 |
 |
 |
| |
A Randomized Trial of Telbivudine vs Adefovir for HBeAg-Positive Chronic Hepatitis B: Efficacy Through Week 76, Predictors of Response and Effects of Switching to Telbivudine
|
| |
| |
Reported by Jules Levin
DDW, May 19-24, 2007, Wash DC
N. Bzowej1; P. Marcellin2; H.L.Y. Chan3; C.L. Lai4; M. Cho5; J. Heathcote6; Y.M. Moon7; Y.C. Chao8; R. Myers9; G. Harb10; N. Brown11
1Sutter Health, San Francisco, CA; 2Hopital Beaujon, Clichy, France; 3Prince of Wales Hospital, Hong Kong, China; 4University of Hong Kong, Hong Kong, China; 5Pusan National University Hospital, Busan, South Korea; 6Toronto Western Hospital, Toronto, ON, Canada; 7Severance Hospital, Seoul, Korea; 8Tri-Service General Hospital, Taipei, Taiwan; 9University of Calgary, Calgary, AB, Canada; 10Novartis Pharmaceuticals, Corp., East Hanover, NJ, USA; 11Idenix Pharmaceuticals, Cambridge, MA, USA
Summary and Conclusions
Telbivudine provided greater and more consistent viral suppression compared with adefovir after 24 weeks of treatment in patients with HBeAg-positive chronic hepatitis B.
Jules' notes: after 24 weeks (primary analysisbefore patients receiving adefovir could switch to telbivudine) HNB DNA decrease was -6.29 log c/ml for telbivudine vs -4.92 for ADV (p<0.001); a significant difference was evident at week 2. Serum HBV DNA was PCR-negative at week 24 was greater for patients receiving telbivudine (38% vs 12%, p<0.001). Baseline viral loads were about 9.5 logs. After 1 year HBV DNA decrease was -6.55 in telbivudine group (n=45), -5.72 in ADV group (n=44) (p 0.11); 58% in telbivudine group and 39% in ADV group were PCR-negative (300 copies/ml (Roche COBAS Amplicor). 77% in telbivudine group, 81% in ADV group had ALT normalization. HBeAg loss: 31% for telbivudine, 20% adefovir; HBeAg seroconversion: 27% telbivudine, 18% adefovir. These 1 year differences were not statistically significant. At week 76, 74% receiving continuous telbivudine were PCR-negative and HBV DNA decrease was -7.42 log c/ml. There was no drug resistance report in the poster but resistance analyses were reported from the large pivotal international GLOBE trial at DDW: preliminary cumulative resistance at 2 years, 17.8% for telbivudine, 30% for lamivudine in HBeAg+; 7.3% for telbivudine, 16.6% for lamivudine in HBeAg-negative. Of interest, patients who achieved <10,000 c/ml by week 24 had good responses after 1 year, see results below (40-50% HBeAg seroconversion; 90-95% PCR-negative; 90-95% ALT normalization).
Patients treated continuously with telbivudine, or switched to telbivudine from adefovir at week 24, showed significantly lower residual viral load at 1 year, and less treatment failure.
A rapid and marked incremental reduction in serum HBV DNA was evident in patients who were switched from adefovir to telbivudine at weeks 24 or 52. This was particularly evident among patients with suboptimal response to adefovir.
Virologic responses to telbivudine were maintained through 76 weeks of continuous treatment.
Consistent with previous findings for telbivudine and lamivudine,4,5 efficacy outcomes at 1 year were associated with viral load at week 24 for adefovir and telbivudine, with similar patterns observed for the combined treatment groups and for telbivudine and adefovir responses analyzed separately.
Both treatments were well tolerated.
Further follow-up of patients from this and other clinical trials will assess the longer term relationships between early viral suppression and treatment responses. Future studies of suboptimal responders may determine whether early treatment modification, using drugs with complementary resistance profiles, can improve subsequent outcomes.
Background
Substantial evidence supports the concept that treatment-induced suppression of hepatitis B virus (HBV) replication is associated with improved clinical outcomes.1-3 Recent studies have demonstrated that longer-term efficacy outcomes and the risk of resistance are related to the degree of viral suppression achieved within the first 24 weeks of therapy.4,5 Telbivudine has shown superior antiviral efficacy and comparable safety compared with lamivudine in phase III trials.6 Although adefovir has been shown to have antiviral efficacy superior to placebo,7,8 25% of nucleoside-naive hepatitis B e antigen (HBeAg)-positive patients showed a reduction of < 2.2 logs after 48 weeks of treatment with adefovir.9 With an increasing array of treatment options for chronic hepatitis B (CHB), there is a need for clinical trials that directly compare available agents.
The telbivudine 018 study is a randomized, multicenter, open-label clinical trial that compared the safety and antiviral efficacy of telbivudine and adefovir at 24 weeks of treatment, followed by a comparison of efficacy and safety outcomes after 1 year of treatment in patients treated with telbivudine or adefovir, or adefovir for 24 weeks followed by a switch to telbivudine for the remaining 28 weeks. Most patients, regardless of initial treatment group, elected to receive open-label treatment with telbivudine in the 022 extension study.
This report describes efficacy responses at weeks 24 and 52 of the 018 study and a exploratory analysis of week 76 data from the ongoing 022 study. We also present a retrospective analysis of relationships between early virologic responses in the telbivudine 018 study (assessed as HBV DNA levels at 24 weeks) and efficacy responses at 1 year in patients treated with telbivudine or adefovir.
Study Design
Eligible patients (n = 135) were adult males or females with compensated CHB who were HBsAg-positive and HBeAg-positive, with serum HBV DNA >6 log10 copies/mL by COBAS Amplicor PCR assay, and serum alanine aminotransferase (ALT) levels 1.3-10 times the upper limit of normal (ULN).
Patients in the 018 study were randomized (1:1:1) to receive telbivudine 600 mg/day for 1 year (Group A), adefovir 10 mg/day for 1 year (Group B), or adefovir 10 mg/day for 24 weeks followed by a switch to telbivudine 600 mg/day for the remaining 28 weeks of study treatment (Group C) (Figure 1).
After week 52, 121 patients received open-label telbivudine treatment in the 022 study, without treatment interruption. After 76 weeks of treatment (combined 018 + 022 studies), virologic responses were assessed, comparing patients maintained on continuous telbivudine for 76 weeks with patients switched from adefovir to telbivudine at week 52.
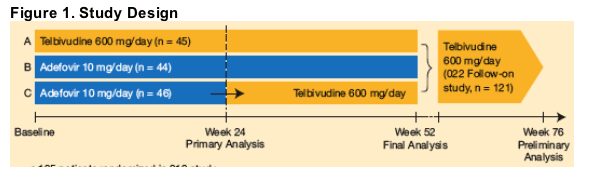
--135 patients randomized in 018 study
--121 patients received telbivudine in 022 follow-on study
Serum samples were collected for analysis at baseline and at weeks 2, 4, 8, 12, 16, 24, 32, 40, 48, and 52 (018 study), 60, 68 and 76 (022 study).
Serum HBV DNA was quantified by COBAS Amplicor HBV Monitor assay (Roche Molecular Systems; Branchburg, NJ; lower limit of detection = 300 copies/mL).
Study Endpoints
Primary efficacy endpoint:
-- Serum HBV DNA reduction at week 24
Key secondary efficacy endpoints (assessed at week 24, week 52 [018] and week 76 [022]):
-- Log10 reduction of serum HBV DNA
-- Clearance of HBV DNA to polymerase chain reaction (PCR) nondetectable levels (<300 copies/mL)
-- ALT normalization
HBeAg loss and seroconversion
Treatment failure and resistance
-- Viral breakthrough: increase of serum HBV DNA to ≥ 5 log10 copies/mL on two consecutive visits for patients in whom serum HBV DNA had previously been suppressed to below that level, or return of HBV DNA levels to within 1 log10 relative to baseline for patients whose serum HBV DNA declined at ≥ 2 log10 copies/mL from baseline (but not to < 5 log10 copies/mL on two consecutive visits
-- Resistance: documented resistance mutations (DNA sequencing of the reverse transcriptase domain of the HBV polymerase gene) in patients with viral breakthrough
-- Primary treatment failure: completion of at least 24 weeks of treatment without achieving two consecutive serum HBV DNA values < 5 log10 copies/mL
After study completion, suboptimal response was defined for exploratory analyses as serum HBV DNA level remaining ≥ 3 log10 copies/mL at week 24, consistent with reports of greater resistance and poorer efficacy outcomes for patients with this level of residual viremia.4,5 We analyzed efficacy outcomes at 1 year for patients in whom HBV DNA levels were < or > 3 log10 copies/mL at week 24.
RESULTS
Baseline Characteristics
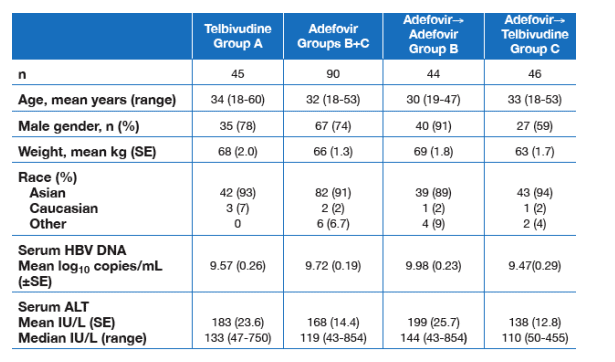
The intent-to-treat population comprised 135 adults with HBeAg-positive CHB, randomized to receive telbivudine 600 mg/day for 1 year (n = 45), adefovir 10 mg/day for 1 year (n = 44); or adefovir for 24 weeks followed by a switch to telbivudine for the remaining 28 weeks of the study (n = 46). Baseline demographic and disease parameters were similar among treatment groups (Table 1).
Table 1. Baseline Demographics and Disease Characteristics
Mean HBV DNA was 9.57 log c/ml in telbivudine group A, 9.72 log c/ml in adefovir groups A+B, 9.98 log c/ml in the ADV to ADV group B, and 9.47 log c/ml in the ADV to telbivudine group B. Mean ALT was 138 to 199. 93% of patients were asian, 32 yrs old.
Primary Analysis at Week 24
Telbivudine treatment (Group A) resulted in a significantly greater reduction in mean serum HBV DNA compared with adefovir (Groups B + C; -6.29 vs -4.92; P < 0.001). A significant difference between the two treatment arms was evident by week 2, and was maintained through week 24. Serum HBV DNA was PCR-negative at week 24 in a significantly greater fraction of patients receiving telbivudine compared with adefovir (38% vs 12%; P < 0.001).
Results at 1 Year
Among patients treated with adefovir initially and then switched to telbivudine at week 24 (Group C), serum HBV DNA levels fell rapidly following the switch to telbivudine, and were nearly identical to those in the telbivudine group (Group A) within 8 weeks (by week 32). Residual viral load at week 52 was significantly lower (P < 0.001) for patients treated with telbivudine (Group A), or switched to telbivudine at week 24 (Group C), compared with patients treated with adefovir for 52 weeks (Group B) (Figure 2, Table 2).
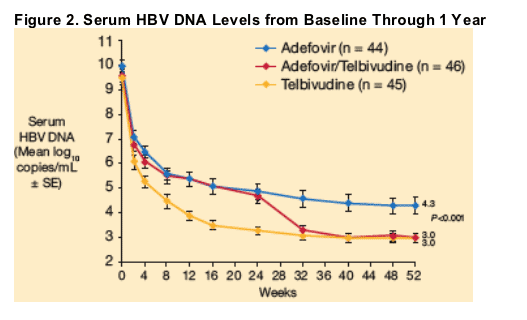
Table 2. Efficacy at 1 Year
HBV DNA decrease from baseline: -6.55 log c/ml for telbivudine (n=450 vs -5.72 for adefovir (n=44); for patiwnts switching at week 24 from ADV to telbivudine, -6.44.
HBV DNA PCr-negative: 58% telbivudine; 39% adefovir; 54% adefovir switch to telbivudine.
ALT normalization: 77% telb; 81% ADV; 85% ADV switch to telb.
HBeAg loss: 31% telb; 20% ADV; 26% ADV to telb switch.
HBeAg seroconversion: 27% telb; 18% ADV; 24% ADV to telb switch.
None of the above differences were statistically significant.
Primary treatment failures: 2% telb; 32% ADV; 11% ADV switch to telb.
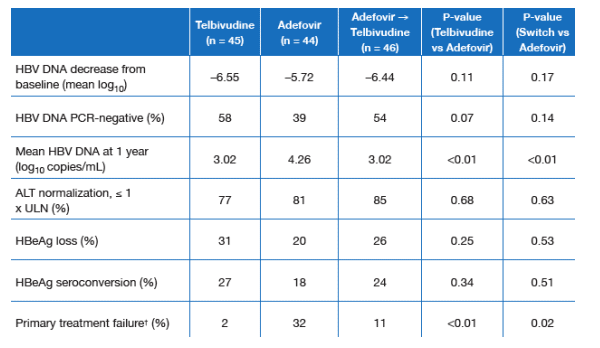
Primary treatment failure (serum HBV DNA levels never < 5 log10 copies/mL) was significantly more frequent in adefovir recipients compared with telbivudine recipients or patients switched to telbivudine after week 24 (Table 2). Serum HBV DNA was PCR-negative in 58% of telbivudine recipients, compared with 39% of adefovir recipients (P = 0.07) and 54% of patients in the switch group. HBeAg loss and seroconversion were more frequent in the telbivudine-treated groups compared with the adefovir group; however, these differences were not statistically significant. Rates of ALT normalization were similar in the three groups.
The incremental antiviral effect associated with switching from adefovir to telbivudine at week 24 was greater in suboptimal responders (HBV DNA levels ≥ 3 log10 copies/mL at week 24) than in the overall group of adefovir patients who switched to telbivudine. At week 24, 78% of patients in the adefovir group had viral load that remained ≥ 3 log10 copies/mL. Of this group, patients who switched to telbivudine at week 24 displayed a further 2.1 log10 mean reduction in HBV DNA between week 24 and 1 year, compared with 0.8 log10 for patients who remained on adefovir (Figure 3).
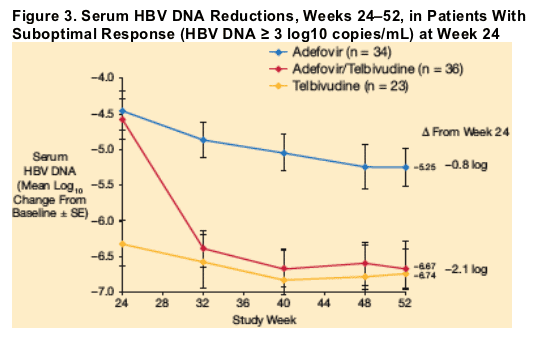
Results at Week 76 (022 Study)
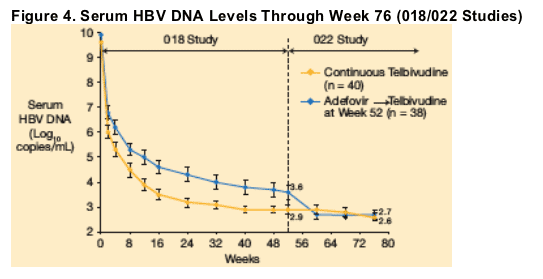
At week 52, residual HBV DNA levels in patients receiving continuous telbivudine (n = 40) were lower than those who had received continuous adefovir. Mean serum HBV DNA levels declined from 3.6 to 2.7 log10 copies/mL between weeks 52 and 76 in patients who switched from adefovir to telbivudine at week 52 (n = 38) (Figure 4).
Among patients who received 76 weeks of continuous telbivudine (n = 40), mean serum HBV DNA levels declined from 2.9 to 2.6 log10 copies/mL between weeks 52 and 76.
The proportion of patients with PCR-negative serum HBV DNA at week 52 was higher in patients who had received telbivudine in the 018 study, compared with adefovir. At week 76, following 24 weeks of telbivudine in the 022 study, similar proportions of patients were PCR-negative (Figure 5).
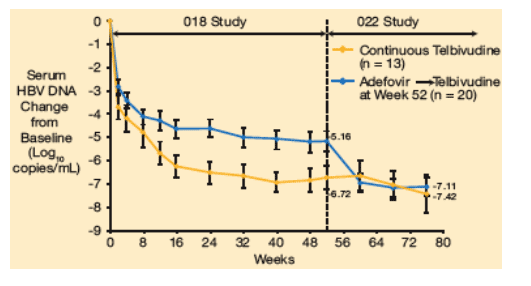
Among the 26 adefovir recipients with persistent suboptimal response (HBV DNA remained ≥3 log10 copies/mL at week 52), a rapid incremental decrease in serum HBV DNA was observed after switching to telbivudine at week 52 (Figure 6). A further mean 1.95 log10 copies/mL reduction of serum HBV DNA levels occurred between weeks 52 and 76 (n = 20).
Relationship of Week 24 Viral Load to Efficacy Responses at 1 Year
Among patients who received 1 year of continuous adefovir or telbivudine treatment (Groups A and B), the highest rates of efficacy outcomes at 1 year occurred in patients with serum HBV DNA levels < 3 log10 copies/mL at week 24 (Figures 7 and 8).
Figure 5. PCR-Negative HBV DNA at Weeks 52 and 76 in Patients Receiving Telbivudine Treatment in the 022 Study
59% in continuous relbivudine group had PCR nondetectable at week 52, and 74% at week 76.
In the ADV switch group 45% had undetectable at week 52 and 76% at week 76.
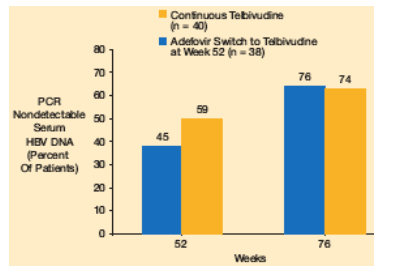
Figure 6. Serum HBV DNA Change in Patients With Suboptimal Response (≥3 log10 copies/mL HBV DNA) at Week 52
At week 52 HBV DNA decrease was -6.72 log in continuous telbivudine group and -5.16 log in adefovir to telbivudine group. After 80 weeks, HBV DNA decrease was -7.42 in the conrinuous telbivudine group and -7.11 in the adefovir switch to telbivudine group.
Relationship of Week 24 Viral Load to Efficacy Responses at 1 Year
Among patients who received 1 year of continuous adefovir or telbivudine treatment (Groups A and B), the highest rates of efficacy outcomes at 1 year occurred in patients with serum HBV DNA levels < 3 log10 copies/mL at week 24 (Figures 7 and 8).
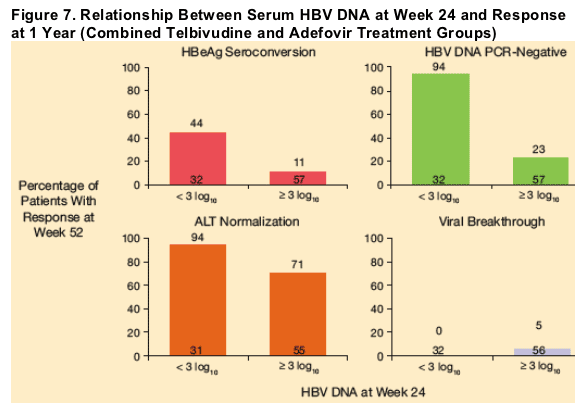
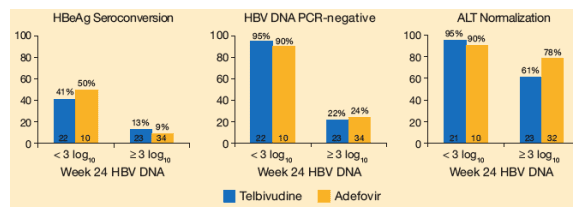
Figure 8. Relationship Between Serum HBV DNA at Week 24 and Response at 1 Year (Telbivudine vs Adefovir)
RESPONSE AT WEEK 52
Of those patients taking telbivudine and who achieved <3 logs at week 24 (<10,000 c/ml), 41% had HBeAg seroconversion, 95% were HBV DNA PCR-negative, and 96% had ALT normalization.
Of those patients taking ADV and who had <3 log at week 24, 50% haf HBeAg seroconversion, 90% were PCR-negative, and 90% had ALT normalization.
The three viral breakthroughs in this study all occurred in patients with serum HBV DNA levels > 4 logs at week 24. Further resistance analyses are ongoing.
Similar relationships between week 24 serum HBV DNA levels and week 52 outcomes were observed for the telbivudine and adefovir treatment groups (Figure 8). However, the greater overall antiviral effect of telbivudine at week 52, compared with adefovir (Table 2), may be attributable to the significantly greater proportion of telbivudine recipients with serum HBV DNA levels below 3 log10 copies/mL at week 24 (49% vs 22%, P < 0.01).
Safety
Both drugs were well tolerated with similar clinical adverse event profiles and no drug-attributed serious adverse events. Adverse events for both agents were primarily those associated with viral respiratory infections and gastrointestinal complaints. Grade 3/4 neutropenia was observed in two patients, one a telbivudine recipient, the other, a patient in the switch group; both events resolved at follow-up 6 days later, with continued treatment.
Limitations
The relationship between early virologic response and treatment outcomes at 1 year was analyzed retrospectively. The telbivudine 018 study was powered for week-24 analysis, with limited statistical power for 1-year (3 group) comparisons. In addition, data from the 022 follow-on study (beyond week 52) are uncontrolled.
References
1. Gauthier J, Bourne EJ, Lutz MW, et al. Quantitation of hepatitis B viremia and emergence of YMDD variants in patients with chronic hepatitis B treated with lamivudine. J Infect Dis. 1999;180:1757-1762.
2. Yuen MF, Sablon E, Hui CK, et al. Factors associated with hepatitis B virus DNA breakthrough in patients receiving prolonged lamivudine therapy. Hepatology. 2001;34:785-791.
3. Lai CL, Leung N, Teo EK, et al. A 1-year trial of telbivudine, lamivudine, and the combination in patients with hepatitis B e antigen-positive chronic hepatitis B. Gastroenterology. 2005;129:528-536.
4. Lai CL, Gane E, Liaw YF, et al. Maximal early HBV suppression is predictive of optimal virologic and clinical efficacy in nucleoside-treated hepatitis B patients: scientific observations from a large multinational trial (the GLOBE study) [Abstract]. Hepatology. 2005;42:232A-233A.
5. Di Bisceglie AM, Lai CL, Gane E, et al. Telbivudine GLOBE trial: maximal early HBV suppression is predictive of optimal two-year efficacy in nucleoside treated hepatitis B patients [Abstract]. Hepatology. 2006;44(4 suppl 1):230A.
6. Lai CL, Gane E, Liaw Y-F, et al. Telbivudine (LdT) vs lamivudine for chronic hepatitis B: first-year results from the international phase III GLOBE trial [Abstract]. Hepatology. 2005;42(4 suppl 1):748A.
7. Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, et al. Adefovir dipivoxil for the treatment of hepatitis B e antigen-negative chronic hepatitis B. N Engl J Med. 2003;348:800-807.
8. Marcellin P, Chang TT, Lim SG, et al. Adefovir dipivoxil for the treatment of hepatitis B e antigen-positive chronic hepatitis B. N Engl J Med. 2003;348:808 816.
9. Durantel S, Werle B, Durantel D, et al. Different profiles of response to adefovir dipivoxil and factors that may influence response in patients with chronic hepatitis B [Abstract]. Hepatology. 2004;40:654A.
|
| |
|
 |
 |
|
|