 |
 |
 |
| |
Darunavir Exhibits High Levels of In Vitro Antiviral Activity against Clinical Samples with Different Levels of Susceptibility to Atazanavir/Ritonavir
|
| |
| |
Reported by Jules Levin
5th European HIV Drug Resistance Workshop
Cascais, Portugal
March 30, 2007
M-P de Bethune1; L Bacheler2; S De Meyer1; B Wasikowski3; AR Rinehart4; and G Picchio5
1Tibotec BVBA, Mechelen, Belgium; 2VircoLab, Inc., Durham, NC, USA; 3xLeo, Inc., Raleigh, NC, USA; 4Tibotec Therapeutics, Bridgewater, NJ, USA; 5Tibotec Inc., Yardley, PA, USA
ABSTRACT
Background: Atazanavir/ritonavir (ATV/r) is used commonly in first- and secondline antiretroviral therapy. Following ATV/r failure, physicians are challenged with selecting an active protease inhibitor (PI) to construct the next regimen.We determined the likelihood of activity of 3 ritonavir-boosted PIs against a large number of recent clinical samples with decreasing levels of susceptibility to ATV/r.
Methods: A total of 78,978 samples were received at Virco for routine resistance testing between January 2004 and September 2006: 29,410 (37.2%) of these samples exhibited decreased susceptibility to at least 1 PI (FC>CCO1, vircoTYPE v. 4.0.01). These samples were further divided into 5 susceptibility groups (G1-G5) according to the relationship of predicted fold change (FC) in EC50 to ATV/r clinical cutoffs CCO1 (FC=2.7) and CCO2 (FC=32.9). FC values that fell into susceptibility groups
--G1 (FC
--G5 (FC>32.9, n=4,340)
represented samples with the highest and lowest susceptibility to ATV/r, respectively.
In order to further explore the resistance-response continuum, 3 intermediate susceptibility groups
--G2 (2.7< FC26.2, n=3,536);
--G3 (6.2< FC214.3, n=3,155); and
--G4 (14.3< FC232.9, n=2,953)
were defined by dividing the FC range between CCO1 and CCO2 on the log scale into 3 equal parts. The proportion of samples with predicted maximal response (FC< /=CCO1), predicted reduced response (RR) (CCO1< FC2CCO2) and predicted minimal response (FC >CCO2) to darunavir/r (DRV/r), lopinavir/r (LPV/r) and tipranavir/r (TPV/r) was determined for G1-G5 according to the respective vircoTYPE clinical cutoffs for each PI.
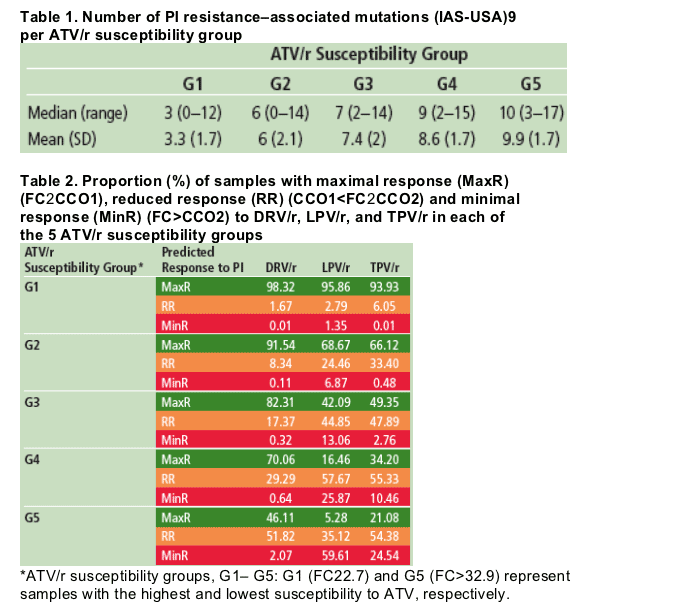
Results: Groups 1 through 5 had a mean number (±SD) of 3.3 (1.7), 6.0 (2.1),
7.4 (2.0), 8.6 (1.7), and 9.9 (1.7) PI resistance-associated mutations (IAS-USA,
October 2006), respectively.
The proportion of samples classified with minimal response to each PI in groups
G1, G2, G3, G4, and G5, respectively, were:
DRV/r -- (0.01%, 0.11%, 0.32%, 0.64%, 2.07%),
LPV/r -- (1.35%, 6.87%, 13.06%, 25.87%, 59.61%), and
TPV/r -- (0.01%, 0.48%, 2.76%, 10.46%, 24.54%).
The proportion of samples classified with maximal response in the same groups were:
DRV/r -- (98.32%, 91.54%, 82.31%, 70.06%, 46.11%),
LPV/r -- (95.86%, 68.67%, 42.09%, 16.46%, 5.28%) and
TPV/r -- (93.93%, 66.12%, 49.35%, 34.20%, 21.08%).
Conclusions: The proportion of samples classified with maximal response to
DRV/r, LPV/r, and TPV/r decreased as phenotypic susceptibility to ATV/r decreased. Within each of the 5 ATV/r susceptibility groups, more samples were classified with maximal response to DRV/r than TPV/r or LPV/r.
Data in the poster body have been updated since the abstract was originally submitted.
Introduction
Following failure with a first- or second-line protease inhibitor (PI), there is a need to identify a therapeutic agent with activity against virus with multiple PI resistance mutations
We determined the likelihood of activity of 3 ritonavir-boosted PIs (darunavir [DRV/r], lopinavir [LPV/r], and tipranavir [TPV/r]) against a large number of recent clinical samples with decreasing levels of susceptibility to atazanavir/ritonavir (ATV/r)
- A similar analysis for DRV has been presented previously, using the biological cutoff value of 2.4 for ATV/r.1 Clinical cutoffs for ATV/r on the vircoTYPE HIV-1 resistance analysis have been presented recently2 and are used for this new analysis
In the POWER 1 and 2 studies and the POWER 3 analysis, DRV co-administered with low-dose ritonavir (DRV/r) 600/100mg bid demonstrated significant and sustained efficacy with a low rate of gastrointestinal side effects. Forty-five percent of patients in the POWER 1 and POWER 2 studies combined, and 40% in the POWER 3 analysis, achieved viral load (VL) <50 copies/mL at week 243,4
DRV/r 600/100mg bid has been approved in the United States, Canada, Russia, Argentina, Switzerland, Australia and the European Union5 for the treatment of HIV infection in antiretroviral treatment-experienced adult patients.
Methods
78,978 samples were received at Virco for routine resistance testing (samples from research studies and clinical trials excluded) between January 2004 and September 2006 (Figure 1)
29,410 (37.2%) of these samples exhibited decreased susceptibility to at least 1 PI (fold-change [FC]>CCO1, vircoTYPE v. 4.1.00, Virco BVBA)
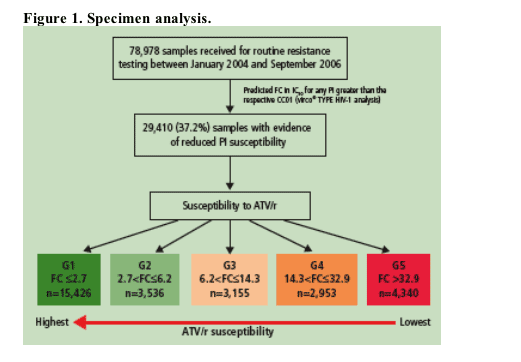
Group 1: FC< /=2.7 (n=15,426)
Group 2: 2.7< FC
Group 3: 6.2< FC< /=14.3 (n=3,155),
Group 4: 14.3< FC< /=32.9 (n=2,953)
Group 5: FC>32.9 (n=4,340)
Samples with reduced PI susceptibility were further divided into 5 ATV/r susceptibility groups (G1-G5) according to predicted FC in EC50 to ATV in relationship with clinical cutoffs for use of ATV: CCO1 (FC=2.7) and CCO2 (FC=32.9) (Figure 2)
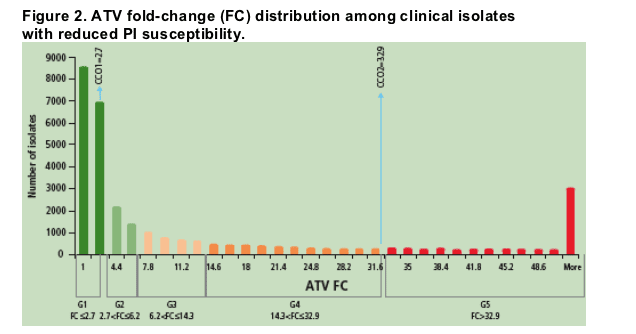
- FC values that fell into susceptibility groups G1 (FC22.7, n=15,426) and G5 (FC>32.9, n=4,340) represented samples with the highest and lowest susceptibility to ATV/r, respectively
- Because of the large interval between ATV/r CCO1 and CCO2, and in order to further explore the resistance-response continuum, 3 intermediate susceptibility groups, G2-G4, were defined by dividing the FC range between CCO1 and CCO2 on the log scale into 3 equal parts
Isolates with ATV/r FC>2.7 (ATV/r CCO1) do not necessarily represent isolates from patients who have failed ATV/r-containing regimens. Virco does not collect data on current or past antiretroviral regimens for isolates submitted for routine clinical resistance testing
The proportion of samples with predicted maximal response (MaxR) (FCCCO2) to DRV/r, LPV/r, and TPV/r was determined for G1-G5 according to the respective vircoTYPE clinical cutoffs for each PI:
- DRV/r: CCO1 = 3.4, CCO2 = 96.96
- LPV/r: CCO1 = 9.7, CCO2 = 56.17
- TPV/r: CCO1 = 1.2, CCO2 = 5.48
Results
The median number of IAS-USA PI resistance-associated mutations was higher in samples with a higher predicted FC to ATV (Table 1).
The proportion of samples classified with MaxR, RR, or MinR to each PI in groups G1, G2, G3, G4, and G5 is described in Table 2, and illustrated in Figure 3
- All three boosted PIs retained MaxR in >90% of the samples in G1
- In G2 (samples with ATV FC slightly greater than CCO1 for ATV/r), DRV/r retained MaxR for >90% of samples, while LPV/r and TPV/r retained MaxR in only two-thirds of the samples
- In all 5 ATV/r susceptibility subgroups, DRV/r showed the largest proportion of samples with MaxR, and the lowest proportion with MinR
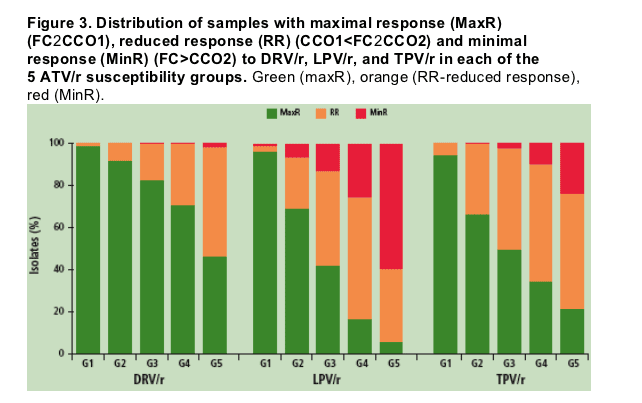
The proportion of samples across all 5 ATV/r susceptibility groups predicted to have MinR to DRV/r ranged from 0-2.1%, whereas the range for TPV/r and LPV/r were 1.4-59.6% and 0-24.5%, respectively
Conclusions
The proportion of samples classified with maximal response to DRV/r, LPV/r, and TPV/r decreased as predicted phenotypic resistance to ATV increased.
Among all categories of ATV/r resistance, the greatest proportion of samples were predicted to have maximal response to DRV/r compared with TPV/r and LPV/r.
Similarly, the lowest proportion of samples were predicted to have minimal response to DRV/r compared with TPV/r and LPV/r.
References
1. Staes M, et al. 15th International HIV Drug Resistance Workshop, June 13-17, 2006, Sitges, Spain. Poster 28.
2. Winters B, et al. 14th Conference on Retroviruses and Opportunistic Infections, February 5-28, 2007, Los Angeles, California. Poster 611.
3. Ruane P, et al. Frontiers in Drug Development of Antiretroviral Therapies, December 10-14, 2006, Cancun, Mexico. Poster 74.
4. PREZISTA (darunavir) Prescribing Information 2006, Tibotec Therapeutics, Bridgewater, NJ, USA. Available online:
http://www.prezista.com/docs/us_package_insert.pdf.
5. EMEA approved product information website. Available online: http://www.emea.eu.int/humandocs/Humans/EPAR/prezista/prezista.htm.
6. Winters B, et al. XV International HIV Drug Resistance Workshop, June 13-17,2006, Sitges, Spain. Poster 160.
7. Virco website: www.vircolab.com.
8. Bacheler L, et al. 4th European HIV Drug Resistance Workshop, March 29-31, 2006, Monte Carlo, Monaco. Poster 40.
9. Johnson VA, et al. Top HIV Med 2006;14:125-31.
|
| |
|
 |
 |
|
|