 |
 |
 |
| |
A Phase III Comparative Trial of Telbivudine vs Lamivudine in Chinese Patients With Chronic Hepatitis B: Two-Year Results
|
| |
| |
Reported by Jules Levin
DDW, May 19-24, 2007, Wash DC
Y. Wang1; J.D. Jia2; J. Hou3; Y.K. Yin4; D. Xu5; D. Tan6; J. Niu7; X. Zhou8; L. Zhu9; N. Brown10
1Southwest Hospital, Chongqing, China; 2Beijing Friendship Hospital, Beijing, China; 3Nanfang Hospital, Guangzhou, China; 4Shanghai Hua Shan Hospital, Shanghai, China; 5Beijing Ditan Hospital, Beijing, China; 6Xiang Ya Hospital, Changsha, China; 7No. 1 Hospital, Changchun, China;
8Ruijin Hospital, Shanghai, China; 9Tianjin Infectious Diseases Hospital, Tianjin, China; 10Idenix Pharmaceuticals, Cambridge, MA, USA
Efficacy at 2 Years
Telbivudine demonstrated significantly greater responses vs lamivudine for key efficacy endpoints:
-- Log10 HBV DNA reduction and PCR nondetectability
-- ALT normalization
-- HBeAg loss in HBeAg-positive patients
-- Therapeutic Response
Jules' notes: HBV DNA decrease was -5.48 log for telbivudine at 2 yrs, -4.00 for LAM. PCR undetectable (<300 c/ml): 63% telbivudine, 39% LAM. ALT normalization: 76% telbivudine, 61% LAM. HBeAg loss: 40% telbivudine, 28% LAM. These differences were statistically significant (p <0.05). HBeAg seroconversion: 29% telbivudine, 20% LAM. 44% of patients with <300 c/ml HBV DNA at week 24 achieved HBeAg seroconversion. Grade 3 or 4 CK elevations, not requiring treatment modification, were more common with telbivudine than with lamivudine (11.4% vs 3.6%).
There was significantly less primary treatment failure in patients treated with telbivudine compared with lamivudine.
Response rates were generally higher in the subset of HBeAg-positive patients considered the best candidates for
treatment by association guidelines (baseline ALT ≥2 x ULN), compared with the overall population.
Both treatments were generally well tolerated. Most frequently reported adverse events included nasopharyngitis,
fatigue, and upper respiratory tract infection.
Impact of Early Viral Suppression on Treatment Outcomes
Rates of efficacy outcomes after 2 years of therapy were highest in patients with the greatest degree of early viral suppression (PCR negative at week 24).
-- HBeAg seroconversion
-- HBV DNA PCR-nondetectability
-- ALT normalization
The same relationship was observed with telbivudine and lamivudine, but greater viral reduction at week 24 was observed with telbivudine, consistent with the generally higher rates of efficacy responses associated with telbivudine at 2 years.
A resistance analysis of viral breakthrough samples up to week 104 is currently being conducted
Background
In a large, international Phase III trial (GLOBE), telbivudine showed significantly greater efficacy vs lamivudine on all direct measures of antiviral efficacy and on several key clinical measures, with significantly less resistance compared with lamivudine through year 2 in adults with chronic hepatitis B (CHB).1-3 CHB remains a significant healthcare concern in many Asian countries, particularly in China. Therefore, the present study was designed to confirm the findings of the GLOBE trial in a large population of Chinese patients with CHB. Results of a preliminary analysis of a locked database at 2 years from the intent-to-treat (ITT) population of this Phase III study are presented here. Maximizing and maintaining hepatitis B virus (HBV) suppression is emerging as a key milestone correlating with clinical improvement in CHB. Better early viral suppression has been linked to improved therapeutic outcomes in several reports. This report therefore includes an analysis of quantitative relationships between early viral suppression (serum HBV DNA levels after 24 weeks of treatment) and subsequent Therapeutic Response.
METHODS
Study Design
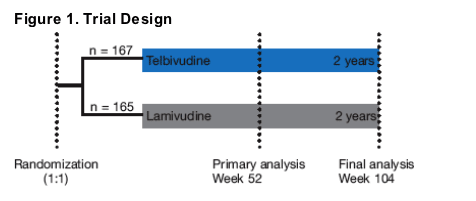
This randomized, double-blind trial compared 2 years of treatment with telbivudine 600 mg/day with lamivudine 100 mg/day in Chinese adults with hepatitis B e antigen (HBeAg)-positive or HBeAg-negative CHB (Figure 1). Key
entry criteria included baseline HBV DNA >6 log10 copies/mL, alanine aminotransferase (ALT) between 1.3 and 10 times the upper limit of normal (ULN), and compensated liver disease.
Key Efficacy Measures and Endpoints
Primary efficacy endpoint:
--Serum HBV DNA reduction from baseline by Roche COBAS® Amplicor assay
Key secondary endpoints:
-- Clearance of HBV DNA to nondetectability by polymerase chain reaction (PCR) assay
-- ALT normalization
-- HBeAg loss and seroconversion
-- Therapeutic Response: HBV DNA <5 log10 AND ALT normalization or HBeAg loss
Treatment failure:
-- Primary treatment failure: serum HBV DNA never <5 log10
Efficacy outcomes at 2 years were assessed according to serum HBV DNA levels at week 24.
-- PCR-nondetectable HBV DNA
-- Serum ALT normalization
-- HBeAg seroconversion
RESULTS
The study enrolled an ITT population of 332 patients in 18 centers in China (290 HBeAg positive, 42 HBeAg negative). Baseline demographic and disease characteristics of the ITT population are shown in Table 1. Treatment groups were well matched at baseline for demographic and disease parameters.*
EFFICACY
At 2 years, telbivudine was superior (P < 0.05) to lamivudine for the primary efficacy endpoint of HBV DNA suppression, with mean log10 HBV DNA reductions of -5.48 vs -4.00 (Table 2). Telbivudine also provided significantly
greater response rates for serum HBV DNA nondetectability by PCR assay, ALT normalization, Therapeutic Response, and HBeAg loss (for HBeAg-positive patients), compared with lamivudine (all P < 0.05) (Table 2).
Significantly fewer patients receiving telbivudine exhibited primary treatment failure, defined as serum HBV DNA levels that remained above 5 log10 copies/mL (3% vs 15% for lamivudine, P < 0.001).
Table 2. Efficacy Outcomes at 2 Years - All Patients
63% PCR-negative for telbivudine, 39% for lamivudine (p <0.05). ALT normalization: 76% telbivudine, 61% lamivudine (p <0.05). HBeAg loss: 40% telbivudine, 28% lamivudine (p <0.05). HBeAg seroconversion: 29% telbivudine, 20% lamivudine.

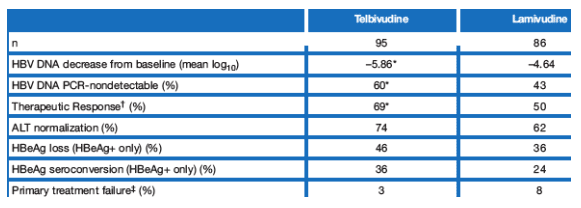
In HBeAg-positive patients with baseline ALT ≥2 x ULN, the population recommended for treatment in European Association for the Study of the Liver (EASL) and Asia-Pacific Association for the Study of the Liver (APASL) guidelines,4,5 telbivudine demonstrated a significantly greater reduction of serum HBV DNA from baseline compared with lamivudine. Telbivudine also provided significantly greater rates of Therapeutic Response and PCR-nondetectable HBV DNA compared with lamivudine (Table 3).
Table 3. Efficacy Outcomes at 2 Years in HBeAg-Positive Patients With Baseline ALT ≥2 x ULN
HBV DNA decrease: -5.86 log for telbivudine, -4.64 for LAM. HBV DNA undetectable: 60% telb, 43% LAM; these 2 parameters were statistically significant (p <0.05). ALT normalization: 74% telb, 62% LAM. HBeAg loss: 46% telb, 36% LAM. HBeAg seroconversion: 36% telbivudine, 24% LAM.
Relationship of Early Virologic Response to Outcomes at 2 Years
To assess the potential relationship between early antiviral response and outcomes at 2 years, patients were categorized into four groups according to serum HBV DNA levels at week 24:
-- PCR nondetectable (<300 copies/mL)
-- Quantitation limit to 103 copies/mL
-- 103 to 104 copies/mL
-- 104 copies/mL
HBV DNA was below the limit of detection at week 24 in a greater proportion of patients (combined HBeAg-positive and HBeAg-negative subpopulations) receiving telbivudine, compared with lamivudine (56% vs 30%) (Figure 2).
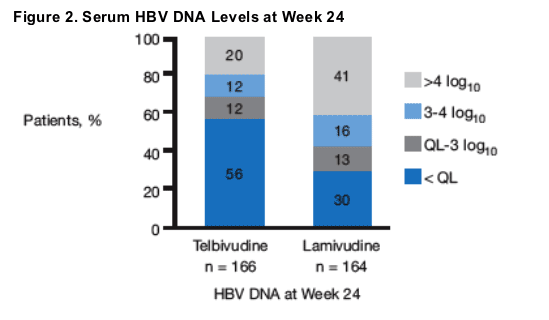
P<0.001 for HBV DNA PCR-nondetectable, nondetectable at week 24, telbivudine vs lamivudine.
QL = quantitation limit of 300 copies/mL by Roche COBAS® Amplicor assay.
Serum HBV DNA levels at week 24 were associated with treatment outcomes at 2 years. Telbivudine-treated patients with minimal or PCR-nondetectable serum HBV DNA at week 24 had a high rate of HBV DNA PCR-nondetectability
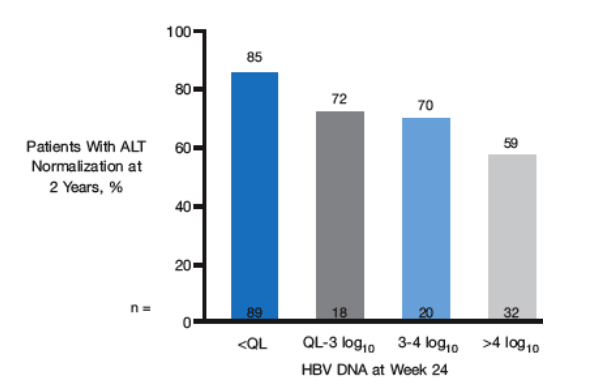
at 2 years (Figure 3). Rates of ALT normalization at 2 years were similarly high for telbivudine-treated patients who achieved maximal viral suppression at week 24 (Figure 4).
Figure 3. Relationship Between the Degree of Week 24
Viral Suppression and Rates of Serum HBV DNA PCR-Nondetectability
at 2 Years in Telbivudine-Treated Patients
83% of patients on telbivudine with undetectable HBV DNA at week 24 had undetectable HBV DNA at 2 years; 75% if HBV DNA was <1,000 c/ml at week 24; 40% if HBV DNA was 1,000 to 10,000, and 12% if HBV DNA was >10,000 c/ml at week 24.
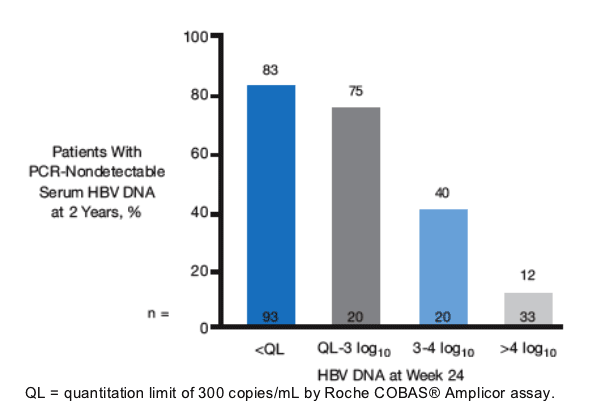
Figure 4. Relationship Between the Degree of Week 24
Viral Suppression and Rates of ALT Normalization at 2
Years in Telbivudine-Treated Patients
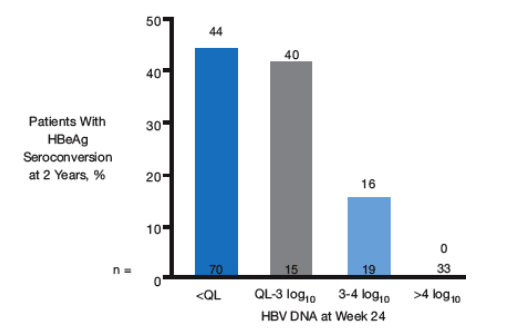
For HBeAg-positive patients treated with telbivudine who had HBV DNA levels ≦300 copies/mL at week 24, the rate of seroconversion at 2 years was 44% (Figure 5).
Figure 5. Degree of Week 24 Viral Suppression Affects Rate of HBeAg Seroconversion at 2 Years in HBeAg-positive Patients Treated with Telbivudine
44% of patients on telbivudine with undetectable HBV DNA at week 24 achieved HBeAg seroconversion at week 24. If viral load was 3-4 log, 16% had HBeAg seroconversion; QL-3 log: 40%. If >4 log: 0.
Safety
Both treatments were generally well tolerated. Reported adverse events were generally mild and transient, occurring at similar rates in both treatment groups (Table 4). The most frequently reported adverse events included nasopharyngitis, fatigue, and upper respiratory tract infection. One patient in the telbivudine group developed a polymyositis that was not attributed to the study drug; the event abated with continued treatment.
Grade 3 or 4 elevations of ALT and aspartate aminotransferase (AST) were more frequent in lamivudine recipients than in telbivudine recipients (Table 5). Grade 3 or 4 CK elevations, not requiring treatment modification, were more common with telbivudine than with lamivudine (Table 5).
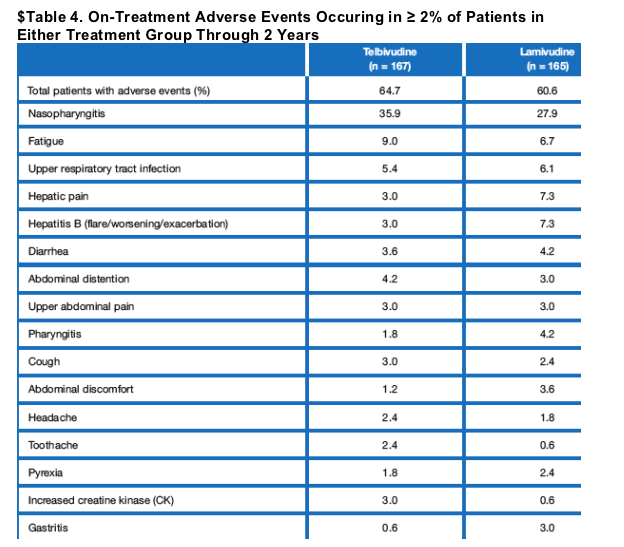
Table 5. New Onset Grade 3/4 Laboratory Abnormalities During the On-Treatment Period*
CK (creatinine kinase) >/= 7.0 x ULN: 11.4% for telbivudine, 3.6% for LAM. Amylase >3.0 x ULN: 0.6% for telbivudine, 0 for LAM. Lipase >2.5 x ULN: 1.8% in both groups.

|
| |
|
 |
 |
|
|