 |
 |
 |
| |
12 weeks VX-950 Therapy
|
| |
| |
Reported by Jules Levin
At DDW, May 2007, Vertex researchers reported results from computer simulation modeling to underscore that 12 weeks therapy with VX-950 can clear HCV. Research studies of pegIFN+RBV have shown that for some patients, those with a Rapid Viral Response (week 4 undetectable), 14-16 weeks therapy may be adequate to clear HCV. So, why wouldn't it be likely that for many patients taking VX-950 + peg/RBV 12 weeks might be adequate to clear HCV? I think it will occur. Of course ongoing research studies will tell us how many patients and for whom this can occur, and identify predictive factors; very rapid response at week 4 will predict 12 week response. The slide presentation by Vertex at DDW is below and reviews the VX950 monotherapy data showing that HCV RNA declines quickly in patients due to the potency of the drug. The slide presentation reviews viral decay in patients with wild-type and low level HCV protease inhibitor drug resistance. Viral decay in these two patient populations may be different and call for different duration of therapy. Nonetheless, Vertex is exploring both 12 weeks and 24 weeks duration of therapy. Study research in treatment-experienced patients is ongoing. There is theoretical data suggesting that treatment with VX950 in peg/RBV nonresponders might re-sensitize these patients to peg/RBV. In other words, the mechanism in these patients that prevents initial response to peg/RBV might be reversible by VX950 due to the way in which the HCV protease inhibitor works in the body. This remains theoretical until we see clinical results from the study in patients. Still, when and if VX950 makes it to the marketplace, in the pharmacy after FDA approval, this will represent a major advance in therapy for patients. VX950 will be the first oral antiviral for HCV and will be used in combination with peg/RBV. Numerous additional oral HCV drugs are in development. Several HCV polymerase inhibitor drugs and protease inhibitor drugs are being developed. There will be a time in the near future, perhaps 5 years, when there will be several oral HCV drugs to combine with pegIFN and perhaps still with RBV. When this happens therapy response rates will improve even more. There will be a time in the near future when Sustained Viral Response rates, HCV clearance, will I think be 70-90% for all patients, including African-Americans, genotype 1, and HIV coinfected, the hardest to treat patient population.
Jules Levin
Author Conclusions:
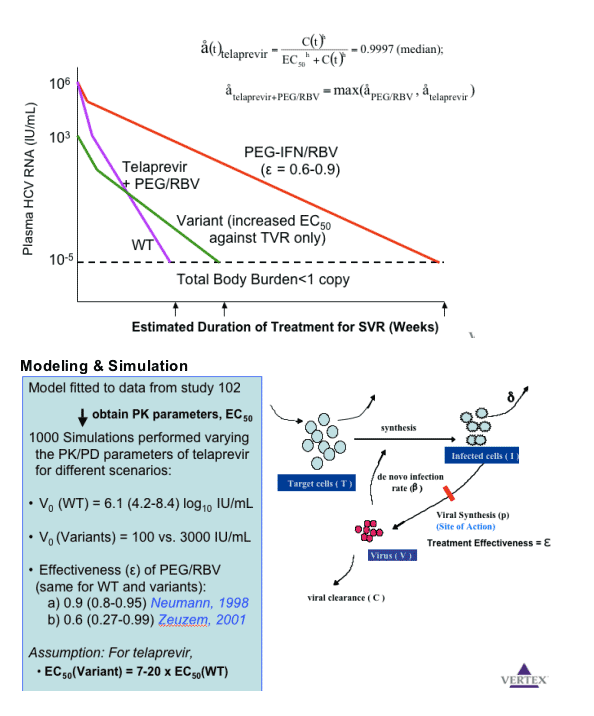
The results of these simulations suggest that
-- Wild-type virus may be eradicated by telaprevir within 10 weeks
-- Telaprevir-resistant variants (7-20 fold increase in IC50) may be eradicated mainly by PEG-IFN/RBV within 10-24 weeks (depending on the effectiveness)
Ongoing clinical trials are evaluating telaprevir-based regimens of 12-24 weeks
Further refinements to the model will be made as more clinical data become available. Modeling supports the 10-24 week approach, hope to suppress hcv.
Abstract
Predicting HCV Treatment Duration with an HCV Protease Inhibitor Co-administered with Peg-IFN/RBV by Modeling both Wild-type Virus and Low Level Resistant Variant Dynamics
V. Garg1; A. Khunvichai1; H. Chu1; J. G. McHutchison2; E. Lawitz3; M. Rodriguez4; T. Kieffer1; J. Alam1
1. Vertex Pharmaceuticals, Inc, Cambridge, MA, USA.
2. Duke Clinical Research Institute, Durham, NC, USA.
3. Alamo Medical Research, San Antonio, TX, USA.
4. Fundacion de Investigacion de Diego, Santurce, Puerto Rico.
Background: HCV protease inhibitors, such as telaprevir, inhibit viral replication and have induced marked decline of plasma HCV RNA titers in genotype-1-infected (GT-1) patients. Viral dynamic (VD) modeling assuming homogenous wild-type (WT) viral population suggested that protease inhibitors may significantly shorten treatment duration required to eradicate the virus (Chu, 2005 AASLD #1260). The objective of this work was to extend the previous empirical model to account for the presence of both WT virus and low-level telaprevir-resistant variants and estimate the required treatment duration.
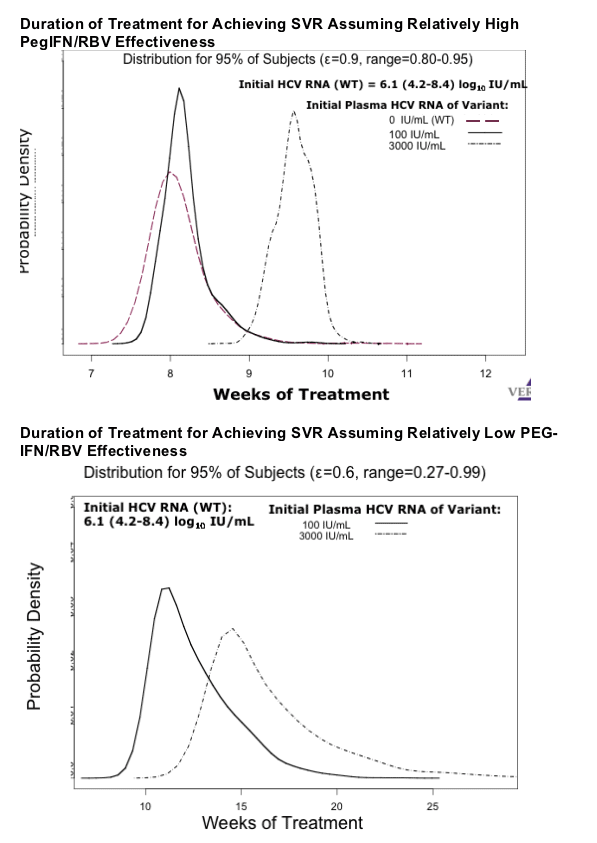
Methods: Plasma HCV RNA data from 12 treatment-naive, GT-1-infected patients dosed for 4 weeks with telaprevir co-administered with peginterferon-alfa-2a (Peg-IFN) and ribavirin (RBV) (Lawitz, 2006 DDW #686F) were analyzed using a VD model (Neumann, Science 1998) incorporating the pharmacokinetics (PK) of telaprevir. Simulations were performed with model parameter values obtained from VD model (ε, Õ, etc.) and PK parameters, for patients with predominately WT virus and low levels of resistant variants (assuming an initial variant level of 100-3000 IU/mL, a variant IC50 value ~7-to 20-fold increase from WT, and similar fitness between resistant variants and WT (Sarrazin, 2005 AASLD #LB06)). Because resistant variants were shown to be sensitive to IFN or RBV in vitro (Lin, 2006 AASLD #89) the effectiveness of Peg-IFN/RBV in blocking virion production was assumed to range 80%-95% against both WT and variant, consistent with clinical observations.
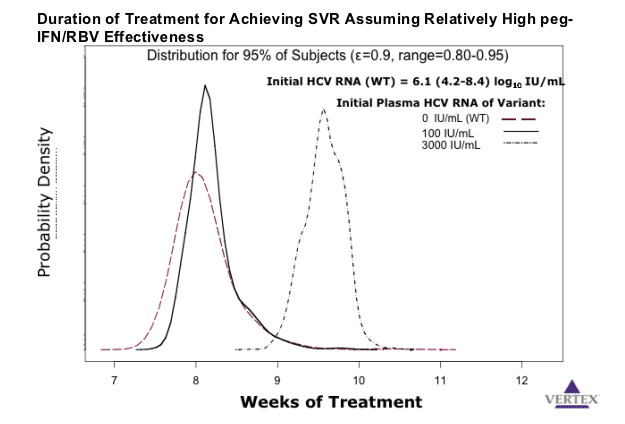
Results: The results of the simulations (Figure 1) indicate that treatment duration with co-administration of telaprevir with Peg-IFN/RBV required to suppress WT and low-level resistant variants below 1 copy (total body burden) is less than 12 weeks.
Conclusions: These results indicate that even in the presence of resistant variants, co-administration of a potent protease inhibitor with Peg-IFN/RBV may result in rapid viral eradication, allowing a substantial reduction in treatment duration from the current 48 weeks. The model supports the study of telaprevir-based treatment with Peg-IFN/RBV for 12 weeks. This hypothesis is being tested in ongoing clinical trials.
Predicting HCV Treatment Duration with Telapravir (VX-950) + Peg-IFN/RBV by Modeling Both Wild-Type and Low level Resistant Variant Dynamics
V Garg (Vertex, A Khunvichai, H-M Chu, JG McHutchison, E Lawitz, M Rodriguez, T Kieffer, J Alam
DDW, May 21, 2007
SLIDES
Earlier HCV RNA Clearance Equals Shorter Treatment
Several studies find that the earlier you clear HCV (reduce to undetectable) shorter duration of HCV therapy is required.
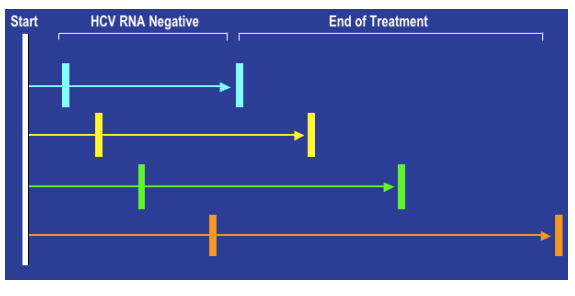
Berg T, et al. Gastroenterology. 2006;130:1086. Mangia, et al. N Engl J Med. 2005;352:2609. Dalgard O, et al. Hepatology. 2005;40:1260. Sanchez-Tapias JM, et al. Gastroenterology. 2006;131:451. von Wagner et al. Gastroenterology. 2004;129:522. Zeuzem S, et al. J Hepatology. 2006;44:97.
Courtesy of Dr. I. Jacobson.
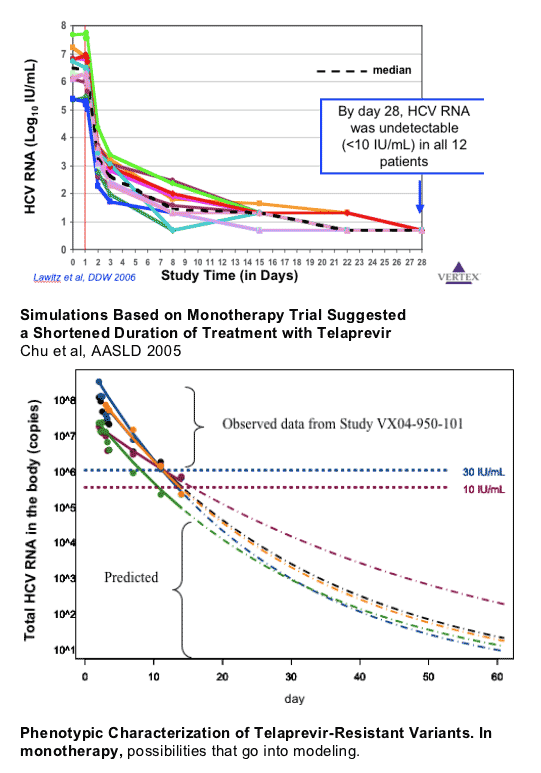
Rapid HCV RNA Response with Telaprevir-Based Regimens
Suggests Shorter Duration of Therapy is Feasible: in the 28 day monotherapy
study you can see VX950 quickly reduces HCV RNA
VX05-950-102: 28-day study in 12 HCV GT1, treatment-naive patients
Telaprevir (loading dose 1250 mg, then 750 mg q8h) + Peg-IFN and RBV

VX05-950-102: Viral Sequencing Results
In patients 1003 & 2002 R155K mutation emerged after stopping VX950 therapy suggesting that R155K may be a mutation to be concerned about and that for some patients longer duration of VX950 therapy may be required. Also note that in pt 1003 the percent of WT virus increased and percent of R155K virus decreased.
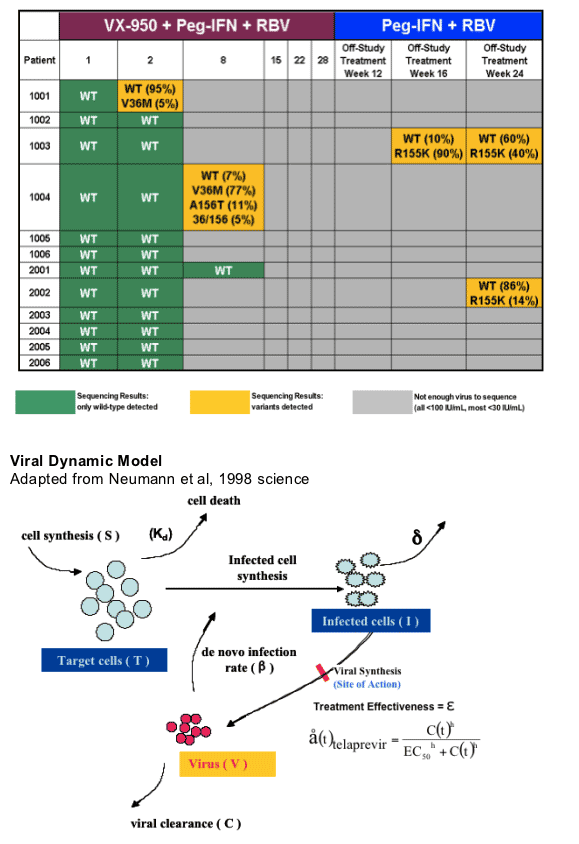
Estimating the Treatment Duration in Presence of
Both Wild-type and Resistant Variants
Slope decline .9 drop is 1 log drop. .9990 is 3 log drop in the first slope decline. .9999 is 4 logs. 1 is a monophasic decline, no need for 2nd slope decline. Telapravir drives down wild-type. What is involved in estimating duration: initial viral load and effectiveness of treatment. Concentration of drug matters over EC50-- equation just below. When you have variants the peg/rbv may be driving effectiveness.
|
| |
|
 |
 |
|
|