the investigator
- 16 patients switched to adefovir monotherapy
- 5 added adefovir to continued telbivudine
Evaluation
Serum HBV markers:
- Serum HBV DNA assessed by Roche COBAS Amplicor polymerase chain reaction (PCR) assay (lower limit of detection [LLOD] ≦300 copies/mL)
- Serum ALT
- Clinical and laboratory safety parameters
Characteristics of 21 Adefovir Salvage Recipients
At baseline, 15 patients were HBeAg positive and 6 patients were HBeA negative.
Breakthrough on telbivudine occurred at a median of 60 weeks (range, 36-111 weeks).
The M204I mutation was detected in all 21 patients.
The A181T adefovir resistance mutation was not detected prior to adefovir treatment.
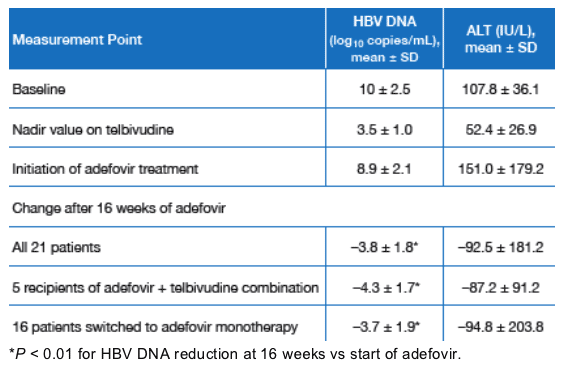
Table 1 summarizes serum HBV DNA and ALT levels in these patients.
Table 1. Serum HBV DNA and ALT Levels
After 16 weeks of adefovir mean HBV DNA decline was -3.8 log copies/ml in all 21 patients. For the 5 recipients of adefovir+telbivudine combination HBV DNA decline was mean -4.3 log copies/ml. For the 16 patients who switched to adefovir monotherapt mean decline in HBV DNA was -3.7 log c/ml.
Serum HBV DNA Response
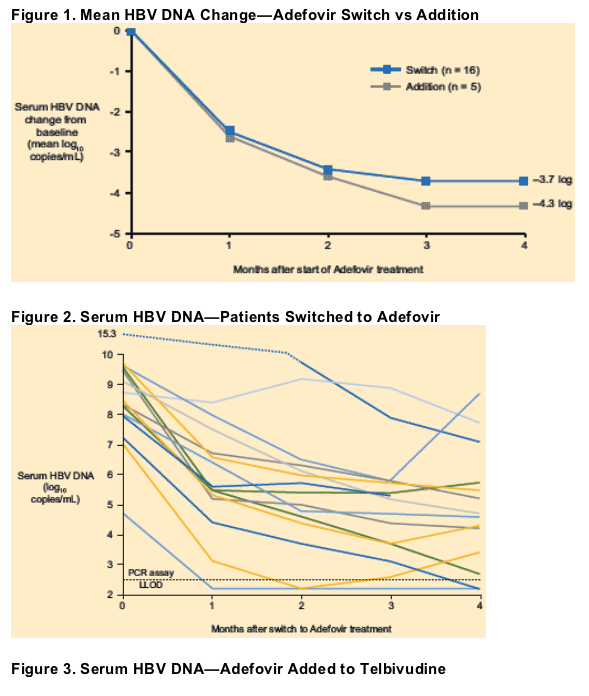
The mean decrease in serum HBV DNA at 16 weeks was 0.6 log10 greater in patients who added adefovir to continued telbivudine than in patients who switched to adefovir monotherapy (Figure 1).
In the switch group, serum HBV DNA at 16 weeks was <5 log10 copies/mL in 8/16 patients and <3 log10 copies/mL in 4/16 patients. There was evidence of possible loss of adefovir response in at least 1 patient (Figure 2).
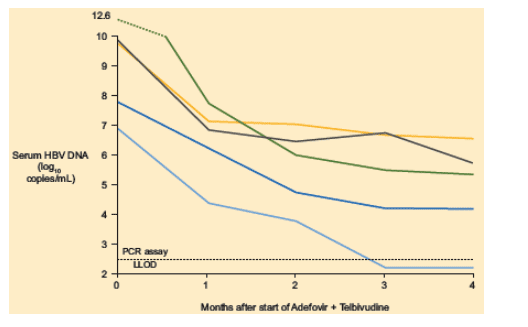
In the combination group (n = 5), HBV DNA was reduced by at least 3 log10 copies/mL at 16 weeks in all patients (Figure 3).
Biochemical Response
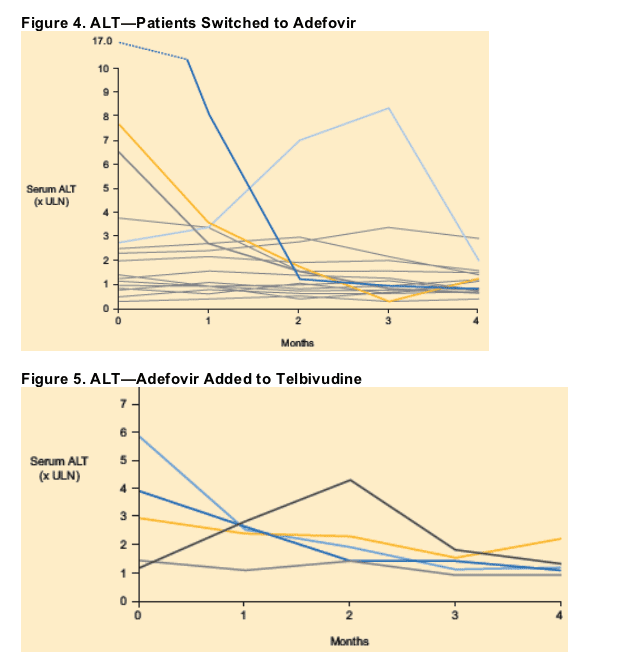
ALT elevations generally improved rapidly after initiation of adefovir treatment in both groups (Figures 4 and 5). One patient in each group experienced a moderate ALT increase during adefovir treatment; these increases resolved over the next 4-8 weeks.
Serologic Response
Two HBeAg-positive patients (one in the adefovir monotherapy group and one in the combination group) lost HBeAg during adefovir treatment and subsequently became HBV DNA PCR negative.
Safety
No serious adverse events were reported during this 16-week analysis.
Limitations
This was a nonrandomized, observational, post-hoc analysis of a small patient cohort; not powered to compare monotherapy vs combination.
More extended follow-up is needed to assess response durability.
|
| |
|
 |
 |
|
|