 |
 |
 |
| |
Treatment Duration and Histological Response to Peginterferon Alfa-2a/Ribavirin: 48 weeks improves liver disease in Non-SVRs better than 24 weeks
|
| |
| |
Reported by Jules Levin
Presented at the 2007 Digestive Disease Week (DDW) Meeting,
May 19-24, 2007; Washington, DC.
This study was supported by Roche Laboratories.
Paul J. Pockros, MD1; Paul Martin, MD2; Ellen Lentz, PhD3; Fayez M. Hamzeh, MD, PhD3; and Anna Lok, MD, FRCP4
1Scripps Clinic, La Jolla, CA, USA; 2Mount Sinai School of Medicine, New York, NY, USA; 3Roche Laboratories, Nutley, NJ, USA; M1854 4University of Michigan Health System, Ann Arbor, MI, USA
"....Improvement in activity and/or fibrosis score was observed in 93 (86%) of 108 patients who achieved SVR, and in 45 (44%) of 103 patients who did not (P<.001).... Of those not achieving an SVR, 50% receiving 48 weeks vs 29% receiving 24 weeks therapy achieved 1 or more grade improvements in fibrosis or inflammation (staging and grading) (p=.049)...."
Introduction
Current treatment practice in HCV, aimed at achieving sustained virologic response (SVR), is to discontinue treatment in patients with detectable serum HCV RNA at 24 weeks.
Improvement in inflammation has been reported in patients with HCV who achieved SVR after receiving interferon with or without ribavirin, and in a smaller percentage of patients who failed to achieve SVR, suggesting that interferon has a direct effect on liver histology.1-4
Longer treatment duration may enhance histologic improvement regardless of SVR.
Study Objective
To determine if treatment of HCV genotype 1-infected patients with peginterferon alfa-2a plus ribavirin for 48 weeks is associated with a greater degree of histologic improvement than treatment for 24 weeks.
Author Conclusions
Among patients chronically infected with HCV genotype 1, those who achieved SVR after treatment with peginterferon alfa-2a plus ribavirin were more likely to have histologic improvement than those who did not.
Among patients who achieved SVR, the proportion with histologic improvement was similar in groups treated for 24 or 48 weeks, regardless of ribavirin dose.
Among patients who did not achieve SVR, 9 of 72 (13%) treated for 48 weeks but none of 31 treated for 24 weeks (P=.054) had improvement in fibrosis stage. The percentage of patients with either inflammation or fibrosis improvement was significantly (P=.044) better for those who were treated for 48 than for 24 weeks, after controlling for ribavirin dose.
These findings suggest that longer treatment duration may be necessary for fibrosis improvement in patients without SVR.
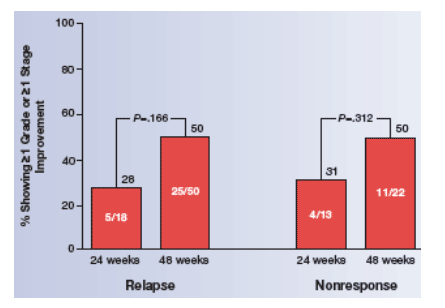
Fifty of 72 (69%) and 18 of 31 (58%) patients without SVR were relapsers in the 48- and 24-week treatment arms, respectively. Although the numbers were small,
the trend favoring 48 weeks in histologic outcome was similar in relapsers and nonresponders.
The numbers in this retrospective analysis were small. The hypothesis generated from this study, that a longer duration of treatment may be beneficial for histologic outcome, should be tested in a larger clinical trial.
Patients
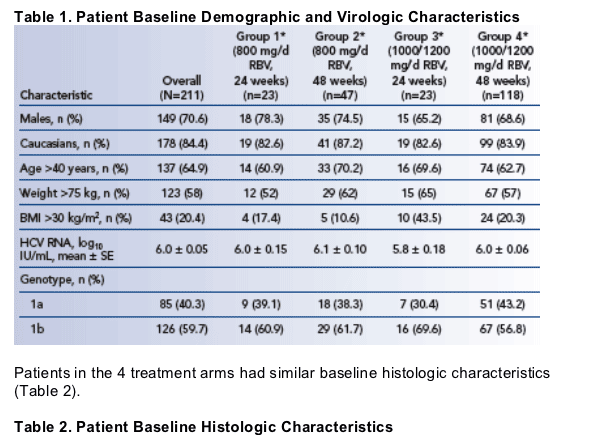
All 211 patients were HCV genotype 1-infected with paired biopsies before and 24 weeks after the end of treatment from 2 registration trials of peginterferon alfa-2a plus ribavirin.5,6
- Paired biopsies were performed from selected investigational sites. The protocols stipulated that patients in the 48-week treatment arm who had positive HCV RNA at Week 24 be withdrawn from treatment. Patients treated for 48 weeks with paired liver biopsies were more likely virologic responders at Week 24. Comparisons between Week 24 and Week 48 treatments were done for SVR, relapsers, and nonresponders separately.
Patients treated with 180 _g/wk peginterferon alfa-2a plus 800 mg/d ribavirin for 24 weeks (Group 1) or 48 weeks (Group 2), or with 180 _g/wk peginterferon alfa-2a plus 1000 or 1200 mg/d ribavirin for 24 weeks (Group 3) or 48 weeks (Group 4).
Relapsers were defined as patients who had at least 1 undetectable HCV RNA measurement during treatment but did not achieve SVR.
Methods
Retrospective histologic analysis
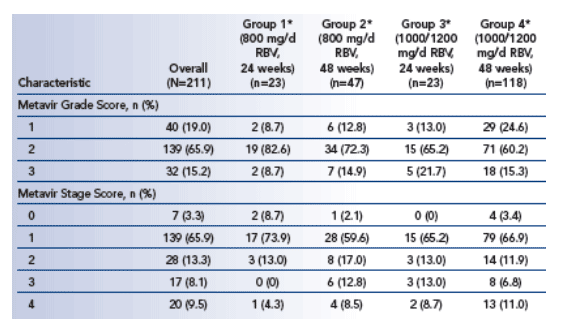
Metavir score used to stage fibrosis (0-4) and to grade activity (0-3). All liver biopsies were evaluated by 1 independent pathologist who was blinded to treatment group, time of biopsy, and patient ID.
Improvement defined as decrease of ≥1 stage or ≥1 grade and worsening as increase of ≥1 stage or ≥1 grade.
Statistical evaluations were performed using the Cochran-Mantel-Haenszel Test stratified by ribavirin dose (800 or 1000/1200 mg/d), Fisher's Exact test, and a general chi-square test.
RESULTS
Patient baseline demographic and virologic characteristics by treatment arm are shown in Table 1.
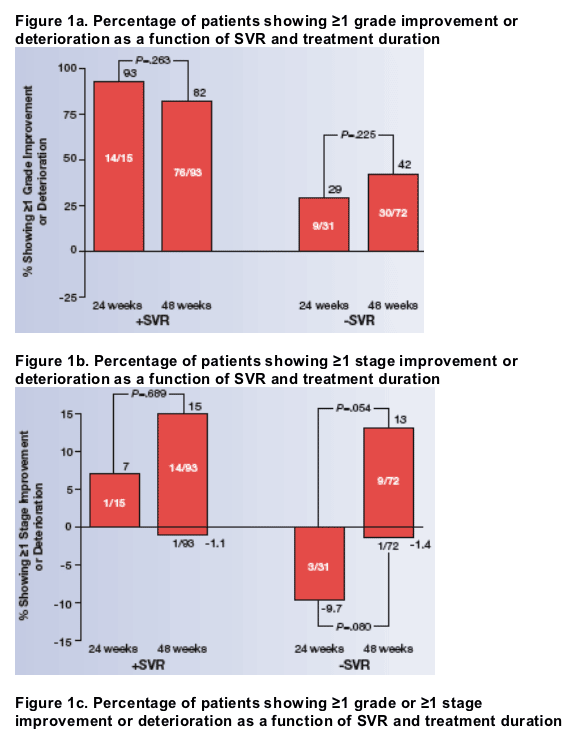
Improvement in activity and/or fibrosis score was observed in 93 (86%) of 108 patients who achieved SVR, and in 45 (44%) of 103 patients who did not (P<.001). Of those not achieving an SVR, 50% receiving 48 weeks vs 29% receiving 24 weeks therapy achieved 1 or more grade improvements in fibrosis or inflammation (staging and grading).
The relationships among histologic improvement, SVR, and treatment duration are shown in Figure 1. Among patients who did not achieve SVR, a higher percentage of those treated for 48 weeks had improvement in activity or fibrosis than those treated for 24 weeks (P=.049), as determined by a general chi-square test.
None of the 15 patients treated for 24 weeks who achieved SVR had worsening of fibrosis. Only 1 of the 93 patients (1.1%) treated for 48 weeks who achieved SVR had ≥1 stage worsening of fibrosis. Of patients who did not achieve SVR, 3/31 treated for 24 weeks (9.7%) and 1/72 treated for 48 weeks (1.4%) had ≥1 stage worsening of fibrosis.
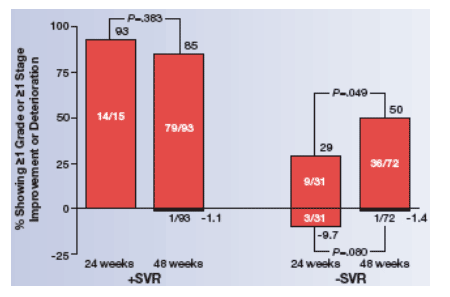
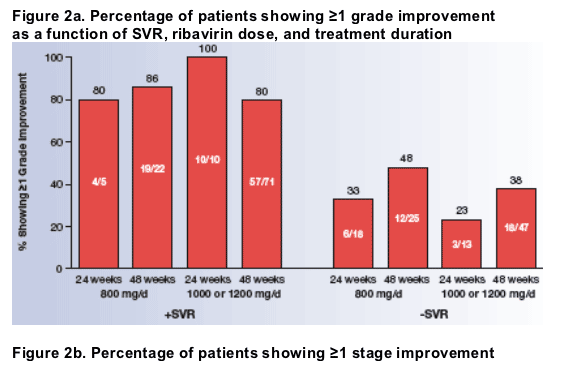
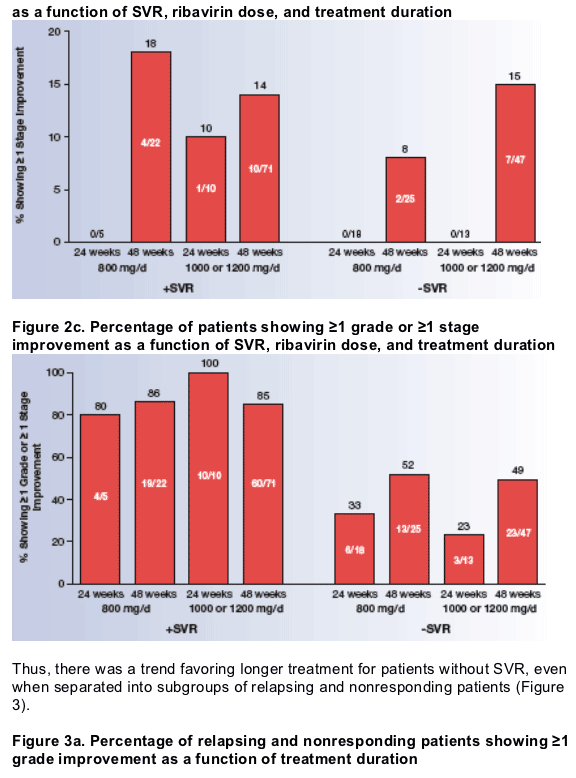
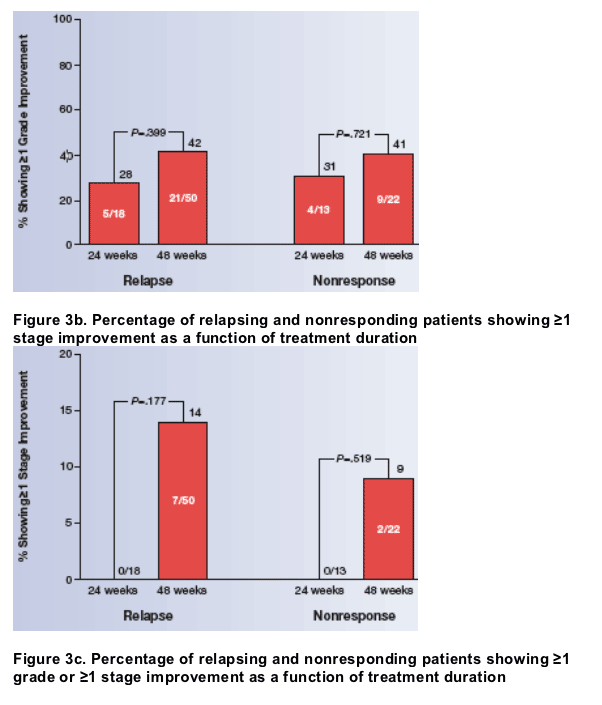
The relationships among histologic improvement and SVR, ribavirin dose, and treatment duration are shown in Figure 2. Among patients who did not achieve SVR, a significantly higher percentage of those treated for 48 weeks had ≥1 grade improvement in activity or ≥1 stage improvement in fibrosis than those treated for 24 weeks (P=.044), as determined by the Cochran-Mantel-Haenszel test stratified by ribavirin dose.
- When improvements in activity and fibrosis were determined separately, a higher percentage of those treated for 48 weeks showed ≥1 grade (P=.167) and ≥1 stage (P=.058) improvements.
REFERENCES
1. Jeffers LJ, Cassidy W, Howell CD, Hu S, Reddy KR. Peginterferon alfa-2a (40 kd) and ribavirin for black American patients with chronic HCV genotype 1. Hepatology. 2004;39:1702-1708.
2. Farrell GC. Two years versus six months of interferon therapy for chronic hepatitis C. Dig Dis Sci. 1996;41:93S-98S.
3. Toyoda H, Kumada T, Nakano S, et al. Effect of the dose and duration of interferon-alpha therapy on the incidence of hepatocellular carcinoma in noncirrhotic patients with a nonsustained response to interferon for chronic hepatitis C. Oncology. 2001;61:134-142.
4. Saito Y, Saito H, Tada S, et al. Effect of long-term interferon therapy for refractory chronic hepatitis c: preventive effect on hepatocarcinogenesis. Hepatogastroenterology. 2005;52:1491-1496.
5. Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347:975-982.
6. Hadziyannis SJ, Sette H Jr, Morgan TR, et al, for the PEGASYS International Study Group. Peginterferon-_2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med. 2004;140:346-355.
|
| |
|
 |
 |
|
|